Advanced Chiral Synthesis of Rasagiline Mesylate: A Cost-Effective Route for Industrial API Manufacturing
The pharmaceutical industry is constantly seeking more efficient pathways to produce critical active pharmaceutical ingredients (APIs), particularly for neurodegenerative disorders like Parkinson's disease. Patent CN102010353B introduces a groundbreaking preparation method for Rasagiline Mesylate, a potent monoamine oxidase-B (MAO-B) inhibitor. This technology represents a significant paradigm shift from traditional resolution-based methods to a direct asymmetric synthesis strategy. By utilizing 1-indanone as a readily available starting material and introducing a chiral auxiliary early in the sequence, the process achieves high optical purity and yield while drastically simplifying the operational workflow. For global procurement teams and R&D directors, this innovation offers a compelling value proposition: a reliable pharmaceutical intermediates supplier can now offer this key compound with reduced dependency on expensive chiral pool resources. The method addresses long-standing bottlenecks in the supply chain, such as the high cost of R-1-indanamine and the low yields associated with racemic resolution, positioning it as a superior choice for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rasagiline has been plagued by economic and technical inefficiencies that hinder large-scale production. Prior art methods, such as those disclosed in earlier patents, often rely on the use of R-1-indanamine as a key starting material. This chiral amine is notoriously expensive and difficult to source in bulk quantities, creating a significant cost barrier for manufacturers. Furthermore, alternative routes that attempt to synthesize the amine from racemic precursors require a resolution step. This classical resolution approach is inherently wasteful, as it theoretically discards half of the produced material (the unwanted enantiomer), leading to a maximum yield of 50% before further processing losses occur. Additionally, direct alkylation strategies often suffer from poor selectivity, generating disubstituted tertiary amine byproducts that are difficult to separate, thereby reducing the overall reaction yield and complicating the purification process. These factors combined result in a lengthy, costly, and environmentally burdensome production cycle that is ill-suited for modern industrial demands.
The Novel Approach
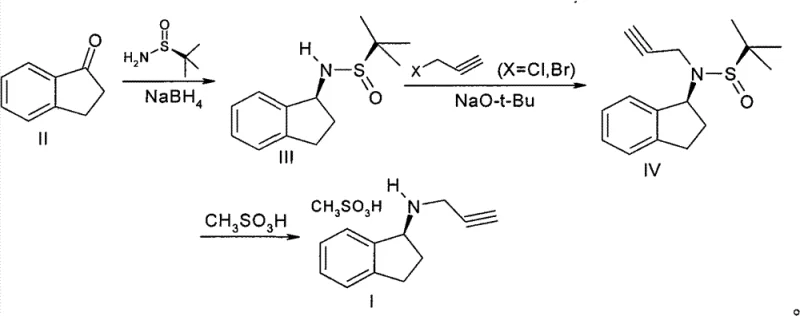
In stark contrast, the method described in Patent CN102010353B employs a clever asymmetric synthesis strategy that bypasses these traditional pitfalls. The core innovation lies in the use of 1-indanone, an inexpensive and abundant ketone, which undergoes a stereoselective reductive amination. By introducing a chiral sulfinamide auxiliary, the process dictates the stereochemistry of the resulting amine with high precision, eliminating the need for wasteful resolution steps. This route not only secures the desired chirality from the outset but also streamlines the synthesis into fewer operational steps. The subsequent N-alkylation with propargyl halide proceeds cleanly under optimized basic conditions, minimizing side reactions. Finally, the removal of the chiral auxiliary and salt formation with methanesulfonic acid yields the target product in high purity. This unique reaction scheme transforms the production landscape, offering a pathway that is both economically viable and technically robust for the commercial scale-up of complex chiral amines.
Mechanistic Insights into Asymmetric Reductive Amination
The heart of this technological advancement is the initial asymmetric reductive amination step, which sets the stereochemical foundation for the entire molecule. In this critical transformation, 1-indanone reacts with a chiral sulfinamide, typically (R)-tert-butyl sulfinamide, in the presence of a Lewis acid catalyst like titanium isopropoxide. This condensation forms a chiral N-sulfinyl imine intermediate, which acts as a rigid template for the subsequent reduction. When sodium borohydride is introduced, the hydride attack occurs selectively on one face of the imine double bond due to the steric bulk of the tert-butyl group on the sulfur atom. This facial selectivity ensures that the resulting amine possesses the desired (R)-configuration with exceptional fidelity. The mechanism effectively transfers chirality from the inexpensive auxiliary to the substrate, a strategy that is far more scalable than enzymatic resolution or chiral chromatography. By controlling the reaction temperature between 50°C and 100°C during imine formation and carefully managing the reduction conditions, the process maximizes diastereoselectivity, which directly translates to high enantiomeric excess in the final free amine after auxiliary removal.
Following the establishment of the chiral center, the synthesis proceeds through a nucleophilic substitution mechanism to install the propargyl group. The chiral amine intermediate, still protected by the sulfinyl group, acts as a nucleophile attacking 3-propargyl halide (chloride or bromide). The use of a strong, non-nucleophilic base such as sodium tert-butoxide is crucial here; it deprotonates the sulfonamide nitrogen, increasing its nucleophilicity without causing elimination side reactions on the propargyl halide. This step is conducted at moderate temperatures (30°C to 60°C) to balance reaction rate with selectivity. The final step involves the acidic hydrolysis of the sulfinyl group using methanesulfonic acid. This dual-purpose step not only cleaves the chiral auxiliary, regenerating the free amine, but simultaneously forms the stable mesylate salt. The precipitation of the product as a crystalline solid from the reaction mixture serves as a powerful purification drive, effectively excluding impurities and ensuring the final API intermediate meets rigorous quality standards without the need for column chromatography.
How to Synthesize Rasagiline Mesylate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and purity. The process begins with the condensation of 1-indanone and the chiral auxiliary, followed by reduction to generate the key chiral intermediate. This intermediate is then alkylated and finally converted to the salt form. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and temperature profiles for each stage, are critical for reproducibility. For R&D teams looking to adopt this technology, understanding the nuances of workup and crystallization is just as important as the reaction chemistry itself. The following guide outlines the essential phases of this streamlined manufacturing process.
- Perform asymmetric reductive amination of 1-indanone with a chiral sulfinamide auxiliary using titanium isopropoxide and sodium borohydride to obtain the chiral amine intermediate.
- Conduct N-alkylation of the chiral amine intermediate with 3-propargyl halide in the presence of a strong base like sodium tert-butoxide to form the protected propargyl amine.
- Remove the chiral auxiliary and form the final salt by reacting the intermediate with methanesulfonic acid in a suitable solvent system to crystallize pure Rasagiline Mesylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The shift from expensive chiral starting materials to commodity chemicals like 1-indanone fundamentally alters the cost structure of the supply chain. By eliminating the reliance on scarce chiral pools, manufacturers can secure a more stable and predictable supply of raw materials, mitigating the risk of price volatility associated with specialized reagents. Furthermore, the reduction in synthetic steps and the avoidance of inefficient resolution processes mean that less material is wasted, directly contributing to substantial cost savings in raw material consumption. The simplicity of the purification process, which relies on crystallization rather than complex chromatographic separations, also reduces the demand for expensive silica gel and solvents, further driving down the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The most significant economic advantage lies in the replacement of high-cost chiral amines with inexpensive achiral ketones. Traditional routes often incur heavy expenses purchasing R-1-indanamine, whereas this method builds the chirality in-house using a catalytic or stoichiometric amount of a reusable auxiliary. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove impurities, reducing the burden on downstream purification units. This efficiency translates to lower energy consumption and reduced waste disposal costs, making the overall manufacturing process significantly more lean and profitable compared to legacy methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of widely available starting materials. 1-indanone and propargyl halides are commodity chemicals produced by numerous global suppliers, ensuring that production is not bottlenecked by a single source of chiral feedstock. The robustness of the reaction conditions, which tolerate standard industrial solvents and reagents, means that the process can be easily transferred between different manufacturing sites without extensive re-optimization. This flexibility allows for diversified sourcing strategies and reduces the lead time for high-purity pharmaceutical intermediates, ensuring consistent delivery to downstream API manufacturers even during market fluctuations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for industrial expansion. The avoidance of column chromatography is a major win for green chemistry, as it eliminates tons of solid waste associated with silica disposal. The process relies on crystallization and filtration, unit operations that are easily scaled from kilograms to multi-ton batches in standard stainless steel reactors. Moreover, the high atom economy and reduced solvent usage align with increasingly strict environmental regulations, facilitating smoother regulatory approvals and reducing the carbon footprint of the manufacturing facility. This makes the technology not just chemically superior, but also a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding the practical implementation, purity profiles, and economic implications of this technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering a clear picture of what partners can expect when adopting this advanced synthetic route.
Q: How does this new method reduce costs compared to traditional resolution routes?
A: Traditional methods often rely on expensive chiral starting materials like R-1-indanamine or require inefficient resolution steps that discard 50% of the material. This novel route uses cheap, achiral 1-indanone and a recoverable chiral auxiliary, significantly lowering raw material costs and improving overall atom economy.
Q: What is the optical purity achievable with this synthetic route?
A: The process utilizes a highly selective asymmetric reduction step which ensures excellent stereocontrol. The patent data indicates that the optical purity (ee value) can reach above 99.5%, meeting the stringent requirements for pharmaceutical grade intermediates without the need for additional chiral purification steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex operations like column chromatography, relying instead on standard crystallization and filtration techniques. The use of common solvents and stable reagents makes it robust for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rasagiline Mesylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral synthesis technology for the global Parkinson's disease medication market. As a dedicated CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into a robust, GMP-compliant manufacturing process. Our facilities are equipped to handle the specific requirements of asymmetric synthesis, including moisture-sensitive reactions and precise temperature control needed for high enantioselectivity. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for optical purity and chemical identity.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this cost-effective route for Rasagiline Mesylate. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this new method for your specific volume requirements. We encourage you to contact us today to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your project timelines. Together, we can accelerate the availability of high-quality, affordable treatments for patients worldwide while optimizing your manufacturing economics.
