Revolutionizing Chiral Amine Synthesis: High-Efficiency Fructose-Derived Catalysts for Industrial Scale-Up
Introduction to Advanced Asymmetric Catalysis
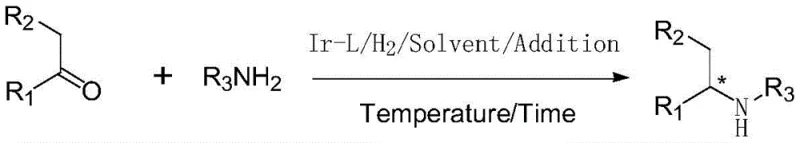
The landscape of chiral amine synthesis is undergoing a significant transformation driven by the need for more sustainable and cost-effective manufacturing processes. Patent CN111285775B introduces a groundbreaking methodology utilizing fructose-derived pyridyl alcohol chiral ligands for the asymmetric reductive amination of ketones. This technology addresses critical bottlenecks in the production of high-value agrochemical and pharmaceutical intermediates by leveraging renewable carbohydrate scaffolds to create highly efficient catalytic systems. Unlike traditional methods that often rely on expensive, non-renewable chiral phosphines, this approach utilizes a readily available fructose backbone, significantly lowering the raw material costs while maintaining high stereocontrol. The innovation lies in the unique coordination chemistry of the iridium-fructose complex, which facilitates direct reductive amination with exceptional turnover numbers, making it a viable candidate for large-scale industrial applications where cost and purity are paramount concerns for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines has been dominated by asymmetric hydrogenation of pre-formed imines or enamines, a process that often requires harsh conditions and multiple synthetic steps. Early attempts at direct asymmetric reductive amination, such as those utilizing ferrocene bisphosphine ligands reported in the late 1990s, suffered from prohibitively high catalyst loadings, often requiring catalyst-to-substrate ratios that were 100 times higher than those needed for simple imine hydrogenation. This inefficiency was largely due to catalyst deactivation caused by the coordination of substrate amines or the reduction of the ketone substrate to an undesired alcohol byproduct. Furthermore, many existing catalytic systems exhibit narrow substrate scopes, failing to perform effectively with aliphatic ketones or sterically hindered amines, which limits their utility in the diverse landscape of fine chemical manufacturing. These limitations result in increased production costs, complex purification workflows, and significant waste generation, posing challenges for procurement teams aiming to optimize margins.
The Novel Approach
The novel approach detailed in this patent overcomes these historical barriers by employing a specialized iridium catalyst system ligated with fructose-derived pyridyl alcohols. This methodology enables the direct conversion of ketones and amines into chiral amines in a single pot, eliminating the need for isolating unstable imine intermediates and thereby improving overall atom economy. The fructose-derived ligand provides a rigid chiral environment that effectively differentiates between enantiotopic faces of the intermediate imine, leading to enantiomeric excess values consistently exceeding 70 percent, with specific examples reaching up to 83 percent ee. Crucially, this system demonstrates remarkable catalytic activity, sustaining high turnover frequencies even at extremely low catalyst loadings, with substrate-to-catalyst ratios reaching as high as 10000:1 in optimized conditions. This leap in efficiency translates directly to reduced reliance on precious metals and simplified downstream processing.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Reductive Amination
The efficacy of this catalytic system stems from the intricate interplay between the iridium center and the multifunctional fructose-derived ligand. The ligand, characterized by a pyridyl alcohol moiety attached to a protected fructose scaffold, coordinates to the iridium precursor, likely forming a cationic or neutral active species depending on the counterions present. The hydroxyl group on the ligand may participate in hydrogen bonding interactions with the substrate or the titanium additive, creating a highly organized transition state that favors the formation of one enantiomer over the other. The presence of a titanium additive, such as titanium isopropoxide, is critical; it acts as a Lewis acid to accelerate the condensation of the ketone and amine into the imine intermediate while simultaneously scavenging the water byproduct, thus preventing catalyst hydrolysis and driving the equilibrium forward. This dual-activation strategy ensures that the rate of imine formation matches the rate of asymmetric hydrogenation, minimizing side reactions such as ketone reduction.
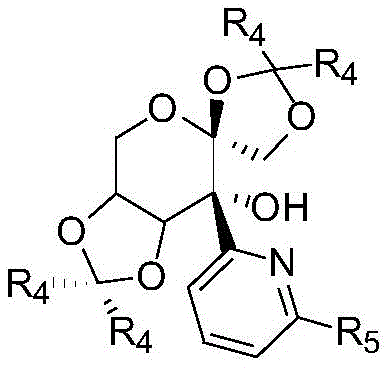
Furthermore, the steric bulk provided by the acetonide protecting groups on the fructose ring plays a pivotal role in enforcing stereoselectivity. By shielding specific quadrants around the metal center, the ligand forces the incoming hydride and the imine substrate to approach in a specific orientation, thereby dictating the absolute configuration of the newly formed chiral center. This level of control is essential for producing pharmaceutical intermediates where even trace amounts of the wrong enantiomer can be detrimental. The robustness of the ligand structure also contributes to the catalyst's longevity, allowing it to withstand the elevated temperatures and pressures required for difficult substrates without decomposing. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as solvent polarity and hydrogen pressure, to maximize both yield and optical purity for specific target molecules.
How to Synthesize Chiral Amines Efficiently
The synthesis of chiral amines using this patented technology involves a straightforward protocol that is amenable to standard high-pressure reactor setups commonly found in pilot and production plants. The process begins with the in-situ generation of the active catalyst by mixing the iridium precursor with the chiral ligand in a suitable solvent like toluene or dichloromethane under an inert atmosphere. Once the catalyst is formed, the ketone and amine substrates are introduced along with the titanium additive, and the mixture is subjected to hydrogen pressure ranging from 20 to 100 bar. The reaction temperature is typically maintained between 20 and 100 degrees Celsius, depending on the reactivity of the substrates, with reaction times varying from 1 to 24 hours. Detailed standardized synthesis steps and specific parameter optimizations for various substrates are provided in the technical guide below.
- Prepare the catalyst system by mixing an iridium-cyclooctadiene complex with the fructose-derived pyridyl alcohol chiral ligand in a solvent like toluene under nitrogen protection.
- Add the ketone substrate, amine source, and a titanium additive (such as titanium isopropoxide) to the reaction mixture.
- Pressurize the reactor with hydrogen gas (20-100 bar) and heat to 20-100°C for 1-24 hours to complete the asymmetric reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this fructose-derived catalytic system presents a compelling value proposition centered on cost stability and operational efficiency. The ability to operate at extremely low catalyst loadings, with substrate-to-catalyst ratios demonstrated up to 10000, drastically reduces the consumption of iridium, a precious metal subject to significant price volatility and supply constraints. This reduction in precious metal usage not only lowers the direct material cost but also simplifies the purification process, as there is less metal residue to remove from the final product to meet stringent regulatory limits. Additionally, the use of fructose, a renewable and abundant feedstock, for the ligand synthesis insulates the supply chain from the geopolitical risks often associated with synthetic chiral ligands derived from petrochemical sources. This shift towards bio-based catalytic components aligns with broader corporate sustainability goals and enhances the long-term reliability of the supply chain.
From a manufacturing perspective, the simplicity of the one-pot procedure eliminates the need for intermediate isolation and drying steps, which are often time-consuming and energy-intensive. This streamlined workflow reduces the overall batch cycle time and increases the throughput of existing production facilities without requiring significant capital investment in new equipment. The method's compatibility with continuous operation further enhances its industrial practicability, allowing for consistent quality and reduced variability between batches. Moreover, the high atom economy of the reductive amination process minimizes waste generation, reducing the burden on waste treatment facilities and lowering environmental compliance costs. These combined factors contribute to a more resilient and cost-effective manufacturing process, enabling suppliers to offer competitive pricing while maintaining high margins and reliable delivery schedules for critical agrochemical intermediates like the Metolachlor precursor.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reductive amination technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. By clarifying these points, we aim to facilitate informed decision-making for R&D and procurement teams evaluating this technology for their specific production needs.
Q: What are the advantages of using fructose-derived ligands over traditional phosphine ligands?
A: Fructose-derived ligands offer a renewable, cost-effective chiral pool source compared to expensive synthetic phosphines. They enable lower catalyst loadings (S/C up to 10000) and simplify the purification process by avoiding complex phosphine oxide byproducts.
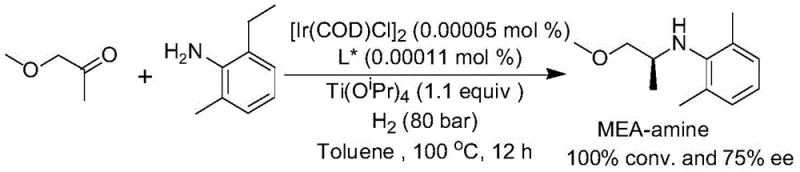
Q: Can this method be applied to the synthesis of Metolachlor intermediates?
A: Yes, the patent specifically highlights the synthesis of the fine Metolachlor intermediate (2-ethyl-6-methylaniline derivative) with excellent results, achieving 95% yield and 75% enantioselectivity at industrially relevant catalyst ratios.
Q: What role does the titanium additive play in this reaction?
A: The titanium additive, such as titanium isopropoxide, acts as a Lewis acid to activate the carbonyl group for imine formation and helps scavenge water produced during the condensation step, driving the equilibrium towards the desired chiral amine product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the fructose-derived iridium system in optimizing the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into robust manufacturing processes. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of handling sensitive organometallic chemistry, guaranteeing that every batch meets stringent purity specifications and enantiomeric excess requirements. We are committed to leveraging such innovative patents to deliver superior quality products that enhance the efficiency of our clients' supply chains.
We invite you to collaborate with us to explore the feasibility of integrating this cost-effective asymmetric reductive amination route into your portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team to request specific COA data for similar chiral amine products and comprehensive route feasibility assessments. Together, we can drive down manufacturing costs and secure a sustainable supply of critical intermediates for the global agrochemical and pharmaceutical markets.
