Revolutionizing Chiral Amine Production: High-Efficiency Asymmetric Reductive Amination for Industrial Scale-Up
Introduction to Advanced Chiral Amine Synthesis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the demand for more sustainable and cost-effective synthetic routes. Patent CN110551033B introduces a groundbreaking methodology for the preparation of chiral amines through the asymmetric reductive amination of ketones. This technology represents a paradigm shift from traditional multi-step sequences to a streamlined, one-pot catalytic process. By utilizing a sophisticated iridium complex coordinated with novel chiral phosphine-phosphoramidite ligands, the invention achieves remarkable enantioselectivity and catalytic turnover numbers. 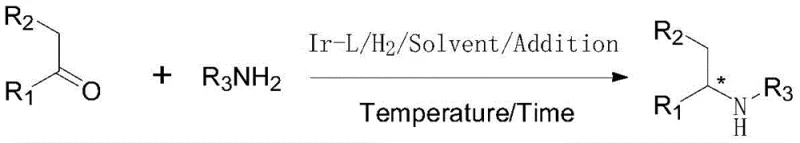 . For R&D directors and procurement strategists, this development signals a new era where high-purity chiral building blocks can be accessed with unprecedented efficiency, directly addressing the critical need for reliable agrochemical intermediate supplier capabilities in a competitive global market.
. For R&D directors and procurement strategists, this development signals a new era where high-purity chiral building blocks can be accessed with unprecedented efficiency, directly addressing the critical need for reliable agrochemical intermediate supplier capabilities in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chiral amines has relied heavily on the asymmetric hydrogenation of pre-formed imines or enamines. While effective, this conventional approach suffers from inherent thermodynamic and operational drawbacks. The formation of the imine intermediate is an equilibrium-controlled reaction that often requires the continuous removal of water to drive completion, adding significant complexity to the reactor setup and energy consumption. Furthermore, many imine intermediates are thermally unstable or prone to polymerization, necessitating low-temperature handling and immediate consumption, which complicates logistics and batch scheduling. Additionally, the catalyst systems used in traditional imine hydrogenation often require high loadings of precious metals to maintain activity, as the free amine product can coordinate to the metal center and inhibit catalysis. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in supply chains that struggle to meet the rigorous demands of modern drug development.
The Novel Approach
The methodology disclosed in CN110551033B elegantly circumvents these challenges by enabling the direct asymmetric reductive amination of ketones with amines in a single vessel. This telescoped process eliminates the need for isolating the imine intermediate, thereby enhancing the overall atom economy and reducing waste generation. The core innovation lies in the specific iridium-chiral phosphine-phosphoramidite catalyst system, which exhibits exceptional tolerance to the reaction conditions and substrates. Unlike previous generations of catalysts that were easily poisoned by amine coordination, this novel system maintains high activity even in the presence of excess amine. The inclusion of titanium alkoxide additives further accelerates the condensation step between the ketone and amine, ensuring rapid conversion to the reactive imine species which is immediately reduced in situ. This synergistic effect allows for milder reaction conditions and significantly shorter cycle times, offering a compelling value proposition for cost reduction in chiral amine manufacturing while maintaining strict control over stereochemical outcomes.
Mechanistic Insights into Ir-Catalyzed Asymmetric Reductive Amination
To fully appreciate the technical superiority of this process, one must examine the intricate interplay between the catalyst architecture and the reaction mechanism. The active catalytic species is generated in situ from an iridium-cyclooctadiene precursor, such as [Ir(COD)Cl]2, and a tailored chiral phosphine-phosphoramidite ligand. 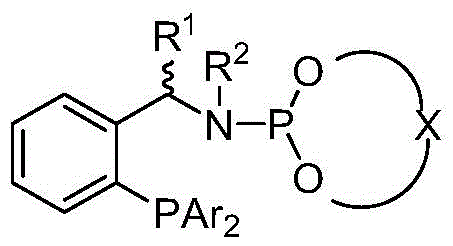 . The ligand design is critical; it features a bulky binaphthyl backbone that creates a well-defined chiral pocket around the iridium center. This steric environment dictates the facial selectivity of the hydride transfer to the prochiral imine intermediate, ensuring the formation of the desired enantiomer with high fidelity. The phosphine moiety provides strong sigma-donation to stabilize the low-valent iridium species, while the phosphoramidite group offers tunable electronic properties and additional steric bulk via the N-substituents. This hemilabile nature allows for the necessary coordination vacancies for hydrogen activation while preventing catalyst decomposition. The result is a robust catalytic cycle that can sustain turnover frequencies far exceeding those of traditional ferrocene-based or simple phosphine ligands, making it ideally suited for the commercial scale-up of complex polymer additives and fine chemicals.
. The ligand design is critical; it features a bulky binaphthyl backbone that creates a well-defined chiral pocket around the iridium center. This steric environment dictates the facial selectivity of the hydride transfer to the prochiral imine intermediate, ensuring the formation of the desired enantiomer with high fidelity. The phosphine moiety provides strong sigma-donation to stabilize the low-valent iridium species, while the phosphoramidite group offers tunable electronic properties and additional steric bulk via the N-substituents. This hemilabile nature allows for the necessary coordination vacancies for hydrogen activation while preventing catalyst decomposition. The result is a robust catalytic cycle that can sustain turnover frequencies far exceeding those of traditional ferrocene-based or simple phosphine ligands, making it ideally suited for the commercial scale-up of complex polymer additives and fine chemicals.
Furthermore, the substrate scope of this catalytic system is remarkably broad, accommodating a diverse array of ketones and amines as defined in the patent structural formulas. 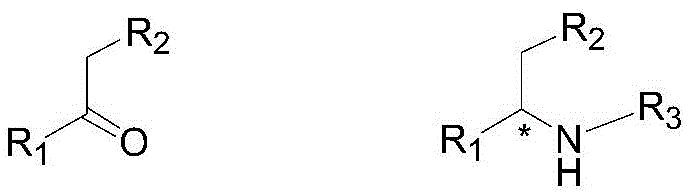 . The R1 and R2 groups on the ketone can range from simple alkyl chains to functionalized aryl groups containing ethers or esters, demonstrating the chemoselectivity of the catalyst. It tolerates potentially reducible functional groups without compromising the integrity of the final product. Similarly, the amine component (R3NH2) can be an aromatic amine, such as 2,6-dimethylaniline, or an aliphatic amine, expanding the utility of this method beyond a single niche application. The mechanism likely involves the initial Lewis acid-assisted condensation of the ketone and amine, facilitated by the titanium additive, to form an iminium ion. This activated species then coordinates to the chiral iridium-hydride complex, undergoing stereoselective hydride insertion. The subsequent protonolysis releases the chiral amine product and regenerates the active catalyst. This detailed understanding of the mechanistic pathway allows process chemists to fine-tune reaction parameters, such as temperature and pressure, to optimize impurity profiles and maximize yield for specific target molecules.
. The R1 and R2 groups on the ketone can range from simple alkyl chains to functionalized aryl groups containing ethers or esters, demonstrating the chemoselectivity of the catalyst. It tolerates potentially reducible functional groups without compromising the integrity of the final product. Similarly, the amine component (R3NH2) can be an aromatic amine, such as 2,6-dimethylaniline, or an aliphatic amine, expanding the utility of this method beyond a single niche application. The mechanism likely involves the initial Lewis acid-assisted condensation of the ketone and amine, facilitated by the titanium additive, to form an iminium ion. This activated species then coordinates to the chiral iridium-hydride complex, undergoing stereoselective hydride insertion. The subsequent protonolysis releases the chiral amine product and regenerates the active catalyst. This detailed understanding of the mechanistic pathway allows process chemists to fine-tune reaction parameters, such as temperature and pressure, to optimize impurity profiles and maximize yield for specific target molecules.
How to Synthesize Chiral Amines Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to leverage the full potential of the iridium catalyst system. The patent outlines a straightforward protocol that begins with the activation of the catalyst precursor under an inert nitrogen atmosphere to prevent oxidation of the sensitive phosphine ligands. The choice of solvent, typically toluene or dichloromethane, plays a vital role in solubilizing both the organic substrates and the organometallic catalyst. Following catalyst activation, the substrates and the titanium alkoxide additive are introduced, and the mixture is subjected to hydrogen pressure in a sealed autoclave. The detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the guide below.
- Prepare the active catalyst by mixing an iridium-cyclooctadiene precursor with a chiral phosphine-phosphoramidite ligand in a solvent like toluene under inert atmosphere.
- Add the ketone substrate, amine source, and a titanium alkoxide additive to the catalyst solution to facilitate imine formation and activation.
- Conduct the hydrogenation reaction in a high-pressure autoclave at 20-100 bar H2 and 20-100°C, followed by purification to isolate the chiral amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel asymmetric reductive amination technology offers tangible strategic benefits that extend beyond mere technical novelty. The most significant advantage lies in the drastic reduction of catalyst loading required to achieve high conversion. The patent reports substrate-to-catalyst (S/C) ratios reaching up to 500,000:1 for the synthesis of key agrochemical intermediates. This extraordinary turnover number implies that the consumption of expensive iridium metal is minimized to trace levels, directly translating into substantial cost savings on raw materials. In an industry where precious metal prices are volatile, reducing dependency on high catalyst loadings provides a hedge against market fluctuations and stabilizes long-term production costs. Moreover, the simplification of the process from a two-step sequence to a one-pot operation reduces the number of unit operations, lowering capital expenditure on equipment and decreasing the labor hours required per batch. This operational efficiency enhances the reliability of the supply chain by shortening manufacturing cycles and reducing the risk of delays associated with intermediate isolation and purification.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of intermediate isolation steps and the ultra-low catalyst loading. By avoiding the need to synthesize and purify imine intermediates separately, manufacturers save on solvents, filtration media, and energy consumption associated with drying and concentration steps. The high atom economy of the reductive amination ensures that the majority of the starting material mass is incorporated into the final product, minimizing waste disposal costs. Furthermore, the ability to use lower pressures and temperatures compared to some harsh traditional methods reduces the energy footprint of the facility. These cumulative efficiencies result in a leaner manufacturing process that significantly lowers the overall cost of goods, allowing companies to offer more competitive pricing for high-purity OLED material precursors or pharmaceutical building blocks without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of this catalytic system. The tolerance of the catalyst to various functional groups means that a single platform technology can be applied to synthesize a wide library of chiral amines, reducing the need for multiple specialized production lines. This flexibility allows manufacturers to respond quickly to changes in market demand or to switch production between different API intermediates with minimal changeover time. Additionally, the high yields reported (often exceeding 90%) ensure that less starting material is required to produce a fixed amount of product, reducing the strain on upstream raw material supplies. The stability of the catalyst system also implies fewer batch failures due to catalyst deactivation, leading to more predictable delivery schedules and improved on-time performance for customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with the principles of green chemistry. The reduction in solvent usage and waste generation simplifies the treatment of effluent streams, making it easier to comply with increasingly stringent environmental regulations. The high selectivity of the reaction minimizes the formation of diastereomeric impurities and side products, which simplifies downstream purification and reduces the volume of hazardous waste sent for incineration. The scalability of the process is evidenced by its successful application in the synthesis of Metolachlor intermediates at industrially relevant scales.
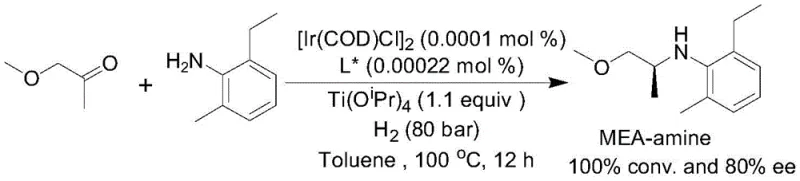 . This proven track record at scale gives confidence to investors and stakeholders that the technology can be seamlessly transferred from the laboratory to multi-ton production facilities, ensuring a continuous and compliant supply of critical chemical ingredients.
. This proven track record at scale gives confidence to investors and stakeholders that the technology can be seamlessly transferred from the laboratory to multi-ton production facilities, ensuring a continuous and compliant supply of critical chemical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reductive amination technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this route for your specific product portfolio.
Q: What is the primary advantage of this asymmetric reductive amination method over traditional imine hydrogenation?
A: The primary advantage is the elimination of the separate imine formation step. This one-pot process improves atom economy, reduces operational complexity, and avoids the equilibrium limitations often associated with isolating unstable imine intermediates, leading to higher overall yields.
Q: How does the catalyst loading in this patent compare to historical methods?
A: This method demonstrates exceptional catalytic efficiency with substrate-to-catalyst (S/C) ratios reaching up to 500,000:1. This is a significant improvement over earlier methods which often required catalyst loadings 100 times higher, drastically reducing the cost contribution of the precious metal iridium.
Q: Is this technology suitable for the synthesis of Metolachlor intermediates?
A: Yes, the patent explicitly highlights the synthesis of the Metolachlor intermediate (S)-2-ethyl-N-(1-methoxy-2-propyl)-6-methylaniline. It achieves 95% yield and 80% ee even at extremely low catalyst loadings, proving its robustness for large-scale agrochemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
The technological advancements described in CN110551033B represent a significant leap forward in the synthesis of chiral amines, offering a pathway to higher purity and lower costs. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of such catalytic innovations and have integrated similar state-of-the-art methodologies into our manufacturing repertoire. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from gram-scale optimization to full industrial output. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and chemical purity at every stage of production. We understand that in the fast-paced world of pharmaceutical and agrochemical development, time is of the essence, and our optimized processes are designed to accelerate your timeline to market.
We invite you to collaborate with us to leverage these cutting-edge synthetic strategies for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this efficient reductive amination route for your specific targets. We encourage you to contact our technical procurement team to request specific COA data for our existing chiral amine catalog or to discuss route feasibility assessments for your custom synthesis needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic ally committed to driving innovation and efficiency in your supply chain.
