Advanced Iridium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Amines
Advanced Iridium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Amines
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN110551037B, which discloses a highly efficient method for the asymmetric hydrogenation of imines utilizing an iridium/chiral diphosphine system. This technology represents a paradigm shift from traditional catalytic approaches, offering a robust pathway to produce chiral amines with exceptional enantiomeric excess values exceeding 80%, and in specific optimized cases, reaching up to 91% ee. The core innovation lies in the in-situ generation of a chiral catalyst through the coordination of an iridium-cyclooctadiene complex with a novel chiral diphosphine ligand. This approach not only simplifies the operational procedure but also dramatically enhances the turnover number, making it a viable solution for industrial-scale manufacturing where cost and efficiency are paramount.
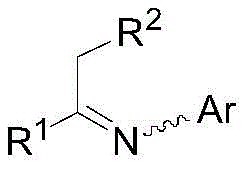
The versatility of this catalytic system is underscored by its ability to handle a broad spectrum of substrates under relatively mild conditions. Unlike legacy processes that often require extreme pressures or temperatures to achieve acceptable conversion rates, this method operates effectively within a pressure range of 20 to 100 bar and temperatures between 20 and 100°C. For procurement managers and supply chain directors, the implications are profound; the ability to achieve a substrate-to-catalyst molar ratio of up to 500,000:1 translates directly into substantial reductions in precious metal consumption. This efficiency is particularly notable in the synthesis of critical agrochemical intermediates, such as those for S-metolachlor, where the process achieved a 95% yield with high stereoselectivity. Such performance metrics position this technology as a cornerstone for any organization seeking a reliable agrochemical intermediate supplier capable of delivering high-volume, high-quality materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of imines has been fraught with challenges that hindered its widespread industrial adoption. Early attempts, such as those reported by Levi et al. in 1975, suffered from disappointingly low enantiomeric excess values, often hovering around merely 22%, which is entirely insufficient for the rigorous purity standards of modern drug manufacturing. Subsequent improvements, like the ferrocene bisphosphine ligand systems introduced in the late 1990s, managed to elevate ee values to approximately 76%, yet these advancements came with significant operational burdens. These conventional methods typically necessitated the use of large quantities of acid additives and required specialized high-pressure equipment capable of withstanding up to 80 atmospheres of hydrogen pressure at elevated temperatures around 50°C. Furthermore, the synthesis of the ligands employed in these older systems was notoriously difficult and costly, involving complex multi-step sequences that compromised the overall atom economy and increased the environmental footprint of the process.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110551037B offers a streamlined and economically superior alternative that addresses the inherent flaws of its predecessors. The novel approach utilizes a chiral diphosphine ligand that is remarkably simple to prepare, involving straightforward reactions that are amenable to kilogram-level production without the need for exotic reagents or cryogenic conditions. By employing an iridium-cyclooctadiene complex that coordinates in situ with this accessible ligand, the system achieves a level of catalytic activity that far surpasses earlier generations. The reaction conditions are significantly milder, eliminating the absolute necessity for aggressive acid additives and allowing for flexibility in temperature and pressure parameters. This flexibility is crucial for cost reduction in pharmaceutical intermediates manufacturing, as it allows producers to optimize energy consumption and extend the lifespan of their reactor vessels. The result is a process that delivers yields up to 95% and enantioselectivity up to 91%, setting a new benchmark for efficiency in the production of chiral amines.
Mechanistic Insights into Iridium-Chiral Diphosphine Catalysis
To fully appreciate the technical superiority of this system, one must delve into the mechanistic intricacies of the iridium-chiral diphosphine catalyst. The active species is generated through the precise coordination of the iridium center with the bidentate phosphine ligand, creating a chiral environment that dictates the stereochemical outcome of the hydrogenation. The ligand architecture, featuring a ferrocene backbone substituted with specific phosphine groups, plays a pivotal role in this induction. The steric bulk and electronic properties of the substituents on the phosphorus atoms create a well-defined chiral pocket around the metal center. When the imine substrate approaches this active site, the spatial arrangement forces the hydrogen addition to occur from a specific face of the double bond, thereby ensuring high enantioselectivity. This level of control is essential for producing high-purity chiral amines, as even minor deviations in the catalyst structure can lead to significant drops in optical purity.
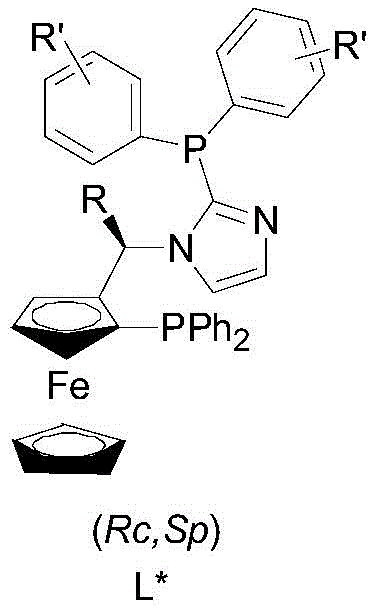
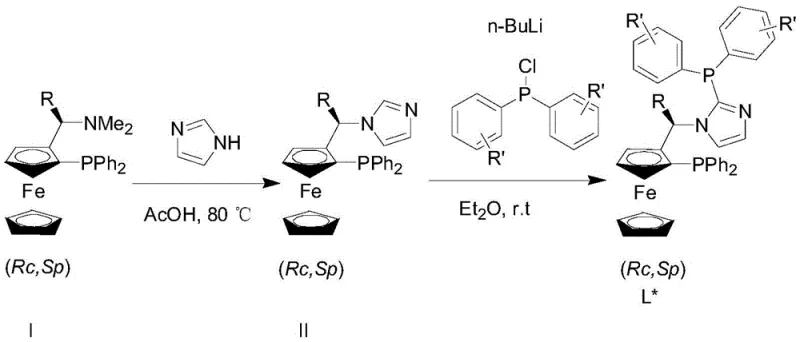
The synthesis of the ligand itself is a testament to the process's elegance and scalability. As illustrated in the patent documentation, the ligand is constructed from readily available precursors through a sequence that avoids hazardous intermediates. The initial step involves the reaction of a ferrocenyl precursor with imidazole under acidic conditions, followed by a lithiation and phosphorylation sequence to install the diphenylphosphine moieties. This synthetic route is not only high-yielding but also robust, ensuring consistent quality of the ligand batch after batch. Consistency in ligand quality is a critical factor for maintaining consistent catalyst performance, which in turn guarantees the reproducibility of the hydrogenation reaction. For R&D directors focused on impurity profiles, this controlled synthesis minimizes the risk of introducing trace metal contaminants or organic byproducts that could complicate downstream purification. The ability to tune the ligand structure by varying the R and R' groups further enhances the system's utility, allowing chemists to tailor the catalyst for specific substrate classes ranging from simple alkyl imines to complex aromatic derivatives.
How to Synthesize Chiral Amines Efficiently
Implementing this advanced hydrogenation technology requires a clear understanding of the operational parameters to maximize yield and selectivity. The process begins with the careful preparation of the catalyst solution, where the iridium precursor and the chiral ligand are mixed in a suitable solvent such as dichloromethane or toluene. This pre-formation step is critical to ensure that the active catalytic species is fully generated before the introduction of the substrate. Once the catalyst is ready, the imine substrate is introduced under an inert atmosphere to prevent oxidation of the sensitive phosphine ligands. The reaction mixture is then subjected to hydrogen pressure, initiating the reduction cycle. Detailed standard operating procedures regarding stoichiometry, mixing rates, and safety protocols are essential for successful scale-up. The following guide outlines the standardized synthesis steps derived from the patent data to assist technical teams in replicating these results.
- Prepare the chiral catalyst by stirring an iridium-cyclooctadiene complex with a chiral diphosphine ligand in a solvent like dichloromethane at room temperature for 2 to 5 hours.
- Under nitrogen protection, add the substrate imine dissolved in solvent to the reaction vessel containing the pre-formed catalyst.
- Replace the atmosphere with hydrogen three times, pressurize to 20-100 bar, and react at 20-100°C for 1-24 hours before workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the adoption of this iridium-catalyzed process offers compelling economic and logistical benefits. The primary driver for cost optimization is the extraordinary turnover number of the catalyst. With substrate-to-catalyst ratios reaching 500,000:1, the amount of expensive iridium required per kilogram of product is negligible compared to traditional methods. This drastic reduction in precious metal loading directly translates to lower raw material costs and simplifies the purification process, as there is less residual metal to remove from the final API or intermediate. Furthermore, the simplicity of the ligand synthesis ensures a stable and cost-effective supply of the chiral inducer, mitigating the risks associated with sourcing complex, proprietary ligands from single-source vendors. This supply chain resilience is vital for maintaining uninterrupted production schedules in a volatile global market.
- Cost Reduction in Manufacturing: The economic impact of this technology extends beyond mere catalyst loading. By eliminating the need for harsh acid additives and reducing the severity of reaction conditions, the process lowers the operational expenditure related to equipment maintenance and energy consumption. The mild conditions allow for the use of standard stainless steel reactors rather than specialized Hastelloy vessels lined for corrosive environments, representing a significant capital expenditure saving for facilities looking to expand capacity. Additionally, the high atom economy of the hydrogenation reaction minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. These factors combined create a leaner, more profitable manufacturing process that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes significantly to supply chain stability. The ligand synthesis relies on commodity chemicals and straightforward transformations, reducing the risk of supply bottlenecks that often plague complex fine chemical supply chains. Because the reaction tolerates a wide range of substrates and conditions, it offers flexibility in sourcing raw materials; if one specific imine precursor is unavailable, the process can often be adapted to accept alternative feedstocks with minimal re-optimization. This adaptability ensures that production can continue even when facing upstream supply disruptions. Moreover, the high conversion rates (>99% in many examples) mean that less unreacted starting material needs to be recovered and recycled, streamlining the workflow and shortening the overall cycle time from raw material intake to finished goods.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor is often where new technologies fail, but this iridium system is explicitly designed for industrial practicality. The patent highlights the potential for continuous operation, a mode of manufacturing that is increasingly favored for its safety and efficiency profile. Continuous flow chemistry allows for better heat and mass transfer control, which is critical for exothermic hydrogenation reactions. From an environmental perspective, the process aligns with green chemistry principles by utilizing hydrogen, the cleanest reducing agent, which produces no byproducts other than the desired amine. The absence of stoichiometric reducing agents like borohydrides or silanes eliminates the generation of large volumes of inorganic waste salts. This eco-friendly profile facilitates easier regulatory approval and supports corporate sustainability goals, making it an attractive option for companies committed to reducing their carbon footprint.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is essential for making informed sourcing and development decisions. The following questions address common inquiries regarding the technical specifications and commercial viability of the iridium/chiral diphosphine system. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's fit for your specific project requirements. Whether you are concerned about catalyst lifetime, substrate scope, or purification needs, these insights clarify the operational realities of implementing this advanced synthesis method.
Q: What is the maximum substrate-to-catalyst ratio achieved in this process?
A: The patent demonstrates exceptional catalytic activity with a substrate-to-catalyst (S/C) molar ratio reaching up to 500,000:1, specifically in the synthesis of S-metolachlor intermediates.
Q: How does this method compare to previous ferrocene-based systems?
A: Unlike earlier methods requiring harsh conditions and difficult ligand synthesis, this novel iridium/chiral diphosphine system operates under milder conditions with simpler ligand preparation while achieving higher enantioselectivity (up to 91% ee).
Q: What types of substrates are compatible with this hydrogenation method?
A: The system is highly versatile, accommodating various imine substrates where R1 can be alkyl, cycloalkyl, or aryl groups, and Ar can be substituted phenyl rings, making it suitable for diverse pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
The transition from patent laboratory data to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of process chemists is adept at optimizing parameters such as temperature, pressure, and catalyst loading to ensure that the theoretical efficiencies described in patent CN110551037B are realized in actual production runs. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and GC instruments to verify enantiomeric excess and chemical purity at every stage of production. This commitment to quality ensures that every batch of chiral amine or agrochemical intermediate we deliver meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to unlock the full potential of this asymmetric hydrogenation technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this catalytic system for your target molecules. We encourage potential partners to contact us to obtain specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you secure a reliable supply of high-purity intermediates while driving down costs and enhancing the sustainability of your manufacturing operations.
