Advancing Chiral Amine Production: High-Efficiency Asymmetric Reductive Amination for Industrial Scale-Up
Advancing Chiral Amine Production: High-Efficiency Asymmetric Reductive Amination for Industrial Scale-Up
The landscape of chiral amine synthesis is undergoing a significant transformation driven by the need for more efficient, atom-economical, and scalable processes. Patent CN110551033B introduces a groundbreaking methodology for the preparation of chiral amines through the asymmetric reductive amination of ketones. This technology leverages a sophisticated catalytic system comprising an iridium precursor and a novel chiral phosphine-phosphoramidite ligand. Unlike traditional multi-step sequences that require the isolation of imine intermediates, this approach facilitates a direct, one-pot conversion of ketones and amines into high-value chiral amines. The implications for the fine chemical industry are profound, offering a pathway to reduce waste, lower energy consumption, and streamline production workflows for critical agrochemical and pharmaceutical intermediates.
This innovation addresses long-standing challenges in asymmetric catalysis, particularly the balance between catalytic activity and stereoselectivity. By utilizing an in-situ generated iridium complex, the method achieves remarkable turnover numbers, making it economically viable for large-scale manufacturing. The process operates under relatively mild conditions compared to historical precedents, yet delivers robust performance across a diverse range of substrates. For R&D directors and process chemists, this represents a pivotal shift towards greener chemistry without compromising on the stringent purity and optical purity requirements demanded by modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines has relied heavily on the asymmetric hydrogenation of pre-formed imines or enamines. While effective in many contexts, this conventional route suffers from inherent thermodynamic and kinetic limitations. The formation of the imine intermediate is an equilibrium process that often requires the continuous removal of water to drive the reaction to completion, adding complexity to the reactor setup and increasing operational costs. Furthermore, the isolation and purification of these unstable imine intermediates can lead to significant material loss and generate additional solvent waste. Another critical drawback is catalyst deactivation; free amines present in the reaction mixture can coordinate strongly with the transition metal center, poisoning the catalyst and necessitating higher catalyst loadings to maintain acceptable reaction rates.
Additionally, earlier attempts at direct reductive amination, such as those reported in the late 1990s using ferrocene-based ligands, often required excessive amounts of catalyst—sometimes up to 100 times more than needed for imine hydrogenation—to overcome these inhibitory effects. This high catalyst consumption rendered such processes economically unfeasible for industrial applications, forcing manufacturers to stick with the less efficient two-step imine hydrogenation routes. The inability to efficiently process aliphatic ketones further restricted the utility of these early catalytic systems, limiting their application to a narrow subset of aromatic substrates and hindering the development of diverse chiral amine libraries.
The Novel Approach
The methodology disclosed in CN110551033B fundamentally disrupts these limitations by introducing a highly active iridium/chiral phosphine-phosphoramidite catalyst system designed specifically for direct reductive amination. This novel approach bypasses the need for separate imine formation and isolation steps, thereby enhancing the overall atom economy of the synthesis. The unique electronic and steric properties of the phosphine-phosphoramidite ligands prevent the catalyst from being inhibited by the amine substrates, allowing for significantly lower catalyst loadings while maintaining high activity. This breakthrough enables the direct conversion of ketones and amines into chiral products with exceptional efficiency, effectively merging two reaction steps into a single, streamlined operation.
Moreover, this system demonstrates unprecedented versatility, successfully catalyzing reactions with both aromatic and aliphatic ketones, a feat that eluded many previous generations of catalysts. The inclusion of titanium alkoxide additives plays a crucial role in this success, acting as Lewis acids to activate the carbonyl group and scavengers to remove water produced during imine formation in situ. This synergistic effect ensures that the reaction proceeds smoothly to high conversion without the need for complex water removal apparatuses. The result is a robust, scalable process that delivers high yields and excellent enantioselectivity, positioning it as a superior alternative for the commercial production of complex chiral amines.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Reductive Amination
The core of this technological advancement lies in the precise design of the chiral ligand and its interaction with the iridium metal center. The catalyst is generated in situ by reacting an iridium-cyclooctadiene precursor, such as [Ir(COD)Cl]2, with a chiral phosphine-phosphoramidite ligand. This specific class of ligands combines a soft phosphine donor with a harder phosphoramidite moiety, creating a hemilabile environment that facilitates both substrate binding and product release. The chirality of the ligand, often derived from binaphthyl backbones, creates a well-defined chiral pocket around the metal center. This pocket dictates the facial selectivity of the hydride transfer to the transient imine or iminium ion intermediate, ensuring the formation of the desired enantiomer with high fidelity.
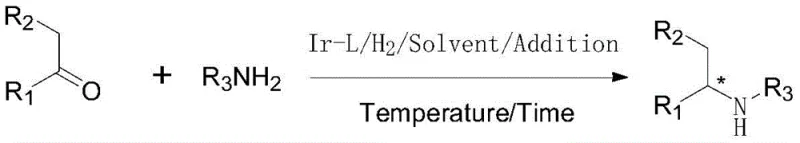
The reaction mechanism involves the initial condensation of the ketone and amine to form an imine or iminium species, a step accelerated by the titanium additive. The iridium-hydride species, formed under hydrogen pressure, then undergoes migratory insertion into the C=N bond of the imine. The steric bulk of the ligand substituents (R1, R2, Ar groups) forces the substrate to approach the metal center in a specific orientation, minimizing the formation of the undesired enantiomer. Following hydride transfer, the resulting chiral amine dissociates from the catalyst, regenerating the active iridium species for the next catalytic cycle. This cycle is highly efficient, capable of sustaining turnover numbers that were previously unattainable in direct reductive amination protocols.
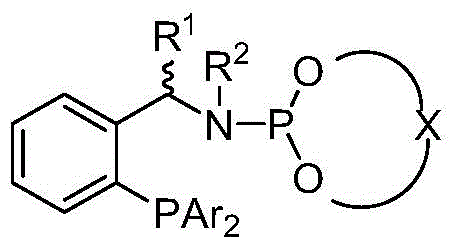
Impurity control is another critical aspect managed by this mechanistic design. In traditional methods, the over-reduction of ketones to alcohols is a common side reaction that competes with amine formation. However, the specific electronic tuning of the iridium-phosphine-phosphoramidite complex favors the reduction of the C=N bond over the C=O bond. The presence of the amine and the titanium additive shifts the equilibrium towards the imine, which is a better electrophile for the iridium-hydride species than the ketone itself. Consequently, the formation of alcohol byproducts is suppressed, leading to cleaner reaction profiles and simplifying downstream purification. This selectivity is vital for meeting the rigorous impurity specifications required for pharmaceutical and agrochemical active ingredients.
How to Synthesize Chiral Amines Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and enantioselectivity. The process begins with the preparation of the catalyst solution under an inert atmosphere to prevent oxidation of the sensitive iridium complex. The choice of solvent, typically toluene or dichloromethane, influences the solubility of the substrates and the stability of the catalyst. The molar ratio of the ligand to iridium is optimized to ensure complete coordination, while the addition of titanium isopropoxide is critical for driving the imine formation equilibrium forward. Detailed standard operating procedures regarding temperature ramps, hydrogen pressurization rates, and workup protocols are essential for reproducible results on a manufacturing scale.
- Prepare the catalyst system by dissolving the Iridium-cyclooctadiene complex and the chiral phosphine-phosphoramidite ligand in a solvent such as toluene under nitrogen protection, stirring at room temperature for 10 minutes to ensure in-situ coordination.
- Add the substrate amine, ketone, and a titanium alkoxide additive (e.g., titanium isopropoxide) to the reaction mixture. Transfer the solution to a high-pressure autoclave.
- Purge the reactor with hydrogen gas three times, pressurize to 20-100 bar, and heat to 20-100°C. Maintain reaction for 1-24 hours, then release pressure and isolate the chiral amine product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
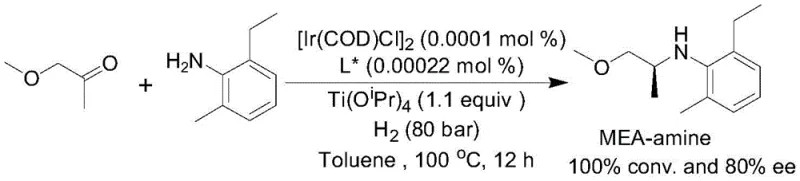
For procurement managers and supply chain leaders, the adoption of this asymmetric reductive amination technology offers tangible strategic benefits beyond mere technical superiority. The most significant advantage is the drastic reduction in catalyst consumption. The patent highlights a substrate-to-catalyst ratio (S/C) of up to 500,000 in the synthesis of the metolachlor intermediate. This level of efficiency translates directly into substantial cost savings, as precious metal catalysts represent a major portion of the raw material cost in fine chemical synthesis. By minimizing the amount of iridium required per kilogram of product, manufacturers can significantly lower their variable costs and reduce exposure to the volatility of precious metal markets.
Furthermore, the simplified one-pot process enhances supply chain reliability by reducing the number of unit operations and intermediate handling steps. Fewer processing stages mean shorter cycle times and reduced risk of batch failures or contamination events. The robustness of the catalyst system allows for continuous operation potential, which is a key factor for securing long-term supply contracts with major agrochemical and pharmaceutical companies. The ability to source high-purity intermediates from a supplier utilizing this efficient technology ensures a stable flow of materials, mitigating the risks associated with complex, multi-step supply chains that are prone to bottlenecks.
From an environmental and compliance perspective, this method aligns perfectly with modern sustainability goals. The high atom economy and reduced solvent usage contribute to a lower environmental footprint, facilitating easier compliance with increasingly strict environmental regulations. The elimination of isolated imine intermediates reduces the generation of hazardous waste streams, lowering disposal costs and improving the overall safety profile of the manufacturing facility. These factors collectively enhance the marketability of the final product, appealing to end-users who prioritize green chemistry principles in their sourcing decisions. The scalability of the process, demonstrated by successful kilogram-level production, assures partners that supply can be ramped up quickly to meet market demand without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reductive amination technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines. The answers reflect the proven capabilities of the iridium/phosphine-phosphoramidite system in real-world synthetic scenarios.
Q: What are the advantages of this asymmetric reductive amination method over traditional imine hydrogenation?
A: This method eliminates the need for pre-forming imine or enamine intermediates, which simplifies the process and improves atom economy. It avoids the equilibrium limitations of imine formation and prevents catalyst inhibition often caused by substrate amines complexing with transition metals in traditional methods.
Q: What represents the maximum catalyst efficiency demonstrated in this technology?
A: The patent demonstrates exceptional catalyst turnover, specifically in the synthesis of the metolachlor intermediate. It achieved a substrate-to-catalyst ratio (S/C) of 500,000 while maintaining a 95% yield and 80% enantiomeric excess, indicating high industrial viability.
Q: Is this catalytic system applicable to both aromatic and aliphatic ketones?
A: Yes, the technology shows broad substrate scope. While earlier methods struggled with aliphatic ketones, this Ir/phosphine-phosphoramidite system successfully catalyzes the reductive amination of various ketones, including acetophenone derivatives, butanone, and methoxyacetone, with high enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110551033B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methods into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields observed in the lab are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral amine meets the exacting standards required for agrochemical and pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Whether you are looking to optimize an existing process or develop a new route for a complex chiral intermediate, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive efficiency and value for your organization.
