Scalable Asymmetric Reductive Amination for High-Purity Chiral Amines and Agrochemical Intermediates
Scalable Asymmetric Reductive Amination for High-Purity Chiral Amines and Agrochemical Intermediates
The landscape of chiral amine synthesis is undergoing a significant transformation driven by the need for more efficient, atom-economical, and scalable processes. Patent CN110551032B introduces a groundbreaking methodology utilizing an iridium chiral phosphine-aminophosphine ligand system to catalyze the asymmetric reductive amination of ketones. This technology represents a pivotal advancement for the production of high-value intermediates, particularly in the agrochemical sector where compounds like S-metolachlor are critical. By enabling the direct conversion of ketones and amines into chiral amines with exceptional enantioselectivity, this process eliminates the need for pre-forming imine intermediates, thereby streamlining the synthetic route and enhancing overall process efficiency for industrial applications.
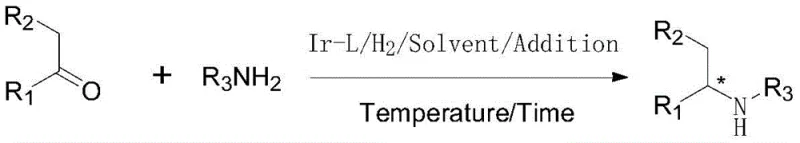
For procurement managers and supply chain directors, the implications of this technology extend far beyond the laboratory. The ability to achieve high yields and enantiomeric excess (ee) values exceeding 80% under relatively mild conditions translates directly into reduced manufacturing costs and improved supply chain reliability. The patent highlights a remarkable substrate-to-catalyst ratio (S/C) of up to 500,000 in the synthesis of key agrochemical intermediates, demonstrating a level of catalytic efficiency that significantly lowers the barrier to entry for large-scale production. This makes the technology an attractive option for companies seeking a reliable agrochemical intermediate supplier capable of delivering high-purity materials at a competitive cost structure without compromising on quality or environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral amines has relied heavily on the asymmetric hydrogenation of pre-formed imines or enamines. While effective, this two-step approach suffers from inherent inefficiencies, including the requirement for separate isolation or stabilization of the imine intermediate, which often exhibits poor stability and can undergo hydrolysis or polymerization. Furthermore, conventional catalytic systems frequently struggle with substrate inhibition, where the amine substrate or the chiral amine product coordinates strongly with the transition metal center, effectively poisoning the catalyst and necessitating high catalyst loadings to drive the reaction to completion. These factors contribute to increased operational complexity, higher raw material consumption, and elevated waste generation, posing significant challenges for cost reduction in agrochemical manufacturing and pharmaceutical intermediate production.
The Novel Approach
The novel approach detailed in the patent overcomes these historical bottlenecks by employing a one-pot asymmetric reductive amination strategy. By utilizing a specialized iridium complex coordinated with a chiral phosphine-aminophosphine ligand, the system facilitates the direct reaction between the ketone and the amine source in the presence of hydrogen. This method not only simplifies the operational workflow by removing the intermediate isolation step but also demonstrates superior tolerance to various functional groups. The inclusion of a titanium alkoxide additive further enhances the reaction kinetics by promoting imine formation in situ. This integrated catalytic system achieves high turnover frequencies and maintains robust enantioselectivity across a broad range of substrates, including challenging aliphatic ketones, marking a substantial improvement over previous generations of catalysts that were often restricted to aromatic systems.
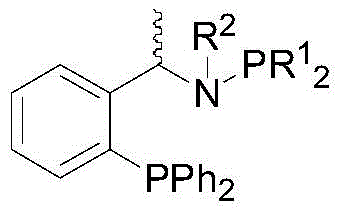
Mechanistic Insights into Iridium-Catalyzed Asymmetric Reductive Amination
The core of this technological breakthrough lies in the unique architecture of the chiral phosphine-aminophosphine ligand system. Unlike simple diphosphine ligands, this bifunctional ligand design incorporates both phosphine and amine moieties, creating a highly defined chiral environment around the iridium center. This specific coordination geometry is crucial for discriminating between the prochiral faces of the transient imine intermediate during the hydride transfer step. The mechanism likely involves the initial formation of an iridium-hydride species, followed by the coordination of the imine substrate. The steric bulk and electronic properties of the ligand substituents (R1 and R2 groups) dictate the trajectory of the hydride attack, ensuring the preferential formation of one enantiomer over the other. This precise control is essential for achieving the high enantiomeric excess values required for regulatory compliance in the production of active pharmaceutical ingredients and agrochemical actives.
Furthermore, the reaction system incorporates a Lewis acid additive, typically titanium tetraisopropoxide, which plays a dual role in the catalytic cycle. Firstly, it activates the carbonyl group of the ketone towards nucleophilic attack by the amine, accelerating the formation of the imine intermediate. Secondly, it may help to sequester water produced during the condensation step, shifting the equilibrium towards imine formation and preventing catalyst deactivation. This synergistic effect between the iridium catalyst and the titanium additive allows the reaction to proceed efficiently even with substrates that are traditionally difficult to reduce. The result is a robust process capable of handling diverse substrate classes, from simple acetophenones to complex aliphatic ketones, while maintaining high levels of stereocontrol and minimizing the formation of unwanted by-products such as reduced alcohols.
How to Synthesize Chiral Amines Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly suitable for commercial adoption. The process begins with the in-situ generation of the active catalyst species by mixing the iridium precursor, such as [Ir(COD)Cl]2, with the chiral ligand in a suitable solvent like toluene. This mixture is then combined with the ketone and amine substrates along with the titanium additive in a high-pressure reactor. The reaction is conducted under a hydrogen atmosphere, typically ranging from 20 to 100 bar, and at temperatures between 20°C and 100°C. The flexibility in reaction conditions allows for optimization based on the specific reactivity of the substrates involved. For detailed standard operating procedures and specific parameter optimization for your target molecule, please refer to the technical guide below.
- Prepare the catalyst in situ by dissolving an iridium-cyclooctadiene complex and a chiral phosphine-aminophosphine ligand in a solvent such as toluene under nitrogen protection.
- Add the substrate amine, ketone, and a titanium alkoxide additive (e.g., Ti(OiPr)4) to the reaction mixture in a high-pressure autoclave.
- Pressurize the reactor with hydrogen gas (20-100 bar) and heat the mixture to 20-100°C for 1-24 hours to complete the reductive amination.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed reductive amination technology offers profound advantages for supply chain optimization and cost management. The most striking feature is the unprecedented catalyst efficiency, with substrate-to-catalyst ratios reaching as high as 500,000:1 in the synthesis of S-metolachlor intermediates. This drastic reduction in catalyst loading directly correlates to significant cost savings, as precious metal consumption is minimized. Additionally, the one-pot nature of the reaction reduces the number of unit operations required, leading to lower energy consumption, reduced solvent usage, and shorter cycle times. These efficiencies collectively contribute to a more sustainable and economically viable manufacturing process, addressing the growing demand for green chemistry solutions in the fine chemical industry.
- Cost Reduction in Manufacturing: The elimination of the separate imine formation and isolation step significantly reduces processing time and raw material costs. By avoiding the need for stoichiometric reducing agents or complex workup procedures associated with traditional methods, the overall cost of goods sold (COGS) is substantially lowered. The high turnover number of the catalyst ensures that the cost contribution of the expensive iridium metal is negligible on a per-kilogram basis, making the process economically competitive even for large-volume commodity chemicals. This efficiency allows manufacturers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive agrochemical market.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent product quality and high yields, reducing the risk of batch failures and supply disruptions. The ability to operate under relatively mild conditions and tolerate a wide range of substrates means that the process is less susceptible to variations in raw material quality. This reliability is paramount for maintaining continuous production schedules and meeting the stringent delivery requirements of global pharmaceutical and agrochemical clients. Furthermore, the scalability of the process from laboratory to industrial scale ensures that supply can be ramped up quickly to meet surging market demand without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: The process aligns well with modern environmental, health, and safety (EHS) standards due to its high atom economy and reduced waste generation. By minimizing the use of hazardous reagents and solvents, the environmental footprint of the manufacturing process is significantly reduced. The high selectivity of the reaction minimizes the formation of by-products, simplifying downstream purification and waste treatment. This not only lowers disposal costs but also facilitates regulatory compliance in jurisdictions with strict environmental regulations. The potential for continuous operation further enhances the sustainability profile of the process, making it an ideal choice for companies committed to green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reductive amination technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the primary advantage of this iridium catalytic system over traditional methods?
A: The primary advantage is the exceptionally high turnover number (S/C ratio up to 500,000), which drastically reduces the consumption of expensive iridium metal compared to conventional asymmetric hydrogenation methods that typically require much higher catalyst loadings.
Q: Can this method be applied to aliphatic ketones?
A: Yes, unlike some earlier catalytic systems that were limited to aromatic ketones, this iridium chiral phosphine-aminophosphine ligand system demonstrates broad substrate scope, successfully catalyzing the reductive amination of both aromatic and aliphatic ketones with high enantioselectivity.
Q: What role does the titanium additive play in the reaction?
A: The titanium alkoxide additive acts as a Lewis acid promoter, facilitating the formation of the imine or iminium intermediate from the ketone and amine, which is the crucial step prior to the asymmetric hydrogenation by the iridium catalyst.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the iridium-chiral phosphine-aminophosphine system described in CN110551032B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. We are committed to delivering high-purity chiral amines and agrochemical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific supply chain needs. Whether you are looking to optimize an existing route or develop a new process for a complex chiral intermediate, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can support your goals for cost reduction in agrochemical manufacturing and ensure a stable supply of high-quality intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
