Advanced L-Menthol Manufacturing: Overcoming Selectivity Barriers with Modified Homogeneous Catalysts
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for producing high-value chiral compounds like L-menthol, a critical ingredient in flavors, fragrances, and pharmaceuticals. Patent CN110845305A introduces a groundbreaking methodology that addresses longstanding challenges in stereoselectivity and catalyst recovery. This technology leverages a novel modified homogeneous organoaluminum catalyst to drive the cyclization of citronellal into isopulegol with exceptional precision. By replacing traditional heavy metal catalysts or low-selectivity Lewis acids, this process offers a robust solution for manufacturers aiming to optimize their production lines. The integration of a specifically designed ligand system ensures that the catalytic activity is not only high but also highly selective, minimizing byproduct formation and simplifying downstream purification processes significantly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-menthol has relied heavily on asymmetric hydrogenation using expensive rhodium catalysts or cyclization using simple Lewis acids like zinc bromide. The rhodium-based routes, while effective, suffer from high raw material costs and significant environmental concerns due to heavy metal contamination, necessitating complex removal steps that increase operational expenditures. Alternatively, the use of ZnBr2 for citronellal cyclization often results in suboptimal yields, typically around 81%, and moderate stereoselectivity near 91%. These limitations lead to substantial product loss during purification and require extensive recycling of unreacted materials. Furthermore, previous organoaluminum catalysts often required ester additives and low-temperature conditions, complicating the reaction setup and making catalyst recovery difficult due to structural degradation during quenching.
The Novel Approach
The methodology outlined in the patent revolutionizes this landscape by introducing a modified homogeneous catalyst that operates efficiently under milder conditions without the need for auxiliary esters. This new catalyst system achieves isopulegol yields exceeding 94% and stereoselectivity greater than 98%, representing a marked improvement over conventional techniques. The key lies in the catalyst's ability to maintain its structural integrity throughout the reaction and recovery phases. By utilizing a ligand derived from 2,6-dimethylpyridine and ketones, the catalyst forms a stable complex with alkyl aluminum that resists decomposition. This stability allows for straightforward recovery via crystallization, enabling multiple reuse cycles without significant loss of activity. Consequently, this approach drastically simplifies the workflow, reduces waste generation, and lowers the overall cost of goods sold for L-menthol production.
Mechanistic Insights into Modified Organoaluminum Catalysis
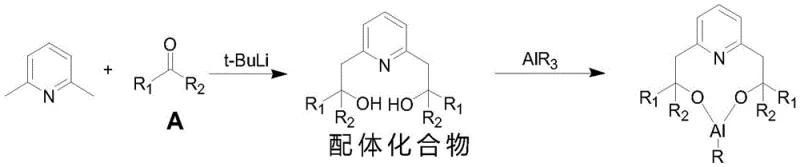
The core innovation of this technology resides in the precise molecular architecture of the ligand and its interaction with the aluminum center. The synthesis begins with the lithiation of 2,6-dimethylpyridine, where the electron-withdrawing nature of the nitrogen atom stabilizes the ring while the ortho-methyl groups provide reactive sites for hydrogen-lithium exchange. Upon reaction with a ketone compound, a bulky ligand framework is established featuring two hydroxyl groups that coordinate tightly with the aluminum atom. This coordination creates a rigid, sterically hindered environment around the active site. When citronellal enters this catalytic pocket, the spatial constraints imposed by the ligand's R1 and R2 groups dictate the trajectory of the cyclization, effectively blocking the formation of unwanted neo-isopulegol isomers. The van der Waals forces between the pyridine nitrogen and the aluminum further stabilize this conformation, ensuring consistent stereochemical outcomes across batches.
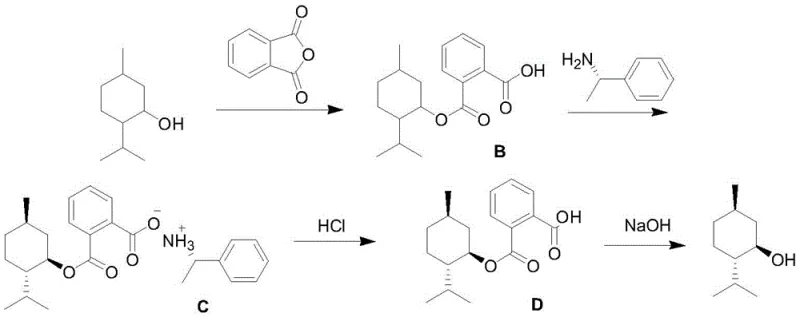
Following the cyclization, the process employs a sophisticated chiral resolution strategy to isolate pure L-menthol from the racemic D,L-mixture. This involves converting the menthol into a monoester derivative using phthalic anhydride, which alters its solubility profile to facilitate crystallization. Subsequent treatment with (S)-α-phenethylamine induces the formation of diastereomeric salts. Due to the specific three-dimensional arrangement of the L-menthol derivative, it forms a less soluble salt with the chiral amine compared to its D-enantiomer counterpart. This difference in solubility allows for the selective precipitation of the desired L-isomer salt, which is then hydrolyzed back to free L-menthol. This chemical induction method avoids the partial racemization often observed in physical separation techniques, ensuring that the final product maintains high optical purity with an ee value reaching 98%.
How to Synthesize L-Menthol Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for implementing this technology in a pilot or commercial setting. It begins with the careful preparation of the catalyst under inert atmosphere to prevent moisture sensitivity issues, followed by the cyclization reaction in common organic solvents like dichloromethane. The subsequent hydrogenation and resolution steps utilize standard unit operations found in most fine chemical facilities, ensuring ease of adoption. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate these high-yield results, please refer to the standardized synthesis guide below.
- Prepare the modified homogeneous catalyst by reacting 2,6-dimethylpyridine with n-butyllithium and a ketone compound to form a ligand, followed by complexation with alkyl aluminum (AlR3).
- Perform the cyclization of citronellal using the prepared catalyst in a solvent like dichloromethane at temperatures between -10°C to 50°C to obtain isopulegol with high stereoselectivity.
- Hydrogenate the isopulegol to D,L-menthol using Pd/C, followed by chiral resolution using (S)-α-phenethylamine to isolate high-purity L-menthol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic system presents a compelling value proposition centered on cost efficiency and supply security. By eliminating the dependency on precious metal catalysts like rhodium, manufacturers can insulate their production costs from the volatile pricing of rare earth metals. The simplified catalyst recovery process means that less fresh catalyst needs to be purchased over time, directly impacting the bottom line through reduced material consumption. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, which reduces the load on purification units and shortens cycle times. This efficiency translates into a more predictable production schedule and enhanced ability to meet tight delivery windows for global clients.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and ester additives significantly lowers the raw material bill. Furthermore, the ability to recover and reuse the organoaluminum catalyst without structural degradation means that the effective cost per kilogram of catalyst consumed is drastically reduced. The high yield and selectivity also mean that less starting material is wasted, maximizing the throughput of existing reactor capacity and lowering the cost per unit of finished L-menthol.
- Enhanced Supply Chain Reliability: The robustness of the catalyst synthesis, which uses readily available precursors like 2,6-dimethylpyridine and common ketones, ensures a stable supply of the catalytic system. Unlike complex chiral ligands that may have long lead times, these components are commodity chemicals. This availability reduces the risk of production stoppages due to raw material shortages. Moreover, the simplified process flow reduces the number of critical control points, making the overall supply chain more resilient to operational disruptions.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions and avoids the generation of heavy metal waste streams, aligning with increasingly stringent environmental regulations. The ease of catalyst recovery via crystallization simplifies waste management and reduces the burden on effluent treatment plants. This environmental compatibility facilitates smoother regulatory approvals for scale-up, allowing manufacturers to expand capacity from pilot scales to multi-ton production without significant infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific performance data and procedural details outlined in the patent documentation to provide clarity on feasibility and operational parameters. Understanding these aspects is crucial for technical teams evaluating the transition from legacy processes to this advanced catalytic system.
Q: How does the modified organoaluminum catalyst improve isopulegol selectivity compared to traditional Lewis acids?
A: The modified catalyst utilizes a specific ligand structure derived from 2,6-dimethylpyridine and ketones, which creates significant steric hindrance around the aluminum center. This spatial arrangement favors the formation of the desired isopulegol isomer over neo-isopulegol, achieving selectivity rates exceeding 98%, significantly higher than traditional ZnBr2 catalysts.
Q: What are the advantages of the chiral resolution method described in the patent?
A: The patent describes a chemical induction method using (S)-α-phenethylamine to form diastereomeric salts. This approach avoids partial racemization often seen in other methods and allows for easy crystallization of the desired enantiomer due to solubility differences, resulting in high optical purity (ee% > 98%) with minimal product loss.
Q: Is the catalyst recovery process scalable for industrial production?
A: Yes, the catalyst is designed for easy recovery. Unlike previous methods requiring quenching and separate ligand recovery, this modified homogeneous catalyst can be recovered via crystallization without destroying its structure. This feature supports continuous reuse, reducing waste and enhancing the economic viability of large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110845305A for the flavor and fragrance sector. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high enantiomeric excess required for premium L-menthol applications. We are committed to delivering consistent quality and reliability for our global partners.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific market needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for high-purity L-menthol and related fine chemical intermediates.
