Advanced Manufacturing of Roxadustat Intermediate via Safe Palladium-Catalyzed Methylation
Advanced Manufacturing of Roxadustat Intermediate via Safe Palladium-Catalyzed Methylation
The pharmaceutical landscape for treating chronic anemia has been significantly reshaped by the development of Hypoxia Inducible Factor (HIF) prolyl hydroxylase inhibitors, most notably Roxadustat (FG-4592). As global demand for this therapeutic agent escalates, the efficiency and safety of its supply chain have become paramount concerns for procurement and R&D leadership. Patent CN110903242B introduces a transformative preparation method for the critical intermediate, 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylate. This innovation addresses longstanding bottlenecks in synthetic efficiency by replacing hazardous reagents with safer, scalable alternatives while maintaining exceptional yield and purity profiles. The structural complexity of Roxadustat necessitates a robust synthetic strategy that balances chemical precision with industrial feasibility.
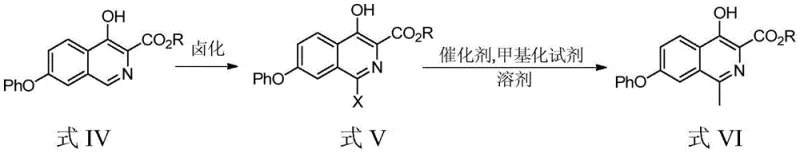
This patented methodology offers a streamlined two-step sequence that bypasses the intricate protection-deprotection cycles and dangerous reagents characteristic of legacy processes. By leveraging a regioselective halogenation followed by a palladium-catalyzed cross-coupling methylation, the process ensures high fidelity in constructing the isoquinoline scaffold. For stakeholders evaluating reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in process chemistry. It not only mitigates the safety risks associated with traditional halogenating agents but also optimizes the cost structure through improved atom economy and reduced waste generation. The following analysis dissects the technical merits and commercial implications of this advanced manufacturing route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key isoquinoline intermediate has been plagued by severe safety hazards and operational inefficiencies that hinder commercial viability. Prior art methods, such as those disclosed in WO2004108681A1, rely heavily on phosphorus oxybromide (POBr3) for bromination, a corrosive liquid that poses significant handling risks and requires specialized containment infrastructure. Furthermore, the subsequent methylation steps in these legacy routes often employ n-butyllithium and methyl iodide, reagents that are pyrophoric and toxic, respectively. These conditions necessitate cryogenic temperatures and inert atmospheres, drastically increasing energy consumption and capital expenditure for specialized reactor systems. Other approaches utilizing phosphorus oxychloride or hydrogen peroxide oxidation introduce similar safety liabilities, including the potential for runaway exothermic reactions and the generation of difficult-to-treat acidic waste streams. The cumulative effect of these drawbacks is a fragile supply chain vulnerable to regulatory scrutiny and production delays.
The Novel Approach
In stark contrast, the process defined in CN110903242B employs a sophisticated yet operationally simple strategy centered on N-halosuccinimides and organoboron chemistry. The initial halogenation utilizes N-bromosuccinimide (NBS) in common solvents like dichloromethane or acetonitrile, eliminating the need for corrosive phosphorus halides. This is followed by a palladium-catalyzed methylation using trimethylboroxine or methylboronic acid, which are stable, non-pyrophoric solids or liquids compared to the dangerous trimethylboron gas used in other methods. This shift in reagent profile fundamentally alters the risk landscape of the manufacturing process. By operating at moderate temperatures (90-110°C) in robust solvents like toluene, the new method facilitates easier heat management and solvent recovery. This approach directly supports cost reduction in pharmaceutical intermediate manufacturing by simplifying the engineering controls required for safe operation and reducing the burden on waste treatment facilities.
Mechanistic Insights into Palladium-Catalyzed Methylation
The core of this technological advancement lies in the precise execution of the methylation step, which constructs the critical C-C bond at the 1-position of the isoquinoline ring. The reaction proceeds via a classic Suzuki-Miyaura cross-coupling mechanism, initiated by the oxidative addition of the palladium(0) catalyst into the carbon-halogen bond of the 1-bromo intermediate. The use of tricyclohexylphosphine as a ligand is particularly noteworthy, as it stabilizes the active palladium species and enhances the rate of transmetallation with the methylboron reagent. This catalytic cycle is highly sensitive to the electronic environment of the substrate; however, the specific conditions outlined in the patent—utilizing potassium phosphate heptahydrate as a base—ensure efficient activation of the boron species without compromising the integrity of the sensitive ester and phenolic functionalities present on the molecule. The result is a highly selective transformation that minimizes the formation of homocoupling byproducts or debrominated impurities.
Impurity control is rigorously managed through the optimization of stoichiometry and reaction parameters. The patent specifies a molar ratio of the bromo-intermediate to the methylating agent between 1:2 and 1:8, ensuring that the equilibrium favors the formation of the desired methyl product. Furthermore, the choice of solvent system, such as toluene or toluene/water mixtures, plays a crucial role in solubilizing the inorganic base while maintaining the organic substrates in solution, thereby maximizing interfacial contact for the catalytic cycle. This mechanistic understanding allows for the consistent production of high-purity pharmaceutical intermediates with purity levels reaching 99.83%. Such high purity is essential for downstream processing, as it reduces the load on crystallization and chromatography steps, ultimately leading to a more economical and environmentally sustainable process.
How to Synthesize 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for replicating this high-yielding process in a GMP-compliant environment. The procedure begins with the dissolution of the precursor isoquinoline in a halogenation solvent, followed by the controlled addition of N-bromosuccinimide to generate the reactive bromo-intermediate. After isolation, this intermediate undergoes the pivotal methylation reaction under nitrogen protection to prevent catalyst oxidation. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and workup procedures, are critical for ensuring batch-to-batch consistency and regulatory compliance.
- Perform halogenation on 4-hydroxy-1-hydrogen-7-phenoxy-3-isoquinoline carboxylate using N-bromosuccinimide (NBS) in dichloromethane to obtain the 1-bromo intermediate.
- Conduct methylation reaction on the bromo-intermediate using trimethylboroxine, palladium acetate catalyst, and tricyclohexylphosphine ligand in toluene at 90-110°C.
- Purify the final crude product via extraction and column chromatography to achieve purity exceeding 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The transition from hazardous, specialty reagents to commodity chemicals like NBS and trimethylboroxine significantly de-risks the supply chain. These materials are widely available from multiple global vendors, reducing the likelihood of single-source bottlenecks that can disrupt production schedules. Moreover, the elimination of cryogenic conditions and pyrophoric reagents lowers the barrier to entry for contract manufacturing organizations (CMOs), expanding the pool of qualified suppliers capable of executing the synthesis. This flexibility is crucial for maintaining supply chain reliability in the face of geopolitical or logistical uncertainties.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the simplification of unit operations and the reduction of safety-related overhead. By avoiding the need for specialized equipment to handle corrosive phosphorus halides or pyrophoric organolithiums, capital expenditure for plant setup is substantially lowered. Additionally, the high reaction yields (up to 88% in the methylation step) mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The use of recyclable solvents like toluene and dichloromethane further enhances the economic profile by allowing for efficient solvent recovery loops, minimizing purchase and disposal costs.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater predictability in manufacturing lead times. Traditional methods prone to exotherms or sensitivity to moisture often suffer from batch failures or extended cycle times due to rigorous drying requirements. In contrast, the tolerance of this new method to hydrated bases (e.g., potassium phosphate heptahydrate) simplifies raw material handling and reduces the risk of batch rejection. This operational stability ensures a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers, supporting just-in-time inventory strategies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns with modern green chemistry principles. The replacement of toxic methyl iodide and corrosive POBr3 reduces the generation of hazardous waste, simplifying effluent treatment and lowering compliance costs. The ability to run the reaction at atmospheric pressure and moderate temperatures facilitates straightforward commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton production reactors. This scalability ensures that the supply can grow in tandem with market demand for Roxadustat without requiring prohibitive infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: How does this new method improve safety compared to prior art using phosphorus oxybromide?
A: The novel process replaces hazardous phosphorus oxybromide (POBr3) and n-butyllithium with solid N-bromosuccinimide (NBS) and stable trimethylboroxine. This eliminates the risks associated with corrosive liquids and pyrophoric reagents, significantly enhancing operational safety during industrial scale-up.
Q: What yields and purity levels can be expected from this synthesis route?
A: Experimental data demonstrates that the halogenation step achieves yields up to 90.7%, while the subsequent methylation step reaches yields of approximately 88.01%. The final product purity consistently exceeds 99.8%, minimizing the need for extensive downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available raw materials and avoids extreme conditions such as cryogenic temperatures or high-pressure hydrogenation. The use of robust solvents like toluene and dichloromethane facilitates easy solvent recovery and waste management, making it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex therapeutics like Roxadustat depends on a partnership grounded in technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from development to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards. Our commitment to process innovation allows us to offer solutions that are not only chemically superior but also economically optimized for the long term.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific financial benefits applicable to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, enabling you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
