Advanced Metal-Free Synthesis of Benzothiophene Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable synthetic routes for heterocyclic scaffolds, particularly benzothiophene derivatives which serve as critical pharmacophores in numerous therapeutic agents. A significant technological breakthrough in this domain is documented in Chinese Patent CN111675690A, which discloses a novel, metal-free methodology for constructing the benzothiophene core. This patent introduces a streamlined one-step radical cyclization strategy that utilizes di-tert-butyl peroxide (DTPB) as a free radical initiator in the presence of disodium hydrogen phosphate. Unlike traditional approaches that rely heavily on precious transition metal catalysts, this innovation leverages a simple thermal radical mechanism in acetone solvent. For R&D directors and process chemists, this represents a paradigm shift towards greener, more cost-effective manufacturing protocols that eliminate the burden of heavy metal removal. The structural versatility of this method is evident in the general formula provided, where various substituents can be tolerated on the aromatic rings, allowing for the rapid generation of diverse compound libraries for drug discovery programs.
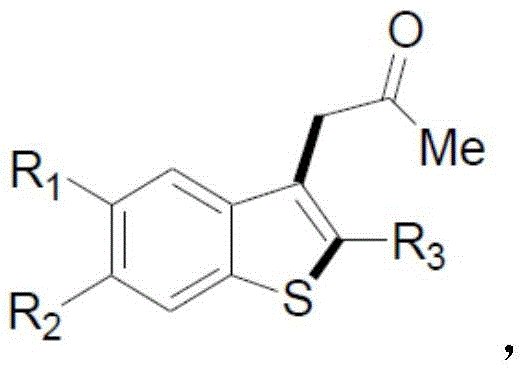
For procurement managers and supply chain heads, the implications of adopting the technology described in CN111675690A are profound. The reliance on commodity chemicals such as acetone and inorganic phosphates, rather than specialized organometallic complexes, drastically simplifies the raw material sourcing landscape. This transition not only mitigates the volatility associated with precious metal pricing but also enhances the overall resilience of the supply chain. Furthermore, the operational simplicity of the process—requiring only standard heating and stirring under nitrogen protection—suggests that existing manufacturing infrastructure can be utilized without significant capital expenditure on specialized reactors. This accessibility positions the technology as a highly viable candidate for immediate commercial scale-up, offering a reliable pathway for producing high-purity pharmaceutical intermediates with reduced lead times and lower operational overheads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiophene derivatives has been dominated by transition metal-catalyzed cyclization reactions, often employing palladium, copper, or gold complexes. While these methods can achieve high yields under mild conditions, they suffer from significant drawbacks that hinder their utility in large-scale industrial applications. The primary concern is the introduction of toxic heavy metal residues into the final product, which necessitates complex and costly purification steps to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). Additionally, the catalysts themselves, particularly gold-based systems like Au(I)-IPr, are prohibitively expensive and sensitive to reaction conditions, requiring rigorous exclusion of moisture and oxygen. Alternative electrophilic cyclization methods using halogens like iodine or bromine generate substantial amounts of hazardous halogenated waste, posing serious environmental compliance challenges and increasing waste disposal costs. These factors collectively inflate the cost of goods sold (COGS) and extend the production timeline, creating bottlenecks for manufacturers aiming to bring new benzothiophene-based drugs to market efficiently.
The Novel Approach
The methodology presented in the patent data offers a compelling alternative by replacing transition metals with an organic peroxide initiator, DTPB. This metal-free approach fundamentally alters the reaction landscape, enabling the intramolecular cyclization of 2-ethynylphenyl methyl sulfides through a radical mechanism. By utilizing acetone as the solvent, the process benefits from a medium that is not only inexpensive and widely available but also participates favorably in the radical propagation steps. The reaction proceeds smoothly at elevated temperatures between 120°C and 140°C, specifically optimized around 130°C to 135°C, ensuring high conversion rates without the need for exotic ligands or sensitive catalytic systems. This simplification of the reaction matrix means that the workup procedure is straightforward, typically involving basic extraction and chromatography, which significantly reduces the complexity of downstream processing. The result is a cleaner reaction profile with fewer by-products, directly translating to higher isolated yields and superior product quality suitable for sensitive pharmaceutical applications.
Mechanistic Insights into DTPB-Mediated Radical Cyclization
The core of this innovative synthesis lies in the generation of sulfur-centered radicals that trigger the cyclization cascade. Upon thermal decomposition, DTPB generates tert-butoxy radicals which abstract a hydrogen atom or interact with the sulfur moiety of the 2-ethynylphenyl methyl sulfide substrate. This initiation step creates a reactive radical species on the sulfur atom or the adjacent carbon framework, which then undergoes an intramolecular addition to the alkyne triple bond. The presence of disodium hydrogen phosphate plays a crucial role as a mild base or buffer, potentially stabilizing intermediate species or facilitating proton transfer steps necessary for the aromatization of the thiophene ring. This mechanistic pathway avoids the formation of stable metal-carbon bonds, thereby eliminating the risk of metal leaching into the product stream. The radical nature of the reaction also imparts a degree of tolerance to various functional groups on the aromatic rings, as evidenced by the successful synthesis of derivatives containing fluorine, methoxy, and bromine substituents. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the stoichiometric ratio of DTPB, to maximize efficiency while minimizing radical-induced side reactions.
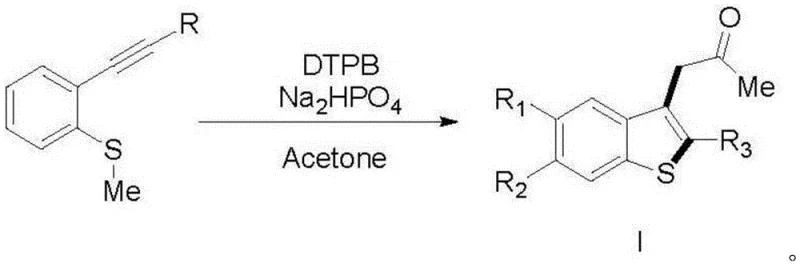
Controlling impurities in radical reactions is often a challenge due to the potential for polymerization or non-selective radical coupling. However, the specific conditions outlined in the patent, particularly the controlled temperature range of 130°C to 135°C and the optimized reaction time of 22 to 25 hours, appear to strike a delicate balance. Operating below 120°C results in incomplete conversion, leaving starting materials that complicate purification, while exceeding 25 hours or higher temperatures can lead to the degradation of the peroxide initiator and the formation of tarry by-products. The use of acetone as a solvent also aids in solubilizing both the organic substrate and the inorganic phosphate salt to some extent, creating a homogeneous or semi-homogeneous reaction environment that promotes consistent radical propagation. This precise control over the reaction kinetics ensures that the desired benzothiophene scaffold is formed with high selectivity, minimizing the formation of regioisomers or over-oxidized species. For quality control teams, this predictability is invaluable, as it leads to a consistent impurity profile that is easier to characterize and control during routine manufacturing batches.
How to Synthesize 3-Acetylbenzothiophene Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings. The protocol involves charging a reactor with the 2-ethynylphenyl methyl sulfide precursor, followed by the addition of DTPB and disodium hydrogen phosphate in acetone. The mixture is then heated under a nitrogen atmosphere to maintain an inert environment, preventing unwanted oxidation of the radical intermediates by atmospheric oxygen. After the designated reaction period, the crude product is isolated via liquid-liquid extraction using dichloromethane, dried, and concentrated. Final purification is achieved through column chromatography using a petroleum ether and ethyl acetate gradient. This straightforward workflow minimizes the need for specialized equipment or hazardous reagents, making it an attractive option for process development teams looking to establish robust manufacturing procedures. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to assist technical teams in replicating these results.
- Charge a reactor with 2-ethynylphenyl methyl sulfide derivative, DTPB (4.0 equiv), and disodium hydrogen phosphate (1.0 equiv) in acetone solvent.
- Heat the reaction mixture to 130-135°C under a nitrogen atmosphere and stir for 22-25 hours to ensure complete cyclization.
- Extract the crude product with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this metal-free synthesis route offers substantial strategic advantages for procurement and supply chain management within the fine chemical sector. The most immediate benefit is the drastic reduction in raw material costs associated with eliminating precious metal catalysts. Palladium and gold catalysts not only carry high price tags but also require complex recovery and recycling processes to be economically viable, adding layers of operational complexity. By switching to DTPB and inorganic salts, manufacturers can secure a more stable and predictable cost structure, insulating themselves from the volatility of the precious metals market. Furthermore, the use of acetone, a bulk commodity solvent, simplifies logistics and storage requirements compared to specialized anhydrous solvents often needed for organometallic chemistry. This simplification extends to the waste stream as well; the absence of heavy metals means that waste disposal is less regulated and less expensive, contributing to overall cost reduction in manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive scavenging resins or activated carbon treatments typically required to reduce metal residues to ppm levels in API intermediates. This simplification of the downstream processing train significantly lowers the consumption of auxiliary materials and reduces the labor hours dedicated to purification. Additionally, the high atom economy of the cyclization reaction, combined with yields reaching up to 80% for certain substrates, ensures that raw material utilization is maximized. The cumulative effect of these factors is a leaner production process with a lower cost base, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can sometimes be subject to geopolitical constraints or supply disruptions, whereas DTPB and sodium phosphate are produced on a massive scale for various industrial applications. This abundance ensures a continuous and reliable supply of key reagents, minimizing the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing greater flexibility in vendor selection. For supply chain heads, this reliability translates into improved planning accuracy and the ability to maintain consistent inventory levels of critical intermediates without the need for excessive safety stocks.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, requiring only heat and stirring in a common solvent, makes this process inherently scalable from gram to ton quantities without significant re-engineering. The absence of halogenated reagents and heavy metals aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance reduces the regulatory burden on the manufacturing site and lowers the risk of environmental fines or shutdowns. Moreover, the use of acetone, which is biodegradable and easier to recover than many chlorinated solvents, further enhances the environmental profile of the process, supporting corporate sustainability goals and improving the company's standing with eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzothiophene synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the long-term viability of the supply chain. The clarity provided here aims to bridge the gap between academic innovation and industrial application, ensuring that all stakeholders have a comprehensive understanding of the process capabilities and limitations.
Q: Does this synthesis method involve transition metal catalysts?
A: No, the method described in patent CN111675690A utilizes DTPB as a free radical initiator instead of expensive transition metals like Palladium or Gold, significantly reducing heavy metal impurity risks.
Q: What is the optimal reaction temperature for this cyclization?
A: The patent data indicates that controlling the temperature between 130°C and 135°C provides the best balance between reaction rate and yield, avoiding decomposition associated with higher temperatures.
Q: Can this method be scaled for industrial production?
A: Yes, the use of acetone as both a reactant component and solvent, along with common inorganic salts, simplifies the supply chain and makes the process highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free radical cyclization technology described in CN111675690A for the production of high-value benzothiophene intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped to handle radical reactions safely and efficiently, with rigorous QC labs dedicated to maintaining stringent purity specifications that exceed industry standards. We understand that the transition to a new synthetic route requires confidence in the partner's technical capability, and our team of expert process chemists is ready to optimize this methodology for your specific portfolio of compounds, ensuring seamless technology transfer and rapid time-to-market.
We invite you to collaborate with us to leverage this advanced synthesis platform for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free process for your specific needs. We encourage you to contact our technical procurement team today to request specific COA data for our benzothiophene library and to discuss route feasibility assessments tailored to your development timelines. Let us help you streamline your supply chain and reduce your manufacturing costs with our proven expertise in heterocyclic chemistry.
