Scalable Synthesis of Spiro Cyclopropane Indene Diketones via Novel Rare Earth Silicon-Amino Catalysis for Global Pharma Supply Chains
Introduction to Next-Generation Spiro Compound Synthesis
The pharmaceutical and agrochemical industries continuously demand more efficient, safer, and environmentally benign pathways for constructing complex molecular scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN110028407B, which discloses a highly innovative method for preparing spiro[cyclopropane-1,2'-indene]-1',3'-diketone compounds. This specific structural motif is prevalent in numerous bioactive natural products and drug candidates, serving as a critical core for antibacterial agents and pesticides. The patent introduces a novel catalytic system utilizing silicon-amino rare earth compounds, specifically [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3, to drive a one-pot cascade reaction. This approach represents a paradigm shift from conventional multi-step syntheses, offering a streamlined route that combines alpha-carbonyl esters, phosphites, and 2-substituted methylene-1,3-indene diones under mild conditions. For R&D directors and procurement strategists, this technology promises not only high chemical efficiency but also substantial improvements in process safety and scalability, addressing key pain points in the manufacturing of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the spiro[cyclopropane-1,2'-indene]-1',3'-diketone skeleton has been fraught with significant technical and safety challenges. Prior art synthetic routes generally fall into three categories, each carrying distinct disadvantages that hinder large-scale commercial adoption. The first common method involves the reaction of diazo compounds with olefins catalyzed by transition metals. This approach is inherently dangerous due to the explosive nature of diazo reagents, requiring specialized equipment and rigorous safety protocols that escalate operational costs. The second method relies on sulfur, phosphorus, or ammonium ylides reacting with activated double bonds under alkaline conditions. These reactions often necessitate the use of toxic reagents, such as arsenic-containing substances, posing severe environmental and health hazards that complicate waste disposal and regulatory compliance. Furthermore, the third method, involving alpha-halogenated active methylene compounds, typically requires harsh basic conditions that can lead to poor functional group tolerance. Crucially, these traditional pathways frequently suffer from low stereoselectivity, yielding mixtures of isomers that require expensive and time-consuming chromatographic separation, thereby reducing overall process throughput and increasing the cost of goods sold.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN110028407B offers a transformative solution through a rare earth-catalyzed one-pot synthesis. This novel approach eliminates the need for hazardous diazo reagents, toxic ylides, or strong stoichiometric bases. By employing a silicon-amino rare earth catalyst, the reaction proceeds under significantly milder conditions, typically between 60°C and 84°C, which reduces energy consumption and thermal stress on the equipment. The process is designed as a telescoped sequence where an active intermediate is generated in situ from the alpha-carbonyl ester and phosphite, which then immediately reacts with the 2-substituted methylene-1,3-indene dione. This integration of steps not only simplifies the operational workflow but also dramatically enhances atom economy. The result is a robust protocol capable of delivering the target spiro compounds with high yields and excellent stereoselectivity, specifically favoring the trans-configuration, which is often the biologically active form. This leap in process design directly translates to a more reliable supply chain for complex pharmaceutical intermediates.
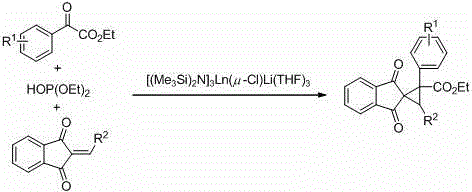
Mechanistic Insights into Rare Earth Silicon-Amino Catalysis
The core of this technological advancement lies in the unique structure and reactivity of the silicon-amino rare earth catalyst. The general formula [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3 features a central trivalent rare earth metal ion (Ln), which acts as a potent Lewis acid to activate the carbonyl and phosphite substrates. The ligand environment, composed of bulky trimethylsilylamino groups and a bridging chloride, creates a specific steric and electronic pocket that governs the stereochemical outcome of the reaction. Among the various rare earth ions tested, including Neodymium, Gadolinium, Erbium, and Ytterbium, Lanthanum (La) has been identified as the most effective metal center. The Lanthanum-based catalyst demonstrates superior ability to coordinate the reactants and stabilize the transition state leading to the cyclopropane ring formation. This precise coordination is critical for achieving the high diastereomeric ratios observed, often exceeding 90:10 in favor of the trans-isomer, thereby minimizing the formation of unwanted byproducts.
![Chemical structure of the silicon-amino rare earth catalyst [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3](/insights/img/spiro-cyclopropane-indene-diketone-rare-earth-catalyst-pharma-supplier-20260305165502-01.png)
Furthermore, the mechanism involves a delicate balance of nucleophilic addition and cyclization steps that are highly sensitive to the reaction medium. The patent data highlights that the solvent plays a pivotal role in modulating the catalyst's activity and the stability of the intermediates. 1,2-Dichloroethane emerges as the solvent of choice, outperforming alternatives like toluene, acetonitrile, or DMF. This is attributed to its optimal polarity, which facilitates the solubility of the polar catalyst species while supporting the necessary addition-cyclization pathway. In non-optimal solvents, the reaction kinetics slow down, and the stereoselectivity drops, leading to mixed products. Understanding this mechanistic nuance allows process chemists to tightly control impurity profiles. By maintaining strict anhydrous and oxygen-free conditions, typically under an inert argon atmosphere, the catalyst retains its activity throughout the 8 to 36-hour reaction window, ensuring consistent batch-to-batch quality essential for GMP manufacturing environments.
How to Synthesize Spiro[cyclopropane-1,2'-indene]-1',3'-diketone Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the sequential addition of reagents to maximize the formation of the active intermediate. The process begins with the synthesis of the silicon-amino rare earth catalyst itself, which involves reacting lithium bis(trimethylsilyl)amide with anhydrous rare earth chlorides in THF, followed by purification to remove lithium chloride salts. Once the catalyst is prepared, the main transformation is executed in a one-pot fashion. First, the catalyst is mixed with diethyl phosphite and the alpha-carbonyl ester at room temperature to generate the reactive species. Subsequently, the 2-substituted methylene-1,3-indene dione and the optimized solvent, 1,2-dichloroethane, are introduced. The mixture is then heated to promote the cyclization. Detailed standardized operating procedures, including specific molar ratios and workup protocols involving ethyl acetate extraction and silica gel chromatography, are critical for reproducing the high yields reported in the patent literature.
- Prepare the silicon-amino rare earth catalyst [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3 under anhydrous and oxygen-free conditions using LnCl3 and lithium bis(trimethylsilyl)amide.
- In a dehydrated reaction flask under argon, mix the catalyst, diethyl phosphite, and alpha-carbonyl ester, stirring at room temperature to form an active intermediate.
- Add 2-substituted methylene-1,3-indene dione and 1,2-dichloroethane solvent, then heat to 60-84°C for 8-36 hours to complete the cyclization and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rare earth-catalyzed methodology offers compelling strategic advantages that extend beyond mere chemical novelty. The primary value driver is the drastic simplification of the manufacturing process. By consolidating what was traditionally a multi-step sequence into a single pot, the number of unit operations is significantly reduced. This reduction directly correlates to lower capital expenditure on equipment, decreased labor hours, and minimized solvent usage, all of which contribute to a leaner cost structure. Moreover, the elimination of hazardous reagents like diazo compounds removes the need for expensive explosion-proof facilities and specialized waste treatment protocols, further driving down the total cost of ownership. The mild reaction conditions also imply lower energy consumption for heating and cooling, aligning with modern sustainability goals and reducing the carbon footprint of the production facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are rooted in the replacement of expensive noble metal catalysts and toxic reagents with a more accessible rare earth system. The catalyst loading is relatively low, ranging from 25% to 30% molar ratio relative to the substrate, which is economically viable given the high turnover and yield. Additionally, the high stereoselectivity means that less material is wasted on separating unwanted isomers, effectively increasing the yield of the desired active pharmaceutical ingredient (API) intermediate. This efficiency gain allows for a more competitive pricing model for the final spiro compounds, providing a buffer against raw material price fluctuations and enhancing margin stability for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on scarce or regulated reagents. This new method utilizes alpha-carbonyl esters and phosphites, which are commodity chemicals with robust global supply chains. The catalyst components, based on lanthanum or other abundant rare earths, are also readily available from established suppliers, mitigating the risk of bottlenecks associated with exotic transition metals. The simplicity of the one-pot procedure also reduces the likelihood of operational errors or batch failures, ensuring a steady flow of high-purity intermediates. This reliability is crucial for pharmaceutical companies managing tight production schedules and regulatory filings, where consistency is paramount.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often exposes hidden inefficiencies, but this methodology is inherently scalable. The use of 1,2-dichloroethane, while requiring proper handling, is a standard industrial solvent with well-established recovery and recycling protocols. The absence of heavy metal contamination in the final product simplifies the purification process, reducing the burden on quality control labs to perform extensive metal scavenging tests. From an environmental perspective, the avoidance of arsenic and phosphorus ylides significantly lowers the toxicity profile of the waste stream, facilitating easier compliance with increasingly stringent environmental regulations. This 'green' advantage not only protects the company's reputation but also future-proofs the manufacturing asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims within patent CN110028407B, providing a factual basis for evaluating its potential integration into your existing manufacturing portfolio. Understanding these specifics helps in making informed decisions about process development and vendor selection.
Q: What are the primary advantages of this rare earth catalytic method over traditional diazo-based routes?
A: Unlike traditional methods that rely on hazardous diazo compounds or toxic arsenic/phosphorus ylides, this patent-disclosed method utilizes mild reaction conditions and avoids noble metals. It achieves high stereoselectivity (trans-configuration) through a one-pot process, significantly reducing safety risks and purification complexity.
Q: Which rare earth metal provides the optimal catalytic performance for this transformation?
A: While the catalyst framework supports various trivalent ions like Neodymium, Gadolinium, Erbium, and Ytterbium, experimental data indicates that Lanthanum (La) delivers superior yields and stereoselectivity. The Lanthanum-based catalyst [(Me3Si)2N]3La(μ-Cl)Li(THF)3 is preferred for maximizing reaction efficiency.
Q: How does the choice of solvent impact the reaction yield and stereoselectivity?
A: Solvent polarity and solubility are critical. 1,2-Dichloroethane is identified as the optimal solvent due to its ability to dissolve both the catalyst and substrates effectively while facilitating the addition-cyclization of the active intermediate. Other solvents like toluene or DMF result in noticeably lower yields and reduced diastereomeric ratios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[cyclopropane-1,2'-indene]-1',3'-diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rare earth silicon-amino system described in CN110028407B. As a leading CDMO partner, we possess the technical expertise to translate such complex laboratory methodologies into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high stereoselectivity and purity specifications achieved in the lab are maintained at the plant level. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and impurity profiles of every batch, guaranteeing that our spiro[cyclopropane-1,2'-indene]-1',3'-diketone intermediates meet the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next-generation drug candidates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal on how we can optimize your supply chain for these high-value intermediates. Let us help you turn this innovative chemistry into a commercial reality.
