Efficient Synthesis of Spiro Cyclopropane Indene Diketones for Pharmaceutical Applications
Efficient Synthesis of Spiro Cyclopropane Indene Diketones for Pharmaceutical Applications
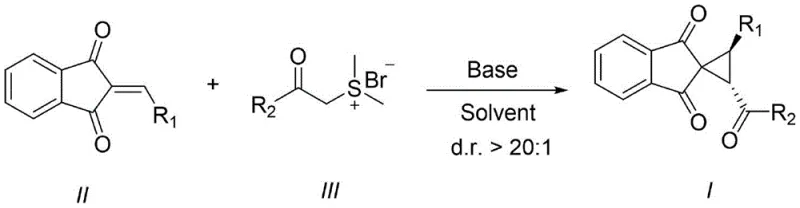
The development of efficient synthetic routes for highly strained cyclic systems remains a pivotal challenge in modern medicinal chemistry, particularly for constructing bioisosteres that enhance metabolic stability. Patent CN113264816A introduces a groundbreaking methodology for the preparation of spiro[cyclopropane-1,2'-indene]-1',3'-diketone derivatives, which serve as critical scaffolds in the design of novel therapeutic agents. This technology leverages a formal [2+1] cycloaddition strategy between substituted 2-ethylidene-1H-indene-1,3(2H)-diones and sulfur ylides under mild basic conditions. By addressing the limitations of previous catalytic systems, this invention provides a robust platform for generating high-purity intermediates essential for drug discovery pipelines. The significance of this advancement lies not only in its chemical elegance but also in its potential to streamline the supply chain for complex pharmaceutical building blocks.
![General structural formula of spiro[cyclopropane-1,2'-indene]-1',3'-diketone derivatives showing variable R1 and R2 groups](/insights/img/spiro-cyclopropane-synthesis-pharma-supplier-20260304182354-01.png)
For procurement specialists and supply chain managers, the implications of this patent are profound, offering a pathway to reliable pharmaceutical intermediate supplier status through simplified processing. The ability to access these structurally complex motifs without relying on precious metal catalysts translates directly into reduced raw material costs and simplified waste management protocols. Furthermore, the broad substrate tolerance described in the documentation ensures that a diverse library of analogs can be produced using a unified operational framework, thereby enhancing the agility of R&D teams responding to evolving biological data. This report analyzes the technical merits and commercial viability of this synthesis, highlighting its capacity for commercial scale-up of complex intermediates while maintaining stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spirocyclopropane rings has relied heavily on diazo compound decomposition or metal-carbene insertion strategies, which often present significant safety and environmental hazards. Traditional methods reported in literature, such as those by Roy, frequently suffer from poor diastereoselectivity, necessitating cumbersome chromatographic separations that drastically reduce overall throughput and increase production costs. Additionally, alternative approaches developed by researchers like Maleki have required the use of sophisticated nanomagnetic organocatalysts functionalized with L-proline, which introduce complexity in catalyst recovery and potential metal contamination issues. These legacy techniques often demand harsh reaction conditions or specialized equipment, creating bottlenecks in the manufacturing of high-purity OLED material or pharmaceutical precursors. The reliance on transition metals also triggers rigorous heavy metal testing requirements in the final active pharmaceutical ingredient, adding layers of analytical burden and risk to the quality control process.
The Novel Approach
In stark contrast, the methodology disclosed in CN113264816A utilizes a metal-free, base-promoted cyclization that operates under remarkably mild conditions, typically ranging from 0°C to 100°C. This innovative route employs readily available sulfur ylides as the carbene equivalent, reacting them with electron-deficient alkenes to forge the cyclopropane ring with exceptional stereocontrol. The process eliminates the need for expensive transition metal catalysts, thereby removing the associated costs of catalyst licensing, removal, and disposal. By achieving a diastereomer ratio (d.r.) greater than 20:1, the method significantly minimizes the formation of unwanted isomers, leading to higher effective yields and reduced solvent consumption during purification. This streamlined approach represents a paradigm shift in cost reduction in API manufacturing, allowing producers to deliver high-value intermediates with superior economic efficiency and environmental compliance.
Mechanistic Insights into Base-Catalyzed Cyclopropanation
The core of this synthetic breakthrough lies in the generation and reactivity of the sulfur ylide species, which acts as a nucleophile towards the activated double bond of the indandione derivative. Upon deprotonation by a suitable base such as cesium carbonate or triethylamine, the sulfonium salt generates a reactive ylide that undergoes a conjugate addition to the beta-position of the alpha,beta-unsaturated ketone system. This initial Michael-type addition creates a zwitterionic intermediate that subsequently collapses via an intramolecular SN2 displacement, expelling the dimethyl sulfide leaving group and closing the three-membered ring. This concerted yet stepwise mechanism ensures that the stereochemical information is preserved and amplified, resulting in the observed high diastereoselectivity favoring the (2S,3R) or (2S,3S) configurations depending on the specific substrate geometry. Understanding this mechanistic pathway is crucial for R&D directors aiming to optimize reaction parameters for specific analogs within the extensive scope defined by the patent.
From an impurity control perspective, the absence of radical pathways or metal-mediated side reactions greatly simplifies the impurity profile of the crude product. Traditional carbene insertions often generate oligomeric byproducts or insertion products at unintended sites, whereas this ionic mechanism is highly chemoselective for the activated alkene. The primary byproduct, dimethyl sulfide, is volatile and easily removed during the concentration or drying steps, preventing it from persisting into the final isolation stage. This cleanliness of reaction allows for simpler workup procedures, often requiring only aqueous washes and crystallization or standard flash chromatography. For quality assurance teams, this predictability means that validation of the cleaning process and analytical method development are more straightforward, reducing the time-to-market for new drug candidates incorporating this spirocyclic scaffold.
How to Synthesize Spiro[cyclopropane-1,2'-indene]-1',3'-diketone Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, making it suitable for both laboratory discovery and pilot plant production. The general procedure involves charging a reaction vessel with the substituted 2-ethylidene-1H-indene-1,3(2H)-dione and the corresponding sulfur ylide precursor in a molar ratio of approximately 1:1. A stoichiometric amount of base, typically 1.2 equivalents, is added along with a solvent such as hexafluoroisopropanol (HFIP), acetonitrile, or dichloromethane to facilitate the reaction. The mixture is stirred at ambient or slightly elevated temperatures until conversion is complete, as monitored by TLC or HPLC. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with substituted 2-ethylidene-1H-indene-1,3(2H)-dione, substituted sulfur ylide, and a suitable base such as Cs2CO3.
- Add an appropriate organic solvent like HFIP or acetonitrile and stir the mixture at a temperature between 0°C and 100°C.
- Upon completion, quench with water or brine, extract with organic solvent, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic advantages beyond mere chemical yield. The elimination of precious metal catalysts removes a significant variable from the raw material cost structure, insulating the production budget from the volatility of rhodium, palladium, or iridium markets. Furthermore, the use of commodity chemicals like inorganic bases and common organic solvents ensures a stable and diversified supply base, reducing the risk of single-source dependency. The high diastereoselectivity inherently reduces the volume of waste generated per kilogram of product, aligning with increasingly stringent environmental regulations and lowering waste disposal fees. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from metal-catalyzed processes to this base-mediated protocol fundamentally alters the cost drivers of production. By avoiding the procurement of expensive chiral ligands or metal salts, the direct material cost is significantly lowered. Additionally, the simplified purification process, driven by high selectivity, reduces the consumption of silica gel and eluents, which are often major cost centers in intermediate manufacturing. The operational simplicity also translates to lower labor hours per batch, as complex catalyst activation or inert atmosphere handling may be minimized. This holistic reduction in operational expenditure allows for more competitive pricing models when supplying high-purity spiro cyclopropane derivatives to downstream partners.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as cesium carbonate, potassium carbonate, or triethylamine ensures that production schedules are not disrupted by niche reagent shortages. Unlike specialized nano-catalysts that may have long lead times or limited suppliers, the inputs for this reaction are standard inventory items for most chemical distributors. This accessibility enhances the reliability of delivery timelines, a critical metric for supply chain heads managing just-in-time inventory for clinical trial materials. Moreover, the robustness of the reaction conditions means that technology transfer between different manufacturing sites can be executed with minimal friction, ensuring continuity of supply across global operations.
- Scalability and Environmental Compliance: The mild reaction temperatures (0°C to 100°C) and ambient pressure conditions make this process inherently safer and easier to scale from gram to ton quantities. There is no need for specialized high-pressure reactors or cryogenic cooling infrastructure, allowing utilization of existing multipurpose reactor trains. From an environmental standpoint, the absence of heavy metals simplifies the effluent treatment process, reducing the load on wastewater treatment facilities and ensuring compliance with discharge limits. The generation of volatile dimethyl sulfide as the only stoichiometric byproduct allows for easy capture and scrubbing, minimizing the environmental footprint of the manufacturing campaign and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing process development workflows.
Q: What is the diastereoselectivity of this synthesis method?
A: The method described in patent CN113264816A achieves excellent diastereoselectivity, with a diastereomer ratio (d.r.) consistently greater than 20:1 across various substrates.
Q: Does this process require transition metal catalysts?
A: No, this novel approach utilizes simple inorganic or organic bases (e.g., Cs2CO3, Et3N) instead of complex transition metal catalysts, simplifying downstream purification.
Q: What is the substrate scope for R1 and R2 groups?
A: The reaction demonstrates wide adaptability, tolerating diverse substituents including alkyl, aryl, halogenated phenyl, nitro, cyano, and heterocyclic groups on both the indandione and sulfur ylide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Cyclopropane Indene Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN113264816A for advancing drug discovery programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and base-catalyzed reactions required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to meet the exacting demands of the pharmaceutical industry. We are committed to delivering high-quality intermediates that accelerate your timeline to clinical milestones.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this metal-free route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted provider of advanced chemical solutions. Let us collaborate to bring your next-generation therapeutics to life with efficiency and precision.
