Advanced Synthesis of Substituted Enamine Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly oxazole derivatives which serve as critical building blocks for bioactive molecules. Patent CN111848450A introduces a groundbreaking approach to synthesizing novel substituted enamine compounds, specifically addressing the limitations of traditional oxidative coupling techniques. This technology enables the efficient production of acyloxyenamine intermediates, which are pivotal precursors for generating oxazole rings found in numerous therapeutic agents ranging from anti-inflammatory drugs to HIV inhibitors. By leveraging hypervalent iodine chemistry in conjunction with formic acid, this invention provides a pathway that is not only chemically elegant but also operationally superior for large-scale manufacturing environments.
The significance of this patent lies in its ability to bypass the hazardous and unstable reagents historically associated with enamine functionalization. Traditional routes often rely on oxidants that pose significant safety risks or suffer from poor solubility profiles, leading to inconsistent batch quality. In contrast, the methodology described in CN111848450A utilizes commercially available starting materials and proceeds under remarkably mild conditions, typically at room temperature. This represents a substantial leap forward for process chemists aiming to streamline the supply chain for high-purity pharmaceutical intermediates while adhering to stringent environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyloxyenamines has been plagued by significant technical hurdles that hinder efficient commercial production. One prevalent method involves the use of iodosobenzene as an oxidant for the intermolecular oxidative coupling of enamines with carboxylic acids. However, iodosobenzene is notoriously unstable, presenting severe explosion hazards and flammability risks that complicate storage and handling in industrial settings. Furthermore, its poor solubility in common organic solvents often results in heterogeneous reaction mixtures, leading to erratic reaction kinetics and unstable yields that are unacceptable for GMP manufacturing. Another existing approach utilizes phenyl iodine (III) diacetate (PIDA) in 2,2,2-trifluoroethanol (TFE); while safer, this method is narrowly scoped and fails to produce diverse acyloxyenamines such as formyl or benzoyl derivatives in high yields, limiting its utility for broad API intermediate synthesis.
The Novel Approach
The innovative process disclosed in the patent overcomes these deficiencies by introducing a versatile three-component reaction system. By reacting an enamine substrate with a hypervalent iodine reagent, such as bis(acetoxy)iodobenzene, in the presence of formic acid and a polar solvent, the method achieves direct formylation or acyloxylation with exceptional efficiency. This approach eliminates the need for dangerous iodosobenzene and expands the substrate scope far beyond what was previously possible with PIDA alone. The reaction proceeds smoothly at ambient temperatures, avoiding the energy costs associated with heating or cooling, and produces the target substituted enamine compounds with high purity and minimal byproduct formation. This strategic shift allows manufacturers to access a wider range of chemical space for drug discovery and development.
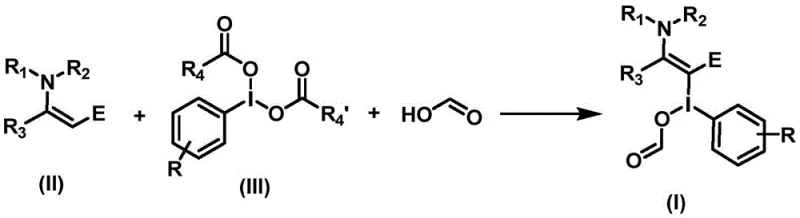
This generalized reaction scheme illustrates the core transformation where the enamine double bond is functionalized via the hypervalent iodine species. The use of formic acid serves a dual purpose, acting both as a reactant to introduce the formyl group and as a promoter for the ligand exchange on the iodine center. This mechanistic nuance ensures that the acyloxy group is installed regioselectively, providing the precise structural motif required for downstream cyclization into oxazoles. The flexibility of the R groups on the enamine and the iodine reagent further underscores the adaptability of this platform for creating diverse libraries of intermediates.
Mechanistic Insights into Hypervalent Iodine-Mediated Oxidative Coupling
The underlying mechanism of this transformation involves the generation of a reactive hypervalent iodine(III) species in situ, which acts as a potent electrophile towards the electron-rich enamine double bond. Upon mixing the bis(acetoxy)iodobenzene with formic acid, a ligand exchange occurs to generate a mixed acetoxy-formyloxy iodine species or a similar active oxidant. This activated complex then coordinates with the enamine nitrogen or attacks the beta-carbon, facilitating the formation of a carbon-iodine bond followed by reductive elimination or nucleophilic substitution that installs the acyloxy functionality. The entire cycle is driven by the strong oxidizing power of the iodine(III) center, which is reduced to iodobenzene, a benign and easily removable byproduct that does not contaminate the final product with toxic heavy metals.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal-catalyzed alternatives. Since no palladium, copper, or other transition metals are required, the risk of metal leaching into the final API is completely eliminated, simplifying the purification workflow and reducing the burden on analytical quality control labs. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups often present in complex enamine substrates. Additionally, the use of polar solvents like TFE or cyclic carbonates helps stabilize the charged intermediates involved in the oxidation process, ensuring consistent reaction rates and minimizing the formation of polymeric side products that can lower overall mass balance.
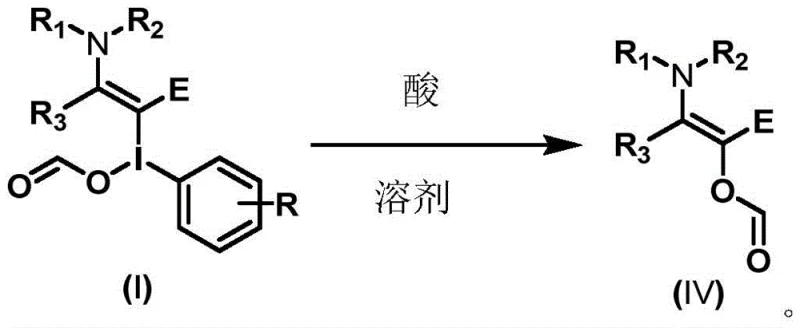
Following the initial synthesis, the resulting substituted enamine compound (I) can be further processed to yield the final acyloxyenamine compound (IV) through a simple acid-mediated reaction. As depicted in the scheme above, treating compound (I) with an organic acid in a non-protic solvent facilitates the displacement of the aryl-iodine moiety, completing the installation of the acyloxy group. This two-step sequence allows for the isolation of stable intermediates if necessary, providing process engineers with greater control over the manufacturing timeline and inventory management.
How to Synthesize Substituted Enamine Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and solvent selection to maximize yield and purity. The process is designed to be user-friendly, operating effectively at room temperature without the need for specialized pressure vessels or inert atmosphere gloveboxes, although standard good manufacturing practices should always be observed. Detailed procedural guidelines regarding reagent addition rates, stirring speeds, and workup protocols are essential for reproducibility. For comprehensive operational instructions, please refer to the standardized synthesis steps provided below which outline the precise molar ratios and reaction times validated in the patent examples.
- React an enamine compound of formula (II) with a hypervalent iodine reagent of formula (III) in a polar solvent.
- Add formic acid to the reaction mixture at room temperature to facilitate the oxidative coupling.
- Stir the mixture until completion to isolate the substituted enamine compound of formula (I) with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible improvements in cost structure and supply reliability. The primary raw materials, including bis(acetoxy)iodobenzene and various enamines, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The elimination of hazardous oxidants like iodosobenzene reduces the costs associated with special handling, storage insurance, and hazardous waste disposal, contributing to a leaner operational budget. Furthermore, the high yields reported in the patent examples, often exceeding 80% and reaching up to 90%, mean that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and the subsequent purification steps required to remove metal residues. The reaction proceeds at room temperature, which drastically lowers energy consumption compared to processes requiring reflux or cryogenic cooling. Additionally, the high atom economy and minimal byproduct formation reduce the volume of solvent and consumables needed for downstream purification, leading to substantial overall savings in manufacturing expenses.
- Enhanced Supply Chain Reliability: By utilizing stable and commercially available reagents, this method ensures a consistent and uninterrupted supply of critical intermediates. The robustness of the reaction against minor variations in conditions means that batch-to-batch variability is minimized, reducing the likelihood of failed batches that could disrupt production schedules. The ability to use a variety of solvents, including green solvents like dimethyl carbonate, provides flexibility to adapt to regional solvent availability and regulatory changes without revalidating the entire process.
- Scalability and Environmental Compliance: The absence of heavy metals and explosive reagents makes this process inherently safer and more environmentally friendly, aligning with modern green chemistry principles. Scaling from gram to kilogram quantities is straightforward due to the exothermic nature being manageable at room temperature, avoiding the heat transfer limitations often encountered in large-scale oxidations. This ease of scale-up facilitates rapid technology transfer from R&D to commercial production, shortening the time to market for new pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel enamine synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the safety advantages of this new enamine synthesis method?
A: Unlike conventional methods using iodosobenzene which is flammable and explosive, this process utilizes stable hypervalent iodine reagents like PIDA under mild conditions, significantly enhancing operational safety.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction operates at room temperature without requiring cryogenic cooling or high-pressure equipment, making it highly suitable for commercial scale-up in standard reactor vessels.
Q: What solvents are compatible with this synthesis route?
A: The method demonstrates versatility with various polar solvents including 2,2,2-trifluoroethanol (TFE), dimethyl carbonate (DMC), and propylene carbonate (PC), allowing for flexible process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Enamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of substituted enamine compounds conforms to the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply chain for your next-generation therapeutics.
