Advanced Manufacturing of Remdesivir Intermediates via Optimized Synthetic Routes
Advanced Manufacturing of Remdesivir Intermediates via Optimized Synthetic Routes
The global demand for effective antiviral therapeutics has placed immense pressure on the pharmaceutical supply chain to deliver high-purity active pharmaceutical ingredients (APIs) with speed and reliability. Patent CN112321589A introduces a groundbreaking synthesis method for Remdesivir (GS-5734), a cyano-substituted adenosine nucleotide analogue with broad-spectrum antiviral activity. This intellectual property outlines a novel pathway that directly utilizes 4-chloropyrrolo[2,1-f][1,2,4]triazine as a starting material, bypassing the need for excessive active hydrogen protection. The technical breakthrough lies in its ability to achieve high reaction yields through a streamlined sequence of addition, cyanation, ammonolysis, and coupling reactions. For R&D directors and procurement specialists, this represents a significant shift towards more efficient manufacturing paradigms, offering a robust alternative to legacy processes that suffer from low efficiency and complex purification requirements.
The Limitations of Conventional Methods vs. The Novel Approach
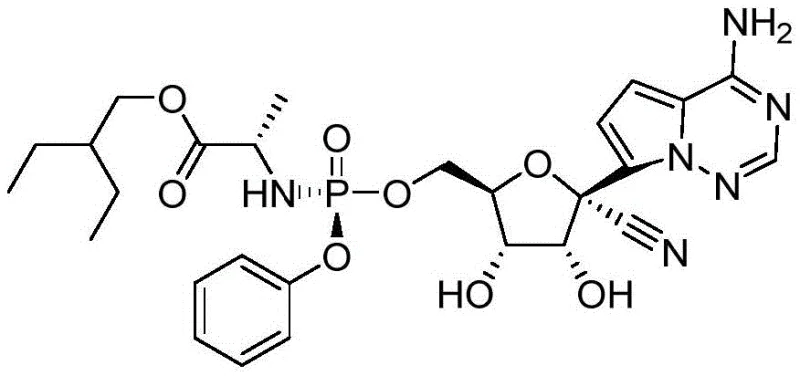
The Limitations of Conventional Methods
Historically, the first-generation synthesis of Remdesivir relied on a convoluted sequence involving 4-chloropyrrolo[2,1-f][1,2,4]triazine reacting with ammonia and bromine, followed by protection with trimethylchlorosilane. This traditional route, often depicted in early literature, necessitates the use of butyllithium for bromine removal and subsequent condensation with tribenzyloxy-D-ribonic acid-1,4-lactone. A critical bottleneck in this legacy approach is the cyanation step using boron trifluoride ether, which frequently results in poor chirality selection and low yields. Furthermore, the debenzylation step typically employs boron trichloride, a hazardous reagent that complicates post-treatment and generates significant hazardous waste. The cumulative effect of these inefficiencies is an overall yield that is too low to be economically viable for large-scale process scale-up, creating supply chain vulnerabilities for critical antiviral medications.
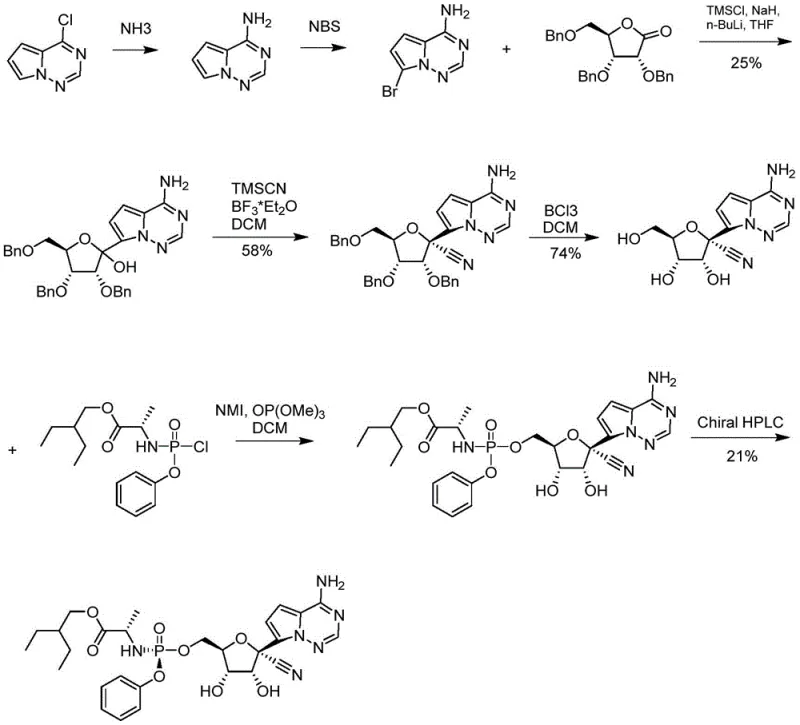
The Novel Approach
In stark contrast, the methodology disclosed in CN112321589A revolutionizes the production landscape by eliminating the need for amino protection during the initial condensation phases. By directly reacting the chloro-triazine derivative with the ribono-lactone under basic conditions, the process avoids the material loss associated with TMS protection and Grignard reagent consumption seen in improved prior art. The innovation extends to the debenzylation stage, where palladium-catalyzed hydrogenation replaces the harsh boron trichloride treatment, ensuring a clean and efficient reaction with convenient catalyst recovery. Additionally, the optimization of the final coupling reaction through the modification of the leaving group in compound 7 allows for an unprotected docking reaction that drastically improves selectivity. This holistic redesign of the synthetic tree results in a route that is not only simpler to operate but also delivers superior product purity essential for regulatory compliance.
Mechanistic Insights into Copper-Catalyzed Ammonolysis and Coupling
The core of this synthetic advancement lies in the precise control of reaction mechanisms to minimize byproduct formation. The ammonolysis step, converting the chloro-intermediate to the amino-derivative, utilizes a copper catalyst system, potentially involving cuprous iodide or cuprous bromide. This transition metal catalysis facilitates the nucleophilic substitution under milder conditions compared to traditional high-pressure ammonia reactions, thereby preserving the integrity of the sensitive nitrile and sugar moieties. Following this, the debenzylation is achieved via heterogeneous catalysis using palladium on carbon. This mechanism allows for the selective cleavage of benzyl ethers in the presence of the nitrile group, a chemoselectivity challenge that often plagues nucleoside synthesis. The ability to recover the palladium catalyst adds a layer of economic and environmental sustainability to the process, aligning with green chemistry principles increasingly demanded by global regulatory bodies.
Furthermore, the final phosphorylation step demonstrates sophisticated mechanistic engineering. By employing a modified phosphoramidate precursor (compound 7) with an enhanced leaving group, the reaction kinetics are significantly accelerated. The addition of titanium tetrachloride or tetraisopropyl titanate as an auxiliary agent plays a crucial role in activating the hydroxyl group of the nucleoside intermediate without the need for prior protection. This "unprotected docking" strategy reduces the step count and eliminates the yield losses associated with protection-deprotection cycles. The result is a highly convergent synthesis where the stereochemical integrity of the chiral centers is maintained throughout, ensuring the final API meets the stringent enantiomeric excess requirements necessary for antiviral efficacy and safety profiles.
How to Synthesize Remdesivir Efficiently
The patented process offers a clear, five-step roadmap for the efficient production of Remdesivir, starting from readily available raw materials. The sequence begins with a low-temperature addition reaction to form the C-N glycosidic bond, followed by a Lewis acid-mediated cyanation to install the critical nitrile functionality. Subsequent steps involve a copper-catalyzed amination and a palladium-catalyzed hydrogenation to reveal the active nucleoside core. The synthesis concludes with a highly optimized coupling reaction to attach the prodrug moiety. This streamlined approach minimizes unit operations and maximizes throughput, making it an ideal candidate for technology transfer and commercial manufacturing. Detailed standardized synthesis steps are provided in the guide below for technical reference.
- Perform addition condensation between 4-chloropyrrolo[2,1-f][1,2,4]triazine and tribenzyloxy-D-ribono-1,4-lactone using a strong base like LDA at low temperature.
- Execute cyanation of the resulting intermediate using trimethylsilyl cyanide under Lewis acid catalysis to form the nitrile group.
- Conduct copper-catalyzed ammonolysis to substitute the chloro group with an amino group, followed by palladium-catalyzed hydrogenation for debenzylation.
- Finalize the synthesis by coupling the deprotected nucleoside with the phosphoramidate prodrug moiety using a magnesium base and titanium additive.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere technical elegance. The elimination of multiple protection and deprotection steps fundamentally alters the cost structure of manufacturing by reducing raw material consumption and solvent usage. The removal of hazardous reagents like boron trichloride simplifies waste management protocols and lowers the environmental compliance burden, which is a critical factor in maintaining uninterrupted production schedules. Moreover, the high purity achieved through crystallization rather than complex chromatography ensures a consistent supply of quality material, reducing the risk of batch failures and delivery delays.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive silyl protecting groups and the associated reagents required for their installation and removal. By utilizing a direct addition strategy that avoids amino interference, the yield of the cyanation reaction is substantially increased, meaning less starting material is wasted per kilogram of final product. The switch to palladium-catalyzed hydrogenation allows for the recovery and reuse of the precious metal catalyst, further driving down the variable costs of production. These cumulative efficiencies result in a leaner manufacturing process that offers substantial cost savings without compromising on the quality of the antiviral intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the new route. The reliance on standard, commercially available reagents such as LDA, trimethylsilyl cyanide, and copper salts reduces dependency on specialized or hard-to-source chemicals. The operational simplicity, characterized by fewer reaction steps and easier work-up procedures like filtration and pulping, shortens the overall production cycle time. This agility enables manufacturers to respond more rapidly to fluctuations in market demand, ensuring a steady flow of high-purity intermediates to downstream API producers and mitigating the risk of shortages during public health emergencies.
- Scalability and Environmental Compliance: The route is explicitly designed with commercial scale-up in mind, addressing the limitations of previous methods that struggled beyond the laboratory bench. The replacement of hazardous debenzylating agents with catalytic hydrogenation significantly reduces the generation of toxic waste streams, aligning the process with stricter environmental regulations. The high selectivity of the final coupling reaction minimizes the formation of difficult-to-remove impurities, simplifying the purification train and reducing solvent intensity. This environmentally conscious design not only facilitates smoother regulatory approvals but also ensures long-term sustainability for large-volume manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: How does this new synthesis route improve upon previous methods regarding yield and waste?
A: The patented method eliminates unnecessary protection and deprotection steps, such as TMS protection and acetonide formation, which were required in prior art. By avoiding harsh reagents like boron trichloride for debenzylation and utilizing recyclable palladium on carbon, the process significantly reduces three wastes and simplifies post-treatment, leading to higher overall yields and purity suitable for industrial scale-up.
Q: What specific catalytic systems are utilized to ensure high stereoselectivity?
A: The process employs specific Lewis acids like boron trifluoride acetic acid or tin tetrachloride during the cyanation step to control stereochemistry effectively. Furthermore, the final coupling reaction utilizes titanium tetrachloride or tetraisopropyl titanate as an auxiliary agent alongside a Grignard base, which optimizes the unprotected docking reaction and greatly improves selectivity without needing additional protecting groups on the sugar moiety.
Q: Is this manufacturing process scalable for commercial production of antiviral APIs?
A: Yes, the route is explicitly designed for scalability. It avoids complex chromatographic separations by relying on crystallization and pulping for purification. The use of robust catalysts like palladium carbon, which can be easily recovered and reused, along with the elimination of sensitive protection steps, makes the process operationally simple and highly suitable for large-scale commercial manufacturing of Remdesivir intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality antiviral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement complex synthetic routes, such as the optimized Remdesivir process described in CN112321589A, positions us as a strategic partner for companies seeking to diversify their supply chain and reduce dependency on single-source vendors.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this optimized route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and reliable supply model.
