Advanced Synthesis of Remdesivir Intermediates via Lewis Acid Catalysis for Commercial Scale-Up
Advanced Synthesis of Remdesivir Intermediates via Lewis Acid Catalysis for Commercial Scale-Up
The global demand for effective antiviral therapeutics has placed immense pressure on the supply chains of critical pharmaceutical intermediates, particularly for nucleoside analogues like Remdesivir. Patent CN111233931B, published in March 2022, introduces a transformative synthetic methodology that addresses the longstanding bottlenecks in producing this high-value active pharmaceutical ingredient (API). This technical disclosure outlines a robust three-step sequence that bypasses the hazardous reagents and complex purification hurdles associated with first-generation processes. By leveraging a novel Lewis acid-catalyzed coupling strategy followed by an efficient chiral resolution, this method offers a pathway to high-purity intermediates with significantly improved process safety and operational simplicity. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is crucial for securing a reliable supply of antiviral intermediates in a volatile market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
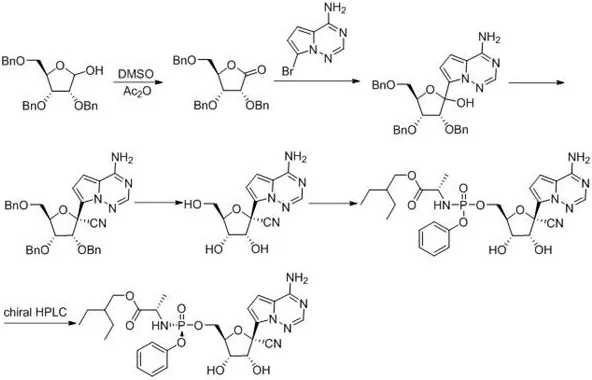
Historically, the synthesis of Remdesivir has been plagued by inefficient multistep sequences that rely heavily on aggressive protecting group chemistry. As illustrated in the prior art routes, such as the one disclosed in WO2016069826A1, the conventional approach typically initiates with heavily benzylated sugar derivatives. These routes necessitate multiple oxidation, addition, and substitution steps, culminating in a final resolution that often relies on preparative chiral HPLC. The reliance on chiral columns is a major bottleneck for commercial manufacturing, as it severely limits throughput and drives up costs due to the expensive stationary phases and low loading capacities. Furthermore, alternative routes reported in literature, such as those utilizing triflic acid to enhance beta-anomer selectivity, introduce significant safety liabilities by employing genotoxic nitro-substituted reagents. These hazardous materials complicate waste management and require stringent containment protocols, thereby increasing the overall cost of goods sold (COGS) and regulatory burden for manufacturers.
The Novel Approach
In stark contrast to the convoluted pathways of the past, the methodology described in CN111233931B presents a streamlined and chemically elegant solution. The core innovation lies in the convergent assembly of the phosphoramidate moiety prior to the coupling with the nucleoside core. Specifically, the process involves the reaction of a chlorophosphoramidate precursor (Compound V) with a hydroxypyridine derivative under basic conditions to generate an activated intermediate (Compound IV). This activated species is then coupled with the protected nucleoside (Compound III) in the presence of a Lewis acid catalyst. This strategic shift eliminates the need for harsh acidic promoters like triflic acid and avoids the generation of genotoxic impurities. The final step utilizes a pragmatic physical separation technique—slurrying and crystallization in an acetone/dichloromethane system—to achieve high optical purity, completely bypassing the need for chromatographic resolution. This approach not only simplifies the unit operations but also drastically reduces the environmental footprint of the synthesis.
Mechanistic Insights into Lewis Acid-Catalyzed Phosphoramidation
The success of this novel synthetic route hinges on the precise activation of the phosphorus center during the coupling of Compound IV and Compound III. In traditional nucleophilic substitutions at phosphorus, achieving high stereoselectivity and conversion rates often requires forcing conditions that can degrade sensitive nucleoside scaffolds. The introduction of Lewis acids, such as zinc chloride (ZnCl2), magnesium chloride (MgCl2), or zinc bromide (ZnBr2), serves to coordinate with the oxygen atoms of the phosphoryl group, thereby increasing the electrophilicity of the phosphorus atom. This coordination lowers the activation energy for the nucleophilic attack by the 5'-hydroxyl group of the nucleoside. Experimental data from the patent indicates that conducting this reaction at moderate temperatures between 50°C and 80°C in solvents like tetrahydrofuran or dioxane provides an optimal balance between reaction kinetics and substrate stability. The choice of base, ranging from triethylamine to inorganic carbonates, further fine-tunes the reaction environment by scavenging the generated acid without promoting side reactions such as epimerization at the anomeric center.
Impurity control is another critical aspect where this mechanism excels. In the absence of Lewis acid activation, competing reactions such as the hydrolysis of the phosphoramidate or the formation of pyrophosphate byproducts can occur. The Lewis acid ensures a rapid and directed coupling, minimizing the residence time of reactive intermediates and thus suppressing the formation of these difficult-to-remove impurities. Furthermore, the subsequent resolution step exploits the subtle solubility differences between the diastereomers of Compound II. By carefully selecting the acetone/dichloromethane ratio, the process induces preferential crystallization of the desired enantiomer (Compound I). This thermodynamic control over the solid-state properties allows for the rejection of the unwanted isomer into the mother liquor, ensuring that the final product meets the stringent purity specifications required for clinical applications without the need for iterative chromatographic polishing.
How to Synthesize Remdesivir Intermediate Efficiently
The implementation of this synthesis route requires careful attention to temperature control and stoichiometry to maximize yield and optical purity. The process begins with the low-temperature formation of the activated phosphoramidate, followed by the Lewis acid-mediated coupling, and concludes with the crystallization-based resolution. Detailed standard operating procedures regarding reagent grades, addition rates, and workup protocols are essential for reproducible results. The following guide summarizes the critical operational parameters derived from the patent examples to assist process chemists in scaling this technology.
- React Compound V with hydroxypyridines under basic conditions at low temperatures (-10°C to 20°C) to generate the activated phosphoramidate Compound IV.
- Couple Compound IV with the nucleoside Compound III in the presence of a Lewis acid (e.g., ZnCl2) and base at elevated temperatures (50-80°C) to form the diastereomeric mixture Compound II.
- Perform chiral resolution of Compound II via slurrying and crystallization using an acetone/dichloromethane solvent system to isolate the target enantiomer Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis method described in CN111233931B offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of genotoxic reagents and the removal of chiral HPLC purification steps fundamentally alter the cost structure and risk profile of manufacturing Remdesivir intermediates. By simplifying the process flow, manufacturers can reduce the number of unit operations, which directly correlates to lower capital expenditure (CAPEX) and operational expenditure (OPEX). Moreover, the use of common, non-proprietary solvents and catalysts enhances supply chain resilience, reducing dependency on single-source specialty chemicals that are prone to allocation issues during global health crises.
- Cost Reduction in Manufacturing: The most significant economic driver of this new method is the replacement of chromatographic purification with crystallization. Chiral HPLC is notoriously expensive due to the high cost of chiral stationary phases and the low throughput associated with batch chromatography. By achieving high optical purity through slurrying and crystallization, the process eliminates this cost center entirely. Additionally, the avoidance of genotoxic nitro-substituted reagents removes the need for specialized waste treatment facilities and extensive analytical testing for mutagenic impurities, further driving down the total cost of production. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to overall operational efficiency.
- Enhanced Supply Chain Reliability: The robustness of the Lewis acid-catalyzed coupling step ensures consistent batch-to-batch quality, which is vital for maintaining a steady supply of API. Traditional routes that rely on sensitive reagents or complex protecting group manipulations are prone to variability, leading to batch failures and supply disruptions. This new method utilizes stable, commercially available starting materials and catalysts, mitigating the risk of raw material shortages. The simplified workflow also shortens the manufacturing cycle time, allowing producers to respond more agilely to fluctuations in market demand. This reliability is crucial for pharmaceutical companies aiming to secure long-term contracts for antiviral drug production.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a significant advancement. The removal of hazardous nitro compounds and the reduction of solvent usage associated with chromatography align with the principles of green chemistry. The process generates less hazardous waste, simplifying disposal and reducing the environmental compliance burden. Furthermore, the scalability of crystallization processes is well-understood in the chemical industry, allowing for seamless transition from pilot plant to multi-ton commercial production. This ease of scale-up ensures that manufacturers can rapidly ramp up capacity to meet surging demand without compromising on product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Remdesivir intermediates using the patented Lewis acid methodology. These insights are derived directly from the experimental data and claims presented in CN111233931B, providing clarity on the practical implementation and advantages of this technology for industry stakeholders.
Q: How does the Lewis acid catalysis improve the synthesis of Remdesivir compared to traditional methods?
A: The use of Lewis acids like zinc chloride or magnesium bromide activates the phosphoramidate coupling step, allowing for milder reaction conditions and avoiding the use of genotoxic nitro-substituted reagents found in earlier literature routes. This results in a cleaner impurity profile and safer handling protocols.
Q: What is the advantage of the acetone/dichloromethane resolution method?
A: This specific solvent system enables effective chiral separation through slurrying and crystallization without the need for expensive and low-throughput chiral HPLC columns, significantly enhancing the scalability and cost-efficiency of the purification process.
Q: Is this synthesis route suitable for industrial scale production?
A: Yes, the patent explicitly states the process features mild reaction conditions, easily controllable parameters, and simple operations, making it highly suitable for large-scale industrial manufacturing compared to routes requiring complex protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team of process chemists has extensively analyzed the synthetic pathways disclosed in CN111233931B and is fully equipped to implement this advanced Lewis acid-catalyzed route. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that employ advanced analytical techniques to guarantee the quality of every batch.
We invite you to collaborate with us to optimize your supply chain for Remdesivir and related antiviral intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
