Optimizing Drospirenone Production: A Technical Analysis of Patent CN102395596B for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value hormonal active pharmaceutical ingredients (APIs), and Drospirenone (CAS#: 67392-87-4) stands as a prime example of a fourth-generation progestogen with unique anti-mineralocorticoid properties. Patent CN102395596B discloses a novel preparation method that addresses critical stability and safety issues inherent in earlier synthetic routes. By utilizing 3β,5-dihydroxy-6β,7β,15β,16β-dimethylene-5β-androst-17,20-epoxy as the starting material, this invention establishes a streamlined sequence involving selective oxidation, ketal protection, lactone construction, and a sophisticated sulfonylation-elimination finale. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, understanding the mechanistic advantages of this specific patent is crucial for securing a supply chain that prioritizes both structural fidelity and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Drospirenone, such as those disclosed in European patent EP0075189 and Chinese patent CN101092443, have long been plagued by significant process hazards and chemical instability issues. The earlier methodologies often rely on the use of flammable and explosive catalysts alongside hydrogen gas, creating a volatile environment that poses severe safety risks during large-scale manufacturing operations. More critically from a chemical integrity standpoint, the dehydration steps in these conventional processes are typically conducted under strong alkaline conditions. This harsh basic environment frequently induces unwanted rearrangement reactions, specifically causing the opening of the sensitive spiro-lactone ring and the cyclopropane ring system. Such side reactions not only drastically reduce the overall yield but also generate complex impurity profiles that are difficult to purge, thereby compromising the quality of the final high-purity pharmaceutical intermediate.
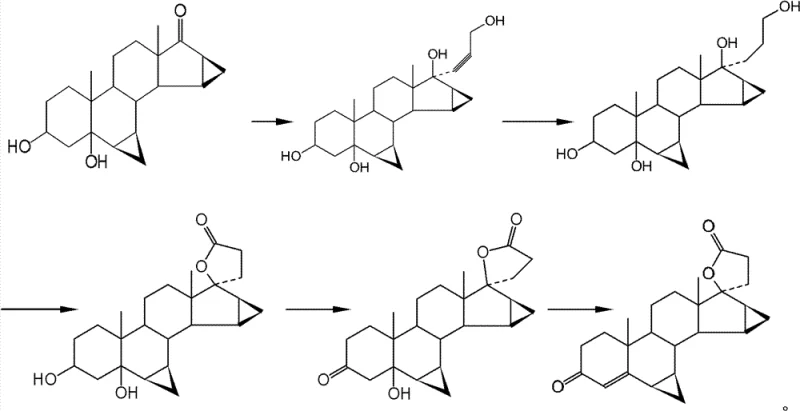
The Novel Approach
In stark contrast to the hazardous and unstable legacy methods, the process defined in CN102395596B introduces a chemically elegant solution that circumvents these pitfalls through a carefully orchestrated sequence of protection and elimination strategies. The innovation lies in the replacement of direct alkaline dehydration with a sulfonylation-desulfonation protocol carried out in a mild reaction system composed of glacial acetic acid and sodium acetate. This shift from strong base to a buffered acidic medium effectively suppresses the ring-opening rearrangement of the spiro and cyclopropane moieties, ensuring the structural integrity of the steroid backbone is maintained throughout the synthesis. By enhancing the specificity of the elimination reaction, this novel approach significantly minimizes by-product formation, leading to a more robust and reproducible manufacturing process that is ideally suited for the commercial scale-up of complex hormonal intermediates.
Mechanistic Insights into Sulfonylation-Mediated Elimination
The core chemical breakthrough of this patent resides in the final transformation steps where the 5-position hydroxyl group is activated and subsequently eliminated to form the requisite 3-keto-4-ene system. Initially, the 3-position hydroxyl is oxidized using agents like chromic anhydride or N-bromosuccinimide, followed by ketal protection with ethylene glycol to shield the ketone functionality during subsequent transformations. The construction of the carboxylic inner ester involves a condensation with dimethyl malonate and subsequent de-esterification using halogen alkali metal salts in DMF at elevated temperatures (100°C–180°C). The pivotal moment occurs when the 5-hydroxyl group is converted into a sulfonate ester (using benzenesulfonyl chloride, p-toluenesulfonyl chloride, or methanesulfonyl chloride). This activation turns a poor leaving group into an excellent one, facilitating elimination under the mild acidic conditions of the acetic acid/sodium acetate buffer.
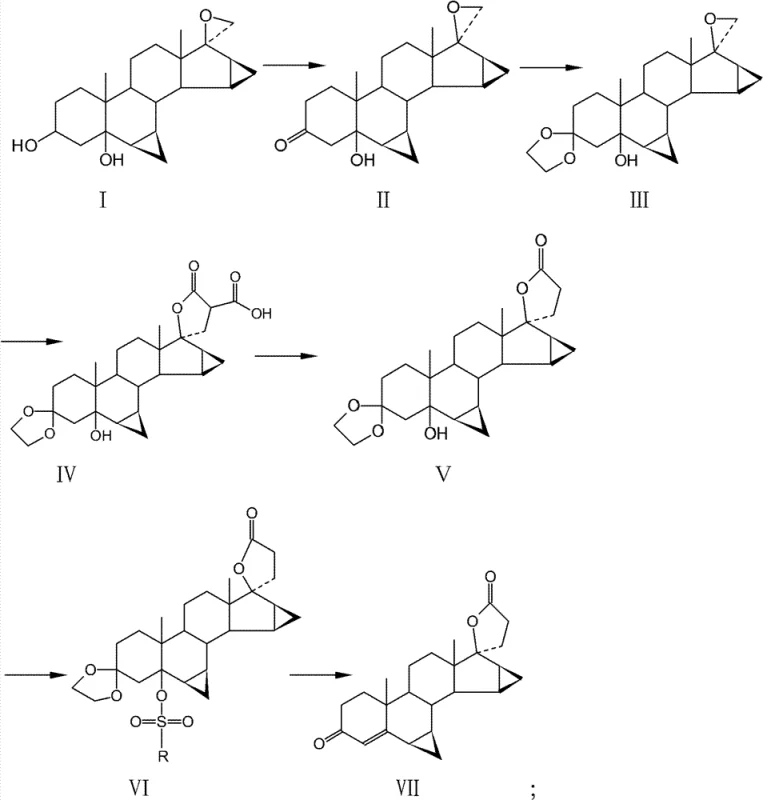
This mechanistic design offers profound advantages for impurity control, a key concern for any R&D Director overseeing API production. By avoiding the nucleophilic attack of hydroxide ions on the electrophilic centers of the spiro-lactone, the process prevents the formation of ring-opened acids or rearranged isomers that typically contaminate batches produced via alkaline routes. The use of specific sulfonyl chlorides allows for fine-tuning the reactivity of the intermediate, ensuring that the elimination proceeds cleanly to form the double bond at the 4,5-position without affecting the delicate cyclopropane rings at positions 6,7 and 15,16. This high degree of regioselectivity and chemoselectivity translates directly into a cleaner crude product, reducing the burden on downstream purification units and ensuring that the final high-purity Drospirenone meets stringent pharmacopeial standards with minimal effort.
How to Synthesize Drospirenone Efficiently
The synthesis of Drospirenone outlined in this patent represents a highly集约 (intensive) and specific route designed to overcome the low yields and quality instability of existing products. The process flows logically from oxidation to protection, then to lactone ring construction, and finally to the critical elimination step. Each stage is optimized for temperature and solvent compatibility, ranging from 0°C for sensitive additions to reflux conditions for cyclization. For technical teams looking to implement this methodology, the detailed standardized synthesis steps provided in the patent documentation serve as a comprehensive guide for laboratory validation and pilot plant trials.
- Oxidize the 3-position hydroxyl group of the starting material using chromic anhydride or N-bromosuccinimide to form the ketone intermediate.
- Protect the 3-ketone group as a ketal using ethylene glycol and an acidic catalyst to enhance hydrophobicity and yield.
- Construct the carboxylic inner ester (lactone) via condensation with dimethyl malonate followed by de-esterification and halogen salt treatment.
- Sulfonylate the 5-position hydroxyl group and perform simultaneous deprotection and elimination in a glacial acetic acid/sodium acetate system to yield Drospirenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance, directly impacting cost structures and supply continuity. The elimination of hazardous reagents like hydrogen gas and explosive catalysts removes the need for specialized high-pressure equipment and rigorous safety protocols associated with hydrogenation units, leading to substantial capital expenditure savings and reduced operational overhead. Furthermore, the avoidance of strong alkaline conditions mitigates the risk of batch failures due to rearrangement side reactions, thereby enhancing the predictability of production schedules and ensuring a more reliable flow of materials to downstream formulation units.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of the purification train. By significantly reducing the generation of ring-opened impurities and rearrangement by-products, the need for extensive chromatographic separation or multiple recrystallization steps is drastically diminished. Additionally, the reagents employed, such as acetic acid, sodium acetate, and common sulfonyl chlorides, are commodity chemicals with stable pricing and widespread availability, avoiding the supply bottlenecks often associated with exotic or proprietary catalysts. This combination of higher crude purity and accessible raw materials drives down the overall cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway directly correlates to improved lead times and supply security. Because the reaction conditions are less sensitive to minor fluctuations in pH or temperature compared to the precarious alkaline dehydration of older methods, the process demonstrates greater reproducibility across different batches and manufacturing sites. This consistency reduces the likelihood of out-of-specification (OOS) results that can halt production lines. Moreover, the starting materials and intermediates are stable and can be stocked strategically, allowing manufacturers to respond rapidly to market demand surges without the long lead times typically required for sourcing specialized catalytic systems.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns well with modern green chemistry principles by minimizing the use of heavy metals and hazardous gases. The waste streams generated are predominantly organic salts and solvents that are easier to treat and dispose of compared to the complex mixtures resulting from rearrangement reactions. This facilitates smoother regulatory approvals and environmental audits, which are critical for maintaining uninterrupted operations in strictly regulated pharmaceutical markets. The process is explicitly designed for industrial-scale production, ensuring that the transition from kilogram-scale R&D to tonnage-scale commercial manufacturing can be executed with minimal technical friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Drospirenone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN102395596B, providing a clear picture of its operational viability.
Q: How does this synthesis method improve safety compared to conventional routes?
A: Unlike prior art methods such as EP0075189 which utilize flammable hydrogen gas and explosive catalysts, this process employs stable oxidants like chromic anhydride or N-bromosuccinimide. Furthermore, the elimination step avoids strong alkaline conditions, significantly reducing operational hazards.
Q: What is the primary advantage regarding product purity and structural integrity?
A: The use of a sulfonylation-desulfonation elimination strategy in a weakly acidic acetic acid/sodium acetate system prevents the ring-opening rearrangement of the sensitive spiro-lactone and cyclopropane rings, which is a common side reaction in alkaline dehydration processes.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the route is designed for industrial production. The steps utilize common solvents like pyridine, DMF, and acetic acid, and the reaction conditions (0°C to 180°C) are manageable within standard pharmaceutical reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drospirenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex steroids like Drospirenone requires more than just a patent license; it demands deep process engineering expertise and a commitment to quality excellence. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel sulfonylation-elimination route are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of high-purity Drospirenone delivered meets the exacting standards required by global regulatory bodies, providing our partners with the confidence needed to navigate the competitive hormonal therapy market.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthetic technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical pharmaceutical intermediate, driving value and efficiency into your final drug products.
