Advanced Drospirenone Manufacturing: Overcoming Safety and Stability Challenges in API Production
The pharmaceutical industry continuously seeks robust manufacturing routes for high-value hormonal APIs like Drospirenone (CAS#: 67392-87-4), particularly those that mitigate safety risks and improve structural fidelity. Patent CN102395596B introduces a transformative preparation method that fundamentally alters the final elimination strategy, moving away from hazardous hydrogenation and harsh alkaline conditions. This innovation addresses critical pain points in steroid synthesis, specifically the instability of the spiro-lactone and cyclopropane moieties under traditional dehydration conditions. By employing a sulfonylation-desulfonation sequence within a glacial acetic acid and sodium acetate buffer system, the process achieves high reaction specificity while eliminating the need for explosive hydrogen gas. For global procurement teams and R&D directors, this patent represents a significant leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a pathway that is not only safer but also inherently more scalable due to its reliance on standard atmospheric pressure reactions and common reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Drospirenone has been plagued by significant safety and chemical stability challenges, as evidenced by earlier patents such as EP0075189. Traditional routes often necessitate the use of flammable and explosive catalysts alongside high-pressure hydrogen gas, creating substantial operational hazards that require specialized, costly infrastructure to manage safely. Furthermore, the critical dehydration step in these legacy processes is typically conducted under strong alkaline conditions. This harsh chemical environment poses a severe threat to the molecular integrity of the steroid backbone, frequently inducing unwanted ring-opening rearrangements of the sensitive spiro-lactone and cyclopropane rings. These side reactions not only degrade the overall yield but also generate complex impurity profiles that are difficult and expensive to remove during downstream purification, ultimately compromising the economic viability and supply reliability of the final API.
The Novel Approach
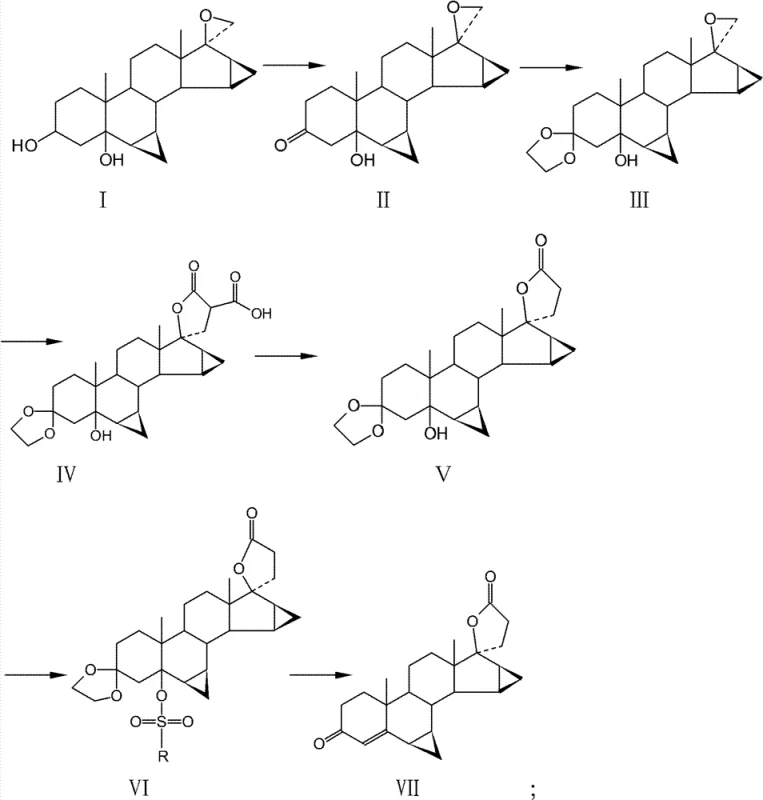
In stark contrast, the methodology disclosed in CN102395596B offers a sophisticated solution by replacing the hazardous elimination step with a mild, highly specific sulfonylation strategy. The process initiates with the oxidation of the 3-hydroxyl group followed by ketal protection, setting the stage for the construction of the carboxylic inner ester via condensation and de-esterification. The true innovation lies in the final transformation: rather than forcing dehydration with strong bases, the 5-hydroxyl group is first converted into a sulfonate ester (using reagents like tosyl chloride or mesyl chloride). This activated intermediate is then subjected to a reaction system comprising glacial acetic acid and sodium acetate. This buffered acidic environment facilitates the simultaneous removal of the ketal protecting group and the elimination of the sulfonate to form the desired 3-keto-4-ene structure. This approach effectively bypasses the rearrangement pitfalls of alkaline methods, ensuring a cleaner reaction profile and superior product quality.
Mechanistic Insights into Sulfonylation-Mediated Elimination
The core mechanistic advantage of this patent lies in the activation of the leaving group at the C-5 position through sulfonylation. In traditional E2 elimination mechanisms driven by strong bases, the abstraction of a proton can sometimes compete with nucleophilic attacks on the strained cyclopropane or lactone rings, leading to ring opening. By converting the hydroxyl group into a sulfonate (such as a tosylate or mesylate), the leaving group ability is dramatically enhanced, allowing elimination to proceed under much milder conditions. The use of sodium acetate in glacial acetic acid provides a weakly basic environment that is sufficient to drive the elimination of the sulfonate but too mild to attack the electrophilic centers of the spiro-lactone. This delicate balance preserves the stereochemical integrity of the 6β,7β and 15β,16β-dimethylene groups, which are crucial for the biological activity of Drospirenone.
Furthermore, the integration of the deprotection step within the same reaction vessel as the elimination demonstrates remarkable process intensification. The acidic nature of the glacial acetic acid medium simultaneously hydrolyzes the ethylene ketal at the C-3 position while the acetate ion promotes the elimination at C-5. This tandem operation reduces the number of unit operations required, minimizing solvent usage and processing time. From an impurity control perspective, this mechanism significantly suppresses the formation of rearranged by-products that typically arise from base-catalyzed degradation. The result is a crude product with a markedly improved purity profile, reducing the burden on crystallization and chromatography steps, which directly translates to lower manufacturing costs and higher throughput for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Drospirenone Efficiently
The synthesis of Drospirenone via this patented route involves a logical sequence of functional group transformations designed to maximize yield and safety. Starting from the epoxy-androstane precursor, the process systematically builds the necessary functionality while protecting sensitive sites until the final reveal. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures for each of the six stages—from oxidation to the final acetic acid reflux—are outlined in the technical guide below.
- Oxidize the starting 3β,5-dihydroxy steroid using chromic anhydride or NBS to form the 3-keto intermediate.
- Protect the 3-ketone as a ketal using ethylene glycol, then perform condensation with dimethyl malonate followed by lactonization.
- Sulfonylate the 5-hydroxyl group and perform simultaneous deprotection and elimination in a glacial acetic acid/sodium acetate system to yield Drospirenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The elimination of high-pressure hydrogenation removes a major bottleneck in production scheduling and safety compliance, allowing for more flexible manufacturing campaigns. Additionally, the avoidance of strong alkaline conditions reduces the corrosion load on reactor vessels and minimizes the generation of hazardous waste streams associated with neutralizing strong bases. These factors collectively contribute to a more resilient and cost-efficient supply chain, ensuring consistent availability of high-purity Drospirenone for downstream formulation.
- Cost Reduction in Manufacturing: The removal of high-pressure hydrogenation equipment drastically lowers capital expenditure requirements, as facilities no longer need specialized autoclaves or extensive gas handling safety systems. Furthermore, the suppression of rearrangement by-products means that less raw material is wasted on off-spec impurities, leading to substantial savings in starting material costs. The streamlined tandem deprotection-elimination step also reduces solvent consumption and energy usage by combining two operations into one, driving down the overall cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: By relying on common, non-hazardous reagents like acetic acid and sodium acetate instead of explosive hydrogen gas, the risk of production shutdowns due to safety incidents or regulatory inspections is significantly mitigated. The robustness of the chemical route against rearrangement ensures batch-to-batch consistency, which is critical for maintaining long-term supply contracts with major pharmaceutical partners. This stability allows for more accurate forecasting and inventory management, reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted delivery to clients.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, as it avoids the generation of heavy metal waste from hydrogenation catalysts and reduces the volume of caustic waste from alkaline treatments. This alignment with green chemistry principles simplifies environmental permitting and waste disposal logistics, facilitating easier scale-up from pilot plants to multi-ton commercial production. The mild reaction conditions also extend the lifespan of manufacturing equipment, reducing maintenance downtime and supporting continuous, large-scale operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential manufacturing partners.
Q: Why is the acetic acid/sodium acetate system preferred for the final elimination step?
A: Traditional alkaline dehydration conditions pose a high risk of opening the sensitive spiro-lactone and cyclopropane rings, leading to rearrangement by-products. The mild acidic buffer system in this patent ensures specific elimination without compromising the structural integrity of the steroid core.
Q: Does this process require high-pressure hydrogenation equipment?
A: No. Unlike prior art methods (e.g., EP0075189) that utilize flammable hydrogen gas and catalysts, this novel route relies on chemical oxidation and sulfonylation, significantly enhancing plant safety and reducing infrastructure requirements.
Q: What are the critical purity controls in this synthesis?
A: The key control point is the prevention of ring-opening rearrangements during the dehydration phase. By utilizing the sulfonylation-desulfonation strategy, the process minimizes impurities related to spiro-ring cleavage, resulting in a cleaner crude product profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drospirenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel sulfonylation route are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature profiles required for this synthesis, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee that every batch meets the highest international standards for hormonal APIs.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the safest, most efficient, and commercially viable production method available for Drospirenone.
