Advanced Industrial Synthesis of Tiagabine: Overcoming Organometallic Limitations for Scalable API Production
Advanced Industrial Synthesis of Tiagabine: Overcoming Organometallic Limitations for Scalable API Production
The pharmaceutical landscape for neurological treatments has long relied on efficient access to high-purity GABA uptake inhibitors, with Tiagabine standing as a cornerstone molecule in epilepsy management. Patent CN1314684C introduces a transformative synthesis methodology that fundamentally restructures the production pathway for Tiagabine, its racemate, and its S-configuration enantiomer. This intellectual property addresses critical bottlenecks in the existing manufacturing paradigm by replacing hazardous, cryogenic organometallic steps with robust, mild-condition organic transformations. For global procurement leaders and R&D directors, this innovation represents a pivotal shift towards more sustainable and economically viable pharmaceutical intermediates sourcing. The disclosed route not only streamlines the construction of the complex bis-thiophene backbone but also integrates a highly efficient resolution strategy for the chiral piperidine moiety, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards for optical purity without excessive downstream processing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically the seminal work reported by Knudsen et al. in 1993, established the foundational chemistry for Tiagabine but relied heavily on operationally demanding techniques that pose significant challenges for modern industrial scale-up. As illustrated in the historical context of synthetic development, the conventional pathways (often referred to as Method A and Method B in early literature) necessitate the use of highly reactive organometallic species such as cyclopropylmagnesium bromide or lithiated thiophenes. These reagents are notoriously pyrophoric and extremely sensitive to moisture and oxygen, requiring rigorous exclusion of atmospheric conditions that drive up facility maintenance and operational overheads. Furthermore, these reactions typically demand cryogenic temperatures, often as low as -70°C, to control selectivity and prevent decomposition, imposing a heavy energy burden on the manufacturing process. The reliance on such specialized conditions inherently limits the batch size and throughput, creating a fragile supply chain vulnerable to disruptions and quality deviations.
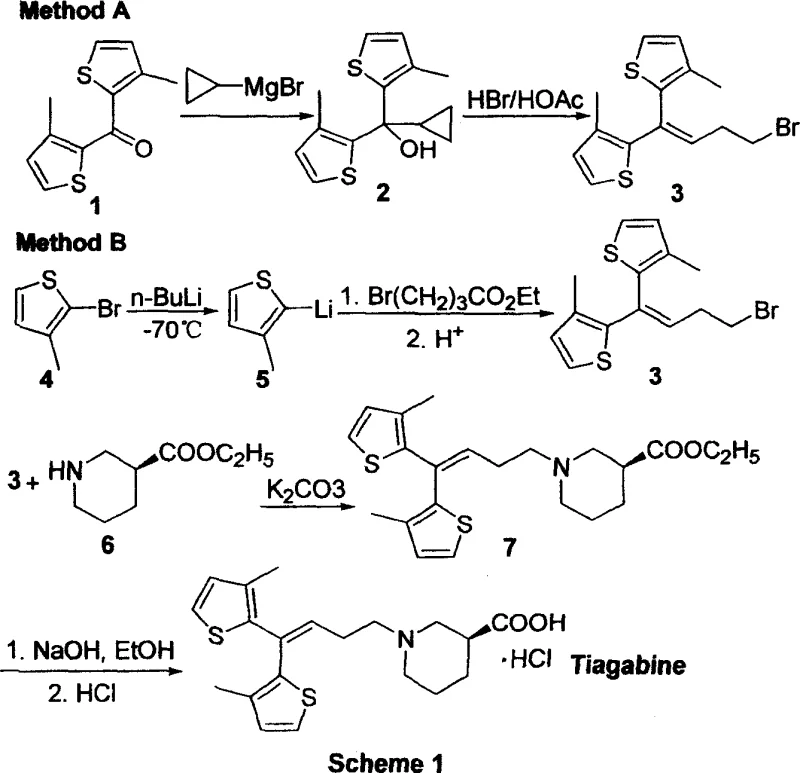
The Novel Approach
In stark contrast, the methodology detailed in CN1314684C circumvents these hazards by employing a Wittig or Horner-Wadsworth-Emmons olefination strategy to construct the critical carbon-carbon double bond linking the bis-thiophene core to the side chain. This approach utilizes phosphonium salts or phosphonates, which are stable, shelf-stable solids that can be handled in standard atmospheric conditions without the need for inert gas gloveboxes or specialized cryogenic reactors. The reaction proceeds efficiently under mild thermal conditions, typically ranging from ambient temperature to moderate reflux (0-110°C), drastically reducing energy consumption and equipment complexity. By shifting from metal-halogen exchange mechanisms to nucleophilic substitution and olefination, the new route minimizes the formation of metallic waste streams, aligning perfectly with green chemistry principles and reducing the environmental footprint associated with cost reduction in pharmaceutical intermediates manufacturing. This strategic pivot allows for a seamless transition from laboratory benchtop to multi-ton commercial production.
Mechanistic Insights into Phosphonium-Mediated Olefination and Coupling
The core of this innovative synthesis lies in the initial olefination step where a bis-thiophene ketone precursor reacts with a functionalized phosphonium ylide or phosphonate anion. Mechanistically, this involves the deprotonation of the phosphonium salt by a mild inorganic base, such as potassium tert-butoxide or sodium hydride, to generate the reactive ylide species in situ. This ylide then attacks the carbonyl carbon of the ketone, forming a betaine intermediate that collapses to release triphenylphosphine oxide and yield the desired alkene product with high geometric selectivity. This transformation is remarkably tolerant of various functional groups, allowing for the presence of protected hydroxyl functionalities that serve as latent handles for subsequent derivatization. The robustness of this mechanism ensures that impurity profiles remain clean, as side reactions typical of radical-based organometallic processes, such as homocoupling or over-addition, are effectively suppressed. This high level of chemoselectivity is crucial for maintaining the integrity of the sensitive thiophene rings, which are prone to degradation under harsh acidic or basic conditions found in older methodologies.
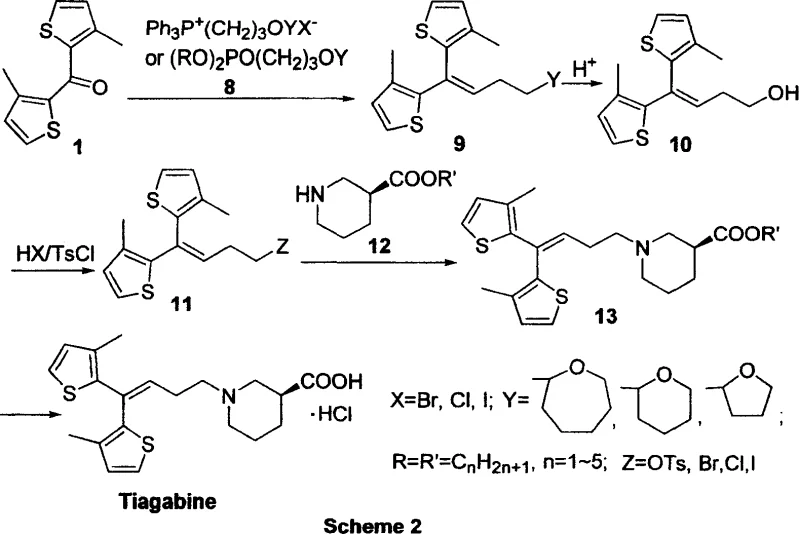
Following the formation of the alkene backbone, the process involves a sophisticated sequence of deprotection and activation to prepare the molecule for coupling with the chiral amine. The protecting group on the hydroxyl terminus is removed under mild acidic conditions, revealing a primary alcohol that is subsequently converted into a superior leaving group, such as a tosylate or halide. This activation step is critical for the final nucleophilic substitution, where the electrophilic side chain reacts with the nitrogen atom of the optically pure beta-piperidine carboxylate. The use of a tosylate or iodide leaving group facilitates this displacement under relatively mild conditions, often aided by catalytic amounts of iodide salts to enhance reaction kinetics via the Finkelstein effect. This mechanistic pathway ensures that the stereocenter on the piperidine ring remains intact throughout the synthesis, preventing racemization and guaranteeing the high enantiomeric excess required for the biological activity of the S-configuration Tiagabine. The final hydrolysis step converts the ester moiety to the free carboxylic acid, completing the synthesis with minimal purification burden.
How to Synthesize Tiagabine Efficiently
The synthesis of Tiagabine via this patented route offers a streamlined protocol that balances high yield with operational simplicity, making it an ideal candidate for technology transfer and commercial adoption. The process begins with the preparation of the olefinic intermediate through the reaction of the bis-thiophene ketone with the appropriate phosphorus reagent, followed by a straightforward workup to isolate the protected alkene. Subsequent steps involve acid-mediated deprotection and activation with tosyl chloride or similar reagents to generate the electrophilic coupling partner. The final assembly involves reacting this activated intermediate with the resolved piperidine ester in the presence of a base and a phase transfer catalyst or iodide source. Detailed standardized operating procedures for each unit operation, including specific solvent choices, temperature ramps, and quenching protocols, are essential for reproducibility. For a comprehensive breakdown of the exact molar ratios, reaction times, and isolation techniques validated in the patent examples, please refer to the technical guide below.
- React bis-thiophene ketone with phosphonium salt or phosphonate reagent under basic conditions to form the protected alkene intermediate.
- Deprotect the hydroxyl group using acid, followed by conversion to a leaving group (halide or tosylate) using thionyl chloride or tosyl chloride.
- Couple the activated intermediate with optically pure beta-piperidine carboxylate ester, followed by hydrolysis and acidification to yield Tiagabine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthesis route delivers profound advantages that extend far beyond simple yield improvements. By eliminating the dependency on pyrophoric reagents and cryogenic infrastructure, manufacturers can significantly lower their capital expenditure requirements and operational risk profiles. The shift to stable, solid reagents simplifies logistics and storage, reducing the need for specialized hazmat handling and allowing for broader supplier qualification. This resilience in the supply chain is critical for maintaining continuous production schedules, especially in a volatile global market where raw material availability can fluctuate. Furthermore, the mild reaction conditions enable the use of standard glass-lined steel reactors that are ubiquitous in the fine chemical industry, removing the barrier to entry for contract manufacturing organizations (CMOs) and facilitating rapid scale-up from pilot plant to commercial tonnage.
- Cost Reduction in Manufacturing: The replacement of expensive organolithium and Grignard reagents with commodity phosphonium salts and inorganic bases results in a drastic reduction in raw material costs. Additionally, the elimination of cryogenic cooling requirements leads to substantial energy savings, while the simplified workup procedures reduce solvent consumption and waste disposal fees. The overall process efficiency is enhanced by the high atom economy of the olefination step and the minimal need for chromatographic purification, driving down the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Utilizing stable, non-hazardous reagents mitigates the risk of supply disruptions caused by the limited availability of specialized organometallics. The robustness of the chemistry allows for longer campaign runs and higher batch success rates, ensuring consistent delivery timelines for downstream formulation partners. This reliability is further bolstered by the versatility of the route, which can accommodate both racemic and chiral starting materials, providing flexibility in sourcing strategies depending on market demand for specific enantiomers.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, as it avoids the production of large quantities of metallic salts and spent organometallic residues. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the burden on wastewater treatment facilities. The scalability is proven by the ability to perform reactions at elevated temperatures without exothermic runaway risks, allowing for safe operation in large-scale reactors and facilitating the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the new synthesis route for Tiagabine considered superior to the 1993 Knudsen method?
A: The new route described in CN1314684C eliminates the need for air-sensitive and expensive organometallic reagents like n-BuLi and Grignard reagents, which require cryogenic conditions (-70°C). Instead, it utilizes robust Wittig or Horner-Wadsworth-Emmons reactions under mild temperatures (0-110°C), significantly improving operational safety and scalability.
Q: How does this process ensure high optical purity for the S-configuration of Tiagabine?
A: The process employs optically pure beta-piperidine carboxylate esters (Compound 12) as starting materials. These chiral building blocks are prepared via resolution with tartaric acid, ensuring that the final coupling step transfers the stereochemistry directly to the final API without the need for complex chiral chromatography at the final stage.
Q: What are the key commercial advantages of this manufacturing method?
A: The method offers substantial cost reduction by replacing precious organometallic catalysts with inexpensive phosphonium salts and inorganic bases. Furthermore, the mild reaction conditions allow for the use of standard industrial reactors, reducing capital expenditure and simplifying waste treatment protocols compared to traditional cryogenic processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tiagabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of essential neurological medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity Tiagabine and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to maximize yield and minimize impurities, guaranteeing a product that exceeds the expectations of regulatory bodies and end-users alike.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits inherent in this process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
