Revolutionizing Tiagabine Production: A Scalable Route for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for central nervous system disorders continues to evolve, with Tiagabine standing out as a critical GABA uptake inhibitor for epilepsy management. The patent CN1651430A introduces a transformative synthesis method that addresses long-standing manufacturing bottlenecks associated with this potent active pharmaceutical ingredient. Unlike traditional approaches that rely on hazardous and costly organometallic chemistry, this innovation leverages stable phosphorus-based coupling strategies to construct the core molecular framework. This shift not only enhances the safety profile of the production facility but also ensures a more consistent supply of high-purity intermediates for downstream drug formulation. For global supply chain leaders, understanding this technological pivot is essential for securing reliable Tiagabine supplier partnerships that can withstand market volatility.
Furthermore, the ability to produce both racemic and S-configuration Tiagabine through this versatile route offers strategic flexibility for generic drug manufacturers aiming to optimize their portfolio. The method specifically targets the elimination of expensive special reagents, which historically acted as a barrier to entry for many potential producers. By utilizing readily available starting materials and mild reaction conditions, the process aligns perfectly with modern green chemistry principles while maintaining rigorous quality standards. This report delves deep into the mechanistic advantages and commercial implications of adopting this patented methodology for large-scale API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
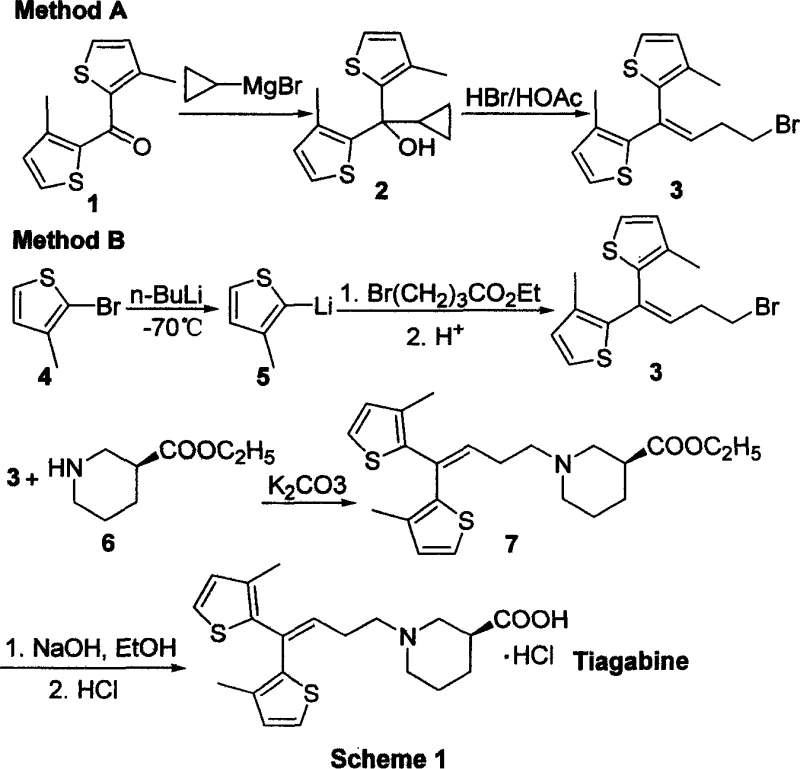
Historical synthetic pathways for Tiagabine, such as those reported by Knudsen et al., heavily depend on the use of highly reactive organometallic reagents like n-butyllithium and Grignard reagents. These substances require stringent cryogenic conditions, often necessitating temperatures as low as -70°C to prevent decomposition and ensure reaction selectivity. Such extreme operational parameters impose a massive burden on industrial infrastructure, requiring specialized cooling systems and increasing energy consumption drastically. Moreover, the air and moisture sensitivity of these reagents introduces significant safety hazards and complicates the handling procedures, leading to potential batch failures and inconsistent yields. For procurement managers, reliance on these conventional methods translates to higher operational expenditures and increased risk of supply chain disruptions due to the complexity of managing hazardous materials.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes phosphorus ylides or phosphonates to effect the crucial carbon-carbon bond formation through Wittig or Horner-Wadsworth-Emmons reactions. This chemical strategy operates effectively under much milder conditions, often at ambient or moderately elevated temperatures, thereby eliminating the need for energy-intensive cryogenic cooling. The reagents involved are significantly more stable and less sensitive to atmospheric conditions, which simplifies the reactor setup and reduces the requirement for inert gas protection. This transition from organometallic to organophosphorus chemistry represents a fundamental improvement in process robustness, allowing for smoother scale-up from laboratory to commercial production volumes. Consequently, this method offers a viable pathway for cost reduction in API manufacturing by streamlining operations and minimizing safety protocols.
Mechanistic Insights into Phosphonate-Mediated Olefination
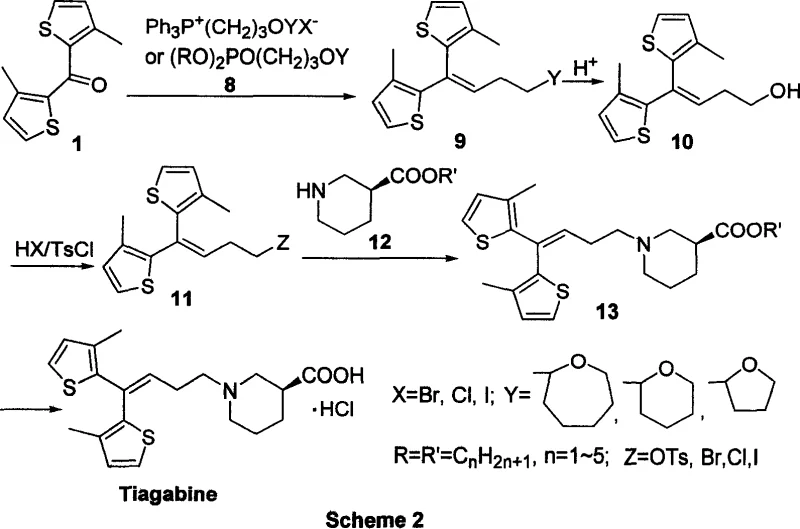
The core of this innovative synthesis lies in the coupling of a thiophene-based ketone with a phosphonate ester to generate the key alkene intermediate with high stereocontrol. The reaction mechanism proceeds through the formation of a betaine or oxaphosphetane intermediate, which subsequently collapses to release the desired olefin and a phosphine oxide byproduct. This step is critical as it establishes the geometric configuration of the side chain, which is essential for the biological activity of the final drug molecule. By optimizing the base and solvent system, the process ensures high conversion rates while minimizing the formation of undesired isomers that could complicate downstream purification. For R&D directors, this level of mechanistic clarity provides confidence in the reproducibility of the synthesis across different manufacturing sites.
Following the olefination, the route incorporates a sophisticated chiral resolution strategy using tartaric acid to isolate the optically pure beta-piperidine carboxylate precursor. This resolution step is pivotal for achieving the specific S-configuration required for the therapeutic efficacy of Tiagabine, ensuring that the final product meets stringent pharmacopoeial standards. The use of classical salt formation and recrystallization techniques allows for the removal of the unwanted enantiomer with high precision, resulting in an optical purity greater than 95%. This focus on impurity control mechanisms ensures that the final API possesses a clean impurity profile, reducing the regulatory burden during drug approval processes. Such attention to stereochemical detail underscores the commitment to producing high-purity pharmaceutical intermediates suitable for sensitive patient populations.
How to Synthesize Tiagabine Efficiently
The implementation of this synthesis route involves a sequence of well-defined chemical transformations that prioritize safety and efficiency at every stage. Starting from readily available thiophene derivatives, the process builds complexity gradually through protected intermediates that are easy to handle and purify. The detailed standardized synthesis steps involve precise control of pH during the hydrolysis and acidification phases to ensure the formation of the stable hydrochloride salt. Operators are guided to maintain specific temperature ranges during the coupling reactions to maximize yield while preventing thermal degradation of sensitive functional groups. For technical teams looking to adopt this technology, the following guide outlines the critical operational parameters necessary for successful execution.
- Perform Wittig or Horner-Wadsworth-Emmons reaction between thiophene ketone and phosphonate reagent to form the alkene intermediate.
- Deprotect the hydroxyl group and convert it to a leaving group such as tosylate or halide under mild acidic conditions.
- Couple the activated intermediate with optically pure beta-piperidine carboxylate followed by hydrolysis to yield Tiagabine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis route offers profound benefits for procurement and supply chain stakeholders who are tasked with optimizing costs and ensuring continuity. The elimination of cryogenic requirements and air-sensitive reagents directly translates to a significant reduction in capital expenditure for specialized equipment and ongoing energy costs. Furthermore, the use of stable, commodity-grade chemicals enhances supply chain reliability by reducing dependence on niche suppliers who may face availability issues. This robustness allows manufacturers to maintain consistent production schedules even during periods of raw material market fluctuation. For supply chain heads, this means a more predictable lead time for high-purity pharmaceutical intermediates and a lower risk of production stoppages.
- Cost Reduction in Manufacturing: The substitution of expensive organolithium reagents with stable phosphonates removes the need for complex safety infrastructure and hazardous waste disposal protocols. This change drastically simplifies the production workflow, leading to substantial cost savings in both labor and material handling. Additionally, the milder reaction conditions reduce energy consumption, contributing to a lower overall carbon footprint and operational expense. By streamlining the synthesis, manufacturers can achieve a more competitive pricing structure without compromising on the quality of the final API product.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. This stability ensures that production can continue uninterrupted, providing a consistent flow of materials to downstream formulation partners. The simplified logistics of handling non-hazardous reagents also accelerate the procurement cycle, allowing for faster response times to market demands. Consequently, partners can rely on a steady supply of critical epilepsy medication components, strengthening the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates easier scale-up from pilot plants to full commercial production without the need for extensive process re-engineering. This scalability is complemented by improved environmental compliance, as the process generates less hazardous waste and consumes fewer resources. The ability to operate within standard chemical processing limits makes this route highly attractive for facilities aiming to meet strict regulatory and sustainability goals. Thus, the method supports sustainable growth and long-term viability in the competitive generic pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tiagabine synthesis technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits for potential manufacturing partners. Understanding these details is crucial for making informed decisions about technology transfer and process adoption. The answers reflect the practical realities of scaling this chemistry in a regulated environment.
Q: Why is the new Tiagabine synthesis route considered superior for industrial scale-up?
A: The new route eliminates the need for cryogenic temperatures and air-sensitive organolithium reagents, significantly reducing infrastructure costs and safety risks associated with large-scale manufacturing.
Q: How is optical purity achieved in this synthesis method?
A: Optical purity is secured through a classical resolution process using tartaric acid salts, which allows for the isolation of the specific S-configuration enantiomer required for therapeutic efficacy with high precision.
Q: Does this method reduce the environmental footprint of Tiagabine production?
A: Yes, by avoiding hazardous pyrophoric reagents and enabling reactions at ambient or mild temperatures, the process minimizes energy consumption and simplifies waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tiagabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthesis routes in securing the global supply of essential medicines like Tiagabine. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to technical excellence allows us to navigate complex chemical challenges, delivering high-purity pharmaceutical intermediates that support your regulatory filings.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next product launch. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you optimize your supply chain and reduce lead time for high-purity pharmaceutical intermediates through our proven manufacturing expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
