Technical Upgrade in Tiagabine Manufacturing for Global Pharmaceutical Supply Chains
The global demand for antiepileptic drugs continues to drive innovation in the manufacturing of key active pharmaceutical ingredients (APIs), with Tiagabine (marketed as Gabitril) representing a critical therapeutic agent for GABA uptake inhibition. A pivotal advancement in this domain is detailed in Chinese Patent CN1288145C, which discloses a robust and economically viable synthesis method for Tiagabine and its stereoisomers. Unlike legacy processes that rely heavily on hazardous and expensive organometallic chemistry, this patented approach utilizes a strategic sequence involving phosphonium salt olefination followed by acid-catalyzed ring opening. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding this technological shift is paramount, as it directly addresses the chronic issues of batch-to-batch variability and high operational costs inherent in older synthetic routes. The patent explicitly highlights the elimination of air-sensitive reagents, positioning this methodology as a superior candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tiagabine has been dominated by routes established in early literature, such as those reported by Knudsen et al., which necessitate the use of highly reactive organometallic species. As illustrated in the prior art schemes, these conventional methods typically involve the generation of Grignard reagents or lithium-halogen exchange intermediates at cryogenic temperatures, often requiring cooling down to -70°C. Such conditions impose severe constraints on manufacturing infrastructure, demanding specialized low-temperature reactors and rigorous exclusion of moisture and oxygen to prevent reagent decomposition. Furthermore, the use of n-butyllithium or similar pyrophoric bases introduces significant safety liabilities, including the risk of thermal runaway and fire, which complicates regulatory compliance and insurance overheads. From a supply chain perspective, the reliance on these sensitive reagents often leads to yield fluctuations and the formation of difficult-to-remove metal impurities, thereby increasing the burden on quality control laboratories and extending production lead times.
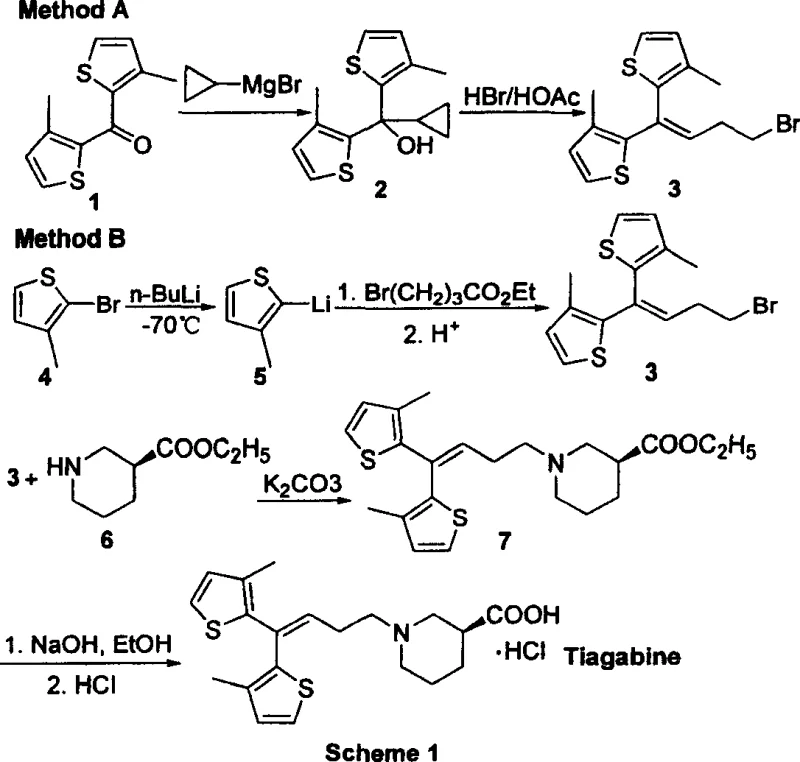
The Novel Approach
In stark contrast to the hazardous legacy pathways, the invention disclosed in CN1288145C introduces a streamlined synthetic strategy that circumvents the need for cryogenic organometallic chemistry entirely. The core innovation lies in the utilization of a functionalized phosphonium salt, which reacts with the bis-thiophene ketone precursor under relatively mild basic conditions to form a cyclopropyl-containing intermediate. This Wittig-type transformation is subsequently followed by a controlled acid-catalyzed ring-opening step, which elegantly extends the carbon chain to install the necessary propyl linker with a terminal halide. This approach not only operates at significantly higher temperatures—ranging from ambient to reflux—thereby eliminating the energy-intensive cooling requirements, but also utilizes reagents that are shelf-stable and easy to handle. By shifting the chemical logic from addition reactions to substitution and rearrangement, the new method offers a pathway that is inherently safer, more reproducible, and ideally suited for cost reduction in API manufacturing through simplified process engineering.
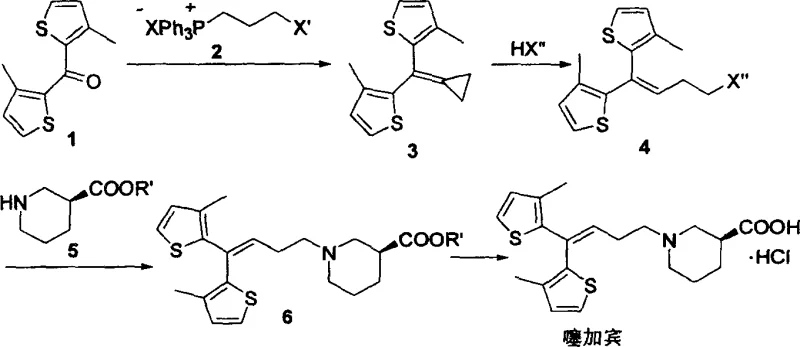
Mechanistic Insights into Phosphonium-Mediated Olefination and Ring Opening
The mechanistic elegance of this new route centers on the formation and subsequent fragmentation of a strained cyclopropane ring, a transformation that provides exceptional control over the regiochemistry of the final alkene. Initially, the deprotonation of the phosphonium salt generates a reactive ylide species in situ, which attacks the carbonyl group of the thiophene ketone to form an oxaphosphetane intermediate. The collapse of this four-membered ring yields the cyclopropyl alkene, a structure that serves as a masked equivalent of the desired linear chain. The subsequent exposure to hydrohalic acids triggers a regioselective ring-opening event, driven by the relief of ring strain and the stabilization of the resulting carbocation or transition state. This step is critical for ensuring the correct placement of the halide leaving group at the terminus of the propyl chain, which is essential for the final nucleophilic substitution with the piperidine moiety. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as acid strength and temperature, to minimize the formation of isomeric byproducts that could compromise the purity of the final drug substance.
Furthermore, the avoidance of strong nucleophiles like organolithiums significantly reduces the risk of side reactions that typically plague thiophene chemistry, such as nucleophilic attack on the sulfur ring or polymerization. In traditional lithiation routes, the high basicity can lead to deprotonation at unwanted positions on the heterocyclic rings, generating complex impurity profiles that are challenging to purge during crystallization. The new method operates under thermodynamic control during the ring-opening phase, favoring the formation of the conjugated alkene system which is thermodynamically more stable. This intrinsic selectivity translates to a cleaner crude reaction mixture, reducing the number of chromatographic or recrystallization steps required to meet stringent pharmacopeial standards. For R&D teams, this implies a more robust process window where minor deviations in stoichiometry or temperature do not result in catastrophic batch failures, thereby enhancing overall process reliability and consistency.
How to Synthesize Tiagabine Intermediates Efficiently
The execution of this synthesis requires careful attention to the preparation of the phosphonium salt and the control of the ring-opening kinetics to ensure high yields. The process begins with the generation of the ylide using a standard alkali base in an aprotic solvent, followed by the addition of the ketone substrate under inert atmosphere to prevent ylide quenching. Once the cyclopropyl intermediate is isolated, it is subjected to acidic conditions where the rate of ring opening must be balanced against potential polymerization or degradation of the sensitive thiophene moieties. The final coupling with the chiral piperidine ester is a straightforward nucleophilic substitution that benefits from the high reactivity of the primary halide generated in the previous step. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- React the bis-thiophene ketone precursor with a functionalized phosphonium salt in the presence of a strong base like potassium tert-butoxide to form a cyclopropyl intermediate.
- Perform an acid-catalyzed ring-opening reaction on the cyclopropyl intermediate using hydrohalic acids to generate the extended alkene chain with a terminal halide.
- Couple the resulting halide with optically pure nipecotic acid ester derivatives followed by hydrolysis to yield the final Tiagabine active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing air-sensitive organolithiums and Grignard reagents with stable phosphonium salts and common mineral acids, the supply chain becomes far more resilient to disruptions. These commodity chemicals are readily available from multiple global vendors, reducing the risk of single-source dependency and allowing for more aggressive negotiation on pricing. Additionally, the elimination of cryogenic processing removes a significant bottleneck in manufacturing capacity, as standard glass-lined or stainless steel reactors can be utilized without the need for specialized low-temperature jackets or brine circulation systems. This flexibility allows manufacturers to maximize asset utilization and respond more rapidly to fluctuations in market demand for antiepileptic medications.
- Cost Reduction in Manufacturing: The economic impact of this process change is profound, primarily driven by the removal of expensive, specialty reagents and the associated infrastructure costs. Organolithium reagents are not only costly to purchase but also require dedicated storage facilities and strict inventory management due to their limited shelf life and hazard classification. By substituting these with inexpensive phosphonium salts and alkali bases, the direct material cost per kilogram of the intermediate is significantly lowered. Moreover, the energy savings from operating at ambient or reflux temperatures rather than -70°C contribute to a lower carbon footprint and reduced utility bills. The simplified purification process, resulting from a cleaner impurity profile, further decreases the consumption of solvents and silica gel, leading to substantial overall cost savings in the production of high-purity Tiagabine.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any pharmaceutical manufacturer, and this new route inherently de-risks the production schedule. The reagents employed are non-pyrophoric and stable under normal atmospheric conditions, which simplifies logistics and warehousing requirements. There is no longer a need for expedited shipping of temperature-controlled hazardous materials, nor the risk of batch rejection due to reagent degradation during transit. This stability ensures that production campaigns can be planned with greater confidence, reducing the likelihood of unplanned downtime caused by reagent quality issues. Consequently, suppliers can offer more reliable lead times and maintain higher safety stock levels of key intermediates, ensuring a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often fraught with challenges, particularly when exothermic organometallic reactions are involved. The thermal profile of the new Wittig-based route is much more manageable, allowing for safer heat dissipation and better temperature control in large-scale reactors. This inherent safety margin facilitates a smoother technology transfer and faster ramp-up to commercial volumes. From an environmental perspective, the process generates less hazardous waste; the absence of heavy metal residues or reactive metal salts simplifies wastewater treatment and disposal protocols. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for meeting the ESG (Environmental, Social, and Governance) criteria of major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does the new synthesis route improve safety compared to traditional methods?
A: The novel route described in CN1288145C eliminates the need for pyrophoric organolithium reagents and cryogenic conditions (-70°C), significantly reducing operational hazards and energy consumption associated with cooling infrastructure.
Q: What are the key purity advantages of avoiding Grignard reagents?
A: By bypassing Grignard or Lithium-Halogen exchange steps, the process minimizes side reactions such as Wurtz coupling or over-addition, leading to a cleaner impurity profile and simplified downstream purification.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the use of stable phosphonium salts and mild reaction conditions (0°C to reflux) makes the process highly scalable, removing the bottlenecks associated with handling air-sensitive reagents on a multi-ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tiagabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific unit operations required for this process, including precise temperature control for the ring-opening step and advanced purification technologies to meet stringent purity specifications. Our rigorous QC labs employ the latest analytical methods to monitor impurity profiles, guaranteeing that every batch of Tiagabine intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your margins. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can secure your supply of this critical antiepileptic ingredient.
