Advanced Manufacturing Strategy for High-Purity Entecavir Intermediates and API
The pharmaceutical landscape for antiviral therapeutics continues to evolve, driven by the relentless demand for higher purity Active Pharmaceutical Ingredients (APIs) and more efficient manufacturing protocols. A pivotal advancement in this domain is documented in Chinese Patent CN101245067B, which discloses a groundbreaking preparation method for Entecavir, a potent inhibitor of hepatitis B virus (HBV) replication. This patent introduces a novel synthetic pathway that fundamentally restructures the approach to constructing the cyclopentene ring system attached to the guanine base, moving away from the cumbersome and low-yielding strategies of the past. The core innovation lies in utilizing a specific trihydroxy intermediate, designated as Compound (II), as the starting material for a streamlined sequence involving hydroxyl protection, cyclization to form the exocyclic double bond, and final deprotection. 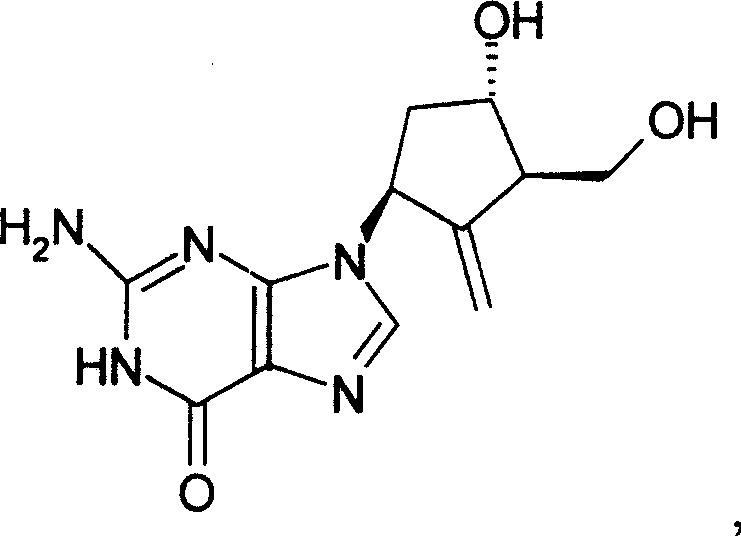 This structural optimization not only enhances the overall chemical yield but also drastically improves the impurity profile, addressing critical pain points for both R&D directors and supply chain managers who require reliable sources of high-quality nucleoside analogues for global distribution.
This structural optimization not only enhances the overall chemical yield but also drastically improves the impurity profile, addressing critical pain points for both R&D directors and supply chain managers who require reliable sources of high-quality nucleoside analogues for global distribution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial synthesis of Entecavir was plagued by significant inefficiencies, as exemplified by the routes disclosed in documents such as US7034152 and WO98/09964. These conventional methods typically relied on a strategy where protecting groups were installed first, followed by cyclization to form the double bond, and finally hydrolysis using boron trifluoride. 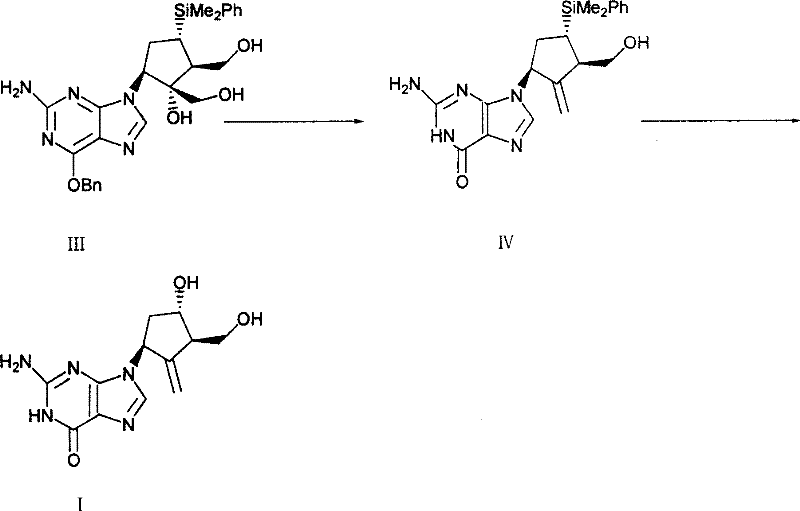 A major drawback of these legacy processes was the notoriously low yield, often stagnating around 34.1% for key intermediates, which translates to massive material loss and inflated production costs. Furthermore, the purification of the resulting products was exceptionally challenging; even after multiple refining steps, purity rarely exceeded 98.5% by area normalization. The chemical nature of the intermediates in these old routes made them prone to generating impurities with polarities very similar to the target product, rendering chromatographic separation difficult and expensive. Additionally, the physical properties of the reaction mixtures posed severe operational hazards, as the intermediates tended to polymerize easily and stick tenaciously to reactor walls, making scraping and recovery labor-intensive and unfavorable for realizing suitability for industrialized production on a multi-ton scale.
A major drawback of these legacy processes was the notoriously low yield, often stagnating around 34.1% for key intermediates, which translates to massive material loss and inflated production costs. Furthermore, the purification of the resulting products was exceptionally challenging; even after multiple refining steps, purity rarely exceeded 98.5% by area normalization. The chemical nature of the intermediates in these old routes made them prone to generating impurities with polarities very similar to the target product, rendering chromatographic separation difficult and expensive. Additionally, the physical properties of the reaction mixtures posed severe operational hazards, as the intermediates tended to polymerize easily and stick tenaciously to reactor walls, making scraping and recovery labor-intensive and unfavorable for realizing suitability for industrialized production on a multi-ton scale.
The Novel Approach
In stark contrast, the methodology presented in CN101245067B offers a robust solution by inverting the traditional logic and optimizing reaction conditions to favor the desired thermodynamic products. 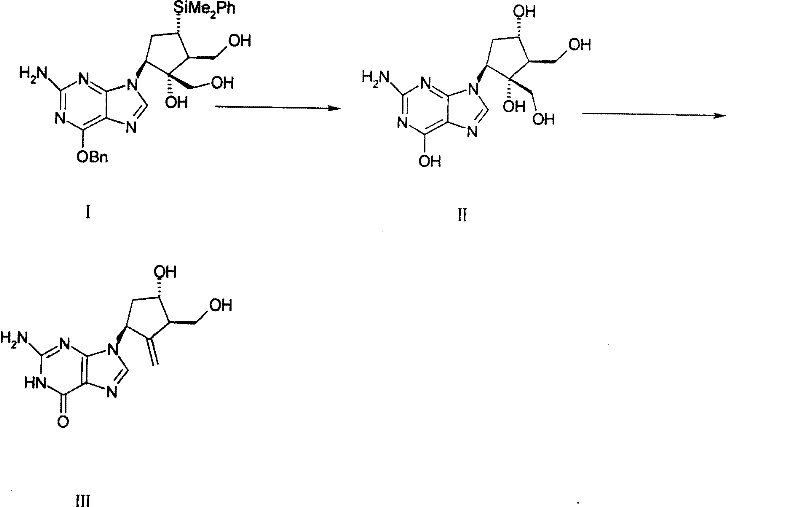 This new route begins with Compound (II), a trihydroxy species, and subjects it to a carefully controlled hydroxyl protection reaction followed by a cyclization step that efficiently generates the crucial exocyclic double bond. By shifting the sequence and utilizing specific reagents like triethyl orthoformate or 2,2-dimethoxypropane under acidic catalysis, the process achieves a remarkable yield improvement, reaching up to 45.3% after technological modifications. More importantly, the purity of the final Entecavir product can reach an impressive 99.7%, surpassing the stringent requirements for antiviral APIs. The operational simplicity of this new route cannot be overstated; it eliminates the sticky polymerization issues of the past, ensuring that the reaction mass remains manageable throughout the synthesis. This ease of operation directly correlates to reduced batch failure rates and lower maintenance costs for manufacturing facilities, making it a superior choice for cost reduction in antiviral drug manufacturing.
This new route begins with Compound (II), a trihydroxy species, and subjects it to a carefully controlled hydroxyl protection reaction followed by a cyclization step that efficiently generates the crucial exocyclic double bond. By shifting the sequence and utilizing specific reagents like triethyl orthoformate or 2,2-dimethoxypropane under acidic catalysis, the process achieves a remarkable yield improvement, reaching up to 45.3% after technological modifications. More importantly, the purity of the final Entecavir product can reach an impressive 99.7%, surpassing the stringent requirements for antiviral APIs. The operational simplicity of this new route cannot be overstated; it eliminates the sticky polymerization issues of the past, ensuring that the reaction mass remains manageable throughout the synthesis. This ease of operation directly correlates to reduced batch failure rates and lower maintenance costs for manufacturing facilities, making it a superior choice for cost reduction in antiviral drug manufacturing.
Mechanistic Insights into Hydroxyl Protection and Cyclization
The success of this novel synthesis hinges on the precise control of the hydroxyl protection and subsequent elimination mechanisms. The process initiates by dissolving Compound (II) in a solvent such as methylene dichloride under an inert argon shield to prevent oxidative degradation. A catalyst, typically pyridinium p-toluenesulfonate (PPTS), is added, and the mixture is cooled to 0°C before the dropwise addition of a radical protection agent like triethyl orthoformate. This low-temperature addition is critical for controlling the exothermic nature of the orthoester formation, ensuring that the primary and secondary hydroxyl groups are selectively protected without inducing premature elimination or degradation of the sensitive purine base. Following the protection phase, the reaction mixture is allowed to warm to room temperature, facilitating the completion of the acetal or orthoester formation. Subsequently, the protected intermediate undergoes a thermal treatment with acetic anhydride at temperatures ranging from 50°C to 120°C. This step is the mechanistic heart of the transformation, where the acetic anhydride acts not merely as an acetylating agent but drives the elimination reaction that forms the exocyclic methylene group. The thermal energy provided in this range overcomes the activation barrier for the E2-type elimination, establishing the double bond geometry required for biological activity while maintaining the stereochemical integrity of the cyclopentane ring.
Impurity control in this mechanism is achieved through the strategic selection of deprotection conditions in the final stage. After the cyclization, the blocking groups are removed using mineral alkalis such as sodium hydroxide or potassium hydroxide, or organic bases like sodium alkoxide, at pH levels between 10 and 12. This alkaline hydrolysis is conducted at moderate temperatures (45-75°C) to ensure complete removal of acetate and orthoester groups without hydrolyzing the glycosidic bond connecting the sugar mimic to the guanine base. The use of specific solvents like C1-C5 lower alcohols during the acid-catalyzed intermediate steps further aids in solubilizing polar byproducts, allowing them to be washed away during the aqueous workup. By avoiding the use of harsh Lewis acids like boron trifluoride in the final deprotection step—a common source of side reactions in prior art—the process minimizes the formation of depurinated byproducts and regioisomers. This meticulous attention to reaction parameters ensures that the final crude product requires minimal purification, often needing only a single recrystallization or a simple column chromatography step to achieve the target 99.7% purity specification.
How to Synthesize Entecavir Efficiently
The synthesis of Entecavir via this patented route represents a paradigm shift in nucleoside analogue manufacturing, offering a clear pathway from readily available intermediates to the high-value final API. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to pilot plant and eventually to commercial production scales. Operators must strictly adhere to the specified temperature gradients and reagent stoichiometry to maximize the yield of the cyclization step, which is the yield-determining step of the entire sequence. The detailed standardized synthesis steps, including exact molar ratios, stirring speeds, and quenching procedures, are outlined below to ensure reproducibility and safety across different manufacturing sites.
- Perform hydroxyl protection on Compound II using radical protection agents like triethyl orthoformate or 2,2-dimethoxypropane under acidic catalysis.
- Execute cyclization and elimination reactions using acetic anhydride at elevated temperatures (50-120°C) to establish the critical exocyclic double bond.
- Complete the synthesis by removing blocking groups through hydrolysis with mineral alkali or organic bases to obtain the final Entecavir product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101245067B translates into tangible economic and logistical benefits that extend far beyond simple chemistry. The primary advantage lies in the substantial increase in overall process yield, which jumps from the historical average of roughly 34% to over 45%. This efficiency gain means that for every ton of raw material purchased, the manufacturer obtains significantly more finished product, effectively lowering the cost of goods sold (COGS) without compromising quality. Furthermore, the elimination of difficult-to-remove impurities reduces the reliance on expensive preparative HPLC or multiple recrystallization cycles, thereby cutting down on solvent consumption and waste disposal costs. This aligns perfectly with the industry's push towards greener chemistry and cost reduction in antiviral drug manufacturing, allowing companies to maintain healthy margins even in a competitive generic market.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly impacts the bottom line by reducing the number of unit operations and the quantity of reagents required. By avoiding the use of exotic or hazardous reagents like boron trifluoride in the final steps and replacing them with common mineral bases, the process lowers the cost of raw materials and simplifies the handling requirements. The higher yield means less starting material is wasted, and the reduced need for extensive purification lowers the consumption of chromatography media and solvents. Consequently, the total manufacturing cost per kilogram of Entecavir is significantly reduced, providing a competitive pricing advantage for suppliers who adopt this technology.
- Enhanced Supply Chain Reliability: One of the most critical supply chain risks in API manufacturing is batch failure due to uncontrollable side reactions or physical processing issues. The prior art methods suffered from severe polymerization and wall-sticking, which often led to lost batches and unpredictable delivery schedules. The new process mitigates these risks by ensuring the reaction mixture remains fluid and manageable throughout the synthesis. This operational robustness ensures consistent batch-to-batch quality and reliable delivery times, which is essential for maintaining the trust of downstream pharmaceutical clients who depend on uninterrupted supply for their finished dosage form production.
- Scalability and Environmental Compliance: Scaling up chemical processes often reveals hidden bottlenecks, but this novel route is inherently designed for industrial feasibility. The avoidance of sticky polymers means that reactor cleaning times are drastically shortened, increasing the overall equipment effectiveness (OEE) of the manufacturing facility. Additionally, the use of more benign reagents and the generation of fewer toxic byproducts simplify the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. This makes the commercial scale-up of complex nucleoside analogues not only technically feasible but also environmentally sustainable, future-proofing the supply chain against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Entecavir synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior alternative to existing commercial processes. Understanding these nuances is vital for technical teams evaluating potential technology transfers or licensing opportunities.
Q: How does the new synthesis route improve yield compared to conventional methods?
A: The novel route described in patent CN101245067B achieves a yield of 45.3%, significantly higher than the 34.1% yield typical of prior art methods like those disclosed in US7034152, primarily by optimizing the cyclization and deprotection sequence.
Q: What specific purity levels can be achieved with this optimized process?
A: Through improved operational controls and the avoidance of difficult-to-remove impurities common in older routes, this process consistently delivers Entecavir with a purity exceeding 99.7% after standard purification steps.
Q: Why is this method considered more suitable for industrial scale-up?
A: The process eliminates the severe polymerization and wall-sticking issues associated with previous methods, ensuring smoother reactor operations, easier product recovery, and reduced downtime for cleaning, which are critical for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a robust commercial process requires deep expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101245067B are fully realized in practice. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Entecavir or its intermediates meets the highest international standards. We understand the critical nature of antiviral supply chains and are committed to delivering high-purity Entecavir that supports the global fight against hepatitis B.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your antiviral product portfolio.
