Advanced Manufacturing of Entecavir Intermediates: A Technical Breakdown of Patent CN101148450A
Advanced Manufacturing of Entecavir Intermediates: A Technical Breakdown of Patent CN101148450A
The pharmaceutical landscape for antiviral therapeutics, particularly for the treatment of Hepatitis B virus (HBV), relies heavily on the efficient and scalable production of high-purity nucleoside analogs. Patent CN101148450A, published in March 2008, presents a pivotal advancement in the preparation of 2-amino-9-[(1S, 3S, 4S)-4-hydroxy-3-hydroxymethyl-2-methylene cyclopentyl]-1,9-dihydro-6H-purine-6-one, widely known as Entecavir. This document outlines a sophisticated synthetic strategy that addresses historical challenges regarding yield and raw material availability. By leveraging a unique starting material, specifically [1S-(1α, 2β, 3α, 5α)]-3-(benzyloxy)-2-[(benzyloxy)methyl]-6-oxabicyclo[3.1.0]hexane, the inventors have constructed a route that bypasses the inefficiencies of earlier methodologies. The structural complexity of this molecule requires precise stereochemical control, which is achieved through a sequence involving imide condensation, selective oxidation, and a critical methylenation step. 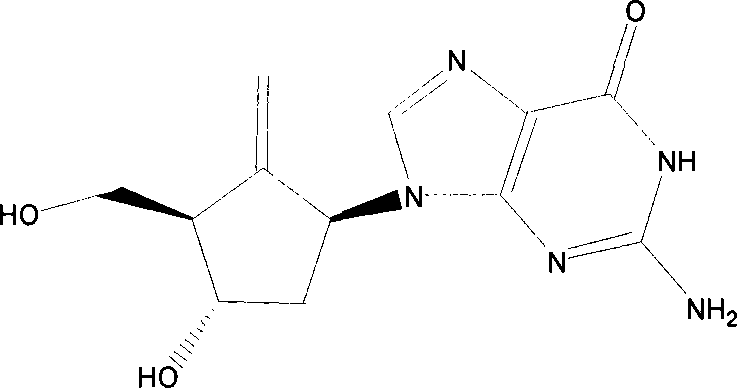
For R&D directors and process chemists, the significance of this patent lies in its detailed manipulation of the cyclopentane core. The introduction of the exocyclic methylene group at the C2 position is a defining feature of Entecavir's biological activity, and achieving this without compromising the integrity of the adjacent chiral centers is non-trivial. The patent describes a pathway that not only secures the correct stereochemistry but also integrates seamlessly with the subsequent formation of the purine ring. This holistic approach to intermediate synthesis ensures that the final active pharmaceutical ingredient (API) meets the rigorous purity standards required for clinical application. Furthermore, the identification of Formula 6 compounds as valuable antiviral intermediates in their own right adds an additional layer of economic value to the production stream, suggesting potential for diversified product portfolios from a single manufacturing line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN101148450A, the industrial synthesis of Entecavir intermediates was plagued by significant inefficiencies that impacted both cost and throughput. Historical methods often struggled with the construction of the carbocyclic ring system, frequently relying on resolution of racemic mixtures or multi-step sequences that eroded overall yield. The background technology section of the patent explicitly notes that existing domestic raw material production methods yielded only approximately 50% of the theoretical maximum. Such low yields are economically unsustainable in the competitive generic pharmaceutical market, where margin compression is a constant pressure. Additionally, conventional routes often utilized harsh reaction conditions or expensive chiral catalysts that were difficult to remove, leading to impurity profiles that complicated downstream purification. The reliance on scarce or difficult-to-synthesize starting materials further exacerbated supply chain vulnerabilities, creating bottlenecks that could delay time-to-market for finished drug products. These limitations necessitated a fundamental rethinking of the synthetic disconnection strategies employed in nucleoside analog manufacturing.
The Novel Approach
The novel approach presented in this patent fundamentally shifts the paradigm by utilizing a readily available chiral pool starting material, the oxabicyclo[3.1.0]hexane derivative. This strategic choice eliminates the need for early-stage resolution, as the starting material already possesses the requisite stereochemical information. The route proceeds through a logical progression of functional group interconversions: first, the ring is opened and functionalized with a nitrogen nucleophile; second, the alcohol is oxidized to a ketone; and third, the critical exocyclic double bond is installed. This sequence is designed to maximize atom economy and minimize the number of isolation steps. By integrating the formation of the purine base later in the sequence, the chemists ensure that the sensitive heterocyclic system is not exposed to the vigorous conditions required for the cyclopentane modification. The result is a streamlined process that reportedly achieves high product yields and utilizes raw materials that are easy to obtain. This represents a substantial improvement over the state of the art, offering a robust platform for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nysted Methylenation and Purine Cyclization
A deep dive into the reaction mechanism reveals the sophistication of the chemical transformations employed, particularly in the installation of the exocyclic methylene group. The conversion of the ketone intermediate (Formula 4) to the methylene compound (Formula 5) utilizes the Nysted reagent in the presence of titanium tetrachloride (TiCl4). This reaction, conducted at cryogenic temperatures ranging from -78°C to 20°C, is a variant of the Tebbe olefination but offers distinct advantages in terms of functional group tolerance and stereoselectivity. The titanium species activates the methylene transfer agent, facilitating the attack on the carbonyl carbon to form an oxatitanacyclobutane intermediate, which subsequently collapses to release the alkene. The low temperature control is crucial here, as it prevents side reactions such as polymerization or isomerization of the sensitive cyclopentane ring. Following this, the coupling with the pyrimidine precursor (Formula 6) involves a nucleophilic aromatic substitution where the amine on the cyclopentane ring displaces a leaving group on the heterocycle. 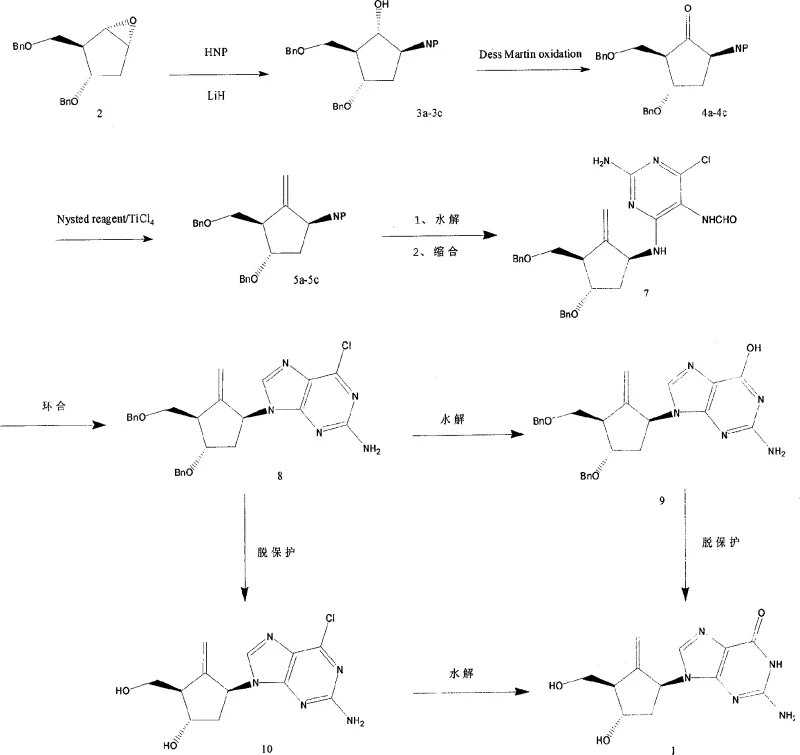
The subsequent cyclization step to form the purine ring is equally critical for ensuring product quality. The patent describes the use of triethyl orthoformate in the presence of concentrated hydrochloric acid to effect ring closure. This acid-catalyzed condensation facilitates the formation of the imidazole portion of the purine system, fusing it to the existing pyrimidine ring. The mechanism likely involves the activation of the formamidine nitrogen by the orthoformate, followed by intramolecular attack by the amino group. Controlling the acidity and temperature (0°C to 85°C) during this step is vital to prevent the hydrolysis of the newly formed glycosidic bond or the degradation of the exocyclic methylene group. From an impurity control perspective, understanding these mechanisms allows process chemists to identify potential byproducts, such as regioisomers or over-alkylated species, and implement specific quenching or purification protocols to mitigate them. The final deprotection using boron trichloride (BCl3) is a standard but delicate operation that requires careful monitoring to ensure complete removal of benzyl groups without affecting the purine core.
How to Synthesize Entecavir Intermediate Efficiently
The synthesis of this high-value nucleoside intermediate requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the succinimide-substituted cyclopentane, followed by oxidation and methylenation to establish the core scaffold. The subsequent coupling with the pyrimidine base and cyclization forms the heterocyclic system, which is then deprotected to reveal the final active structure. Each step demands precise control of stoichiometry, temperature, and reaction time to maintain the integrity of the chiral centers and the exocyclic double bond. While the general workflow is established, the specific operational details regarding workup procedures, solvent choices, and crystallization conditions are critical for achieving the reported high yields and purity levels. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol below.
- Functionalize the cyclopentane core by opening the oxabicyclo hexane epoxide with an imide using LiH, followed by Dess-Martin oxidation and Nysted methylenation to introduce the exocyclic double bond.
- Couple the functionalized cyclopentane amine with 2-amino-4-chloro-5-nitropyrimidinone, followed by cyclization using triethyl orthoformate and acid to form the purine ring system.
- Perform final deprotection of benzyl groups using boron trichloride and hydrolyze any remaining protecting groups to yield the target nucleoside compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the methodology described in CN101148450A offers tangible strategic benefits that extend beyond simple chemical curiosity. The primary advantage lies in the significant reduction of production costs driven by the dramatic improvement in process yield. By moving from historical yields of roughly 50% to the substantially higher efficiencies reported in this patent, manufacturers can drastically reduce the amount of raw material required per kilogram of final product. This efficiency gain directly translates to lower variable costs and improved margin potential, which is essential in the highly price-sensitive generic antiviral market. Furthermore, the elimination of complex resolution steps early in the synthesis simplifies the manufacturing workflow, reducing the demand for specialized chiral columns or expensive resolving agents. This simplification not only lowers direct material costs but also reduces the operational overhead associated with managing complex inventory and waste streams.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route inherently drives down manufacturing expenses by minimizing the number of unit operations and maximizing throughput. The use of readily available starting materials, specifically the oxabicyclo hexane derivative, mitigates the risk of price volatility associated with exotic or custom-synthesized reagents. Additionally, the high yield of the key coupling and cyclization steps means that less solvent and energy are consumed per unit of output, contributing to a leaner cost structure. The ability to utilize common reagents like Dess-Martin periodinane and Nysted reagent, which are commercially scalable, ensures that the supply chain remains robust and cost-effective. Consequently, this process enables a reliable pharmaceutical intermediate supplier to offer competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for API manufacturers, and this route enhances reliability by relying on commodity chemicals and stable intermediates. The starting materials are described as "easily obtained," which suggests a broad supplier base and reduced risk of single-source dependency. This diversification of the supply base protects against disruptions caused by geopolitical issues or production failures at specific vendor sites. Moreover, the robustness of the reaction conditions, particularly the tolerance of the Nysted methylenation to various functional groups, reduces the likelihood of batch failures due to minor variations in raw material quality. This predictability allows for more accurate production planning and inventory management, ensuring that lead times for high-purity pharmaceutical intermediates remain consistent and reliable for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process demonstrates favorable characteristics for large-scale production. The reactions are conducted in standard organic solvents like DMF, dichloromethane, and ethanol, which are well-understood in terms of recovery and recycling infrastructure. The high atom economy of the key steps reduces the generation of hazardous waste, aligning with increasingly stringent environmental regulations. The ability to scale this process from laboratory quantities to commercial tonnage is supported by the use of standard equipment and non-exotic catalysts. This scalability ensures that the manufacturing capacity can be ramped up quickly to meet surges in demand, such as those seen during public health crises, without requiring massive capital investment in new specialized reactors. Thus, the process supports the commercial scale-up of complex pharmaceutical intermediates in an environmentally responsible manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Entecavir intermediates as described in the patent literature. These answers are derived directly from the experimental data and claims presented in CN101148450A, providing a factual basis for decision-making. Understanding these nuances is critical for stakeholders evaluating the feasibility of adopting this technology for their own production lines. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application.
Q: What are the primary yield advantages of this synthesis method compared to prior art?
A: The patent explicitly highlights that previous methods suffered from low yields (around 50%), whereas this novel route utilizing the specific oxabicyclo starting material and Nysted methylenation achieves significantly higher yields, with specific steps reporting yields up to 82% and overall process efficiency improvements.
Q: Is the starting material for this Entecavir intermediate synthesis commercially viable?
A: Yes, the patent emphasizes that the starting material, [1S-(1α, 2β, 3α, 5α)]-3-(benzyloxy)-2-[(benzyloxy)methyl]-6-oxabicyclo[3.1.0]hexane, is easily obtained, which resolves a major bottleneck in the supply chain for manufacturing antiviral nucleoside analogs.
Q: How does this process handle the critical exocyclic methylene group installation?
A: The process utilizes a robust Nysted methylenation reaction mediated by titanium tetrachloride (TiCl4) at low temperatures (-78°C to 20°C), ensuring precise stereocontrol and high conversion of the ketone intermediate to the desired methylene cyclopentane structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antiviral medications. Our technical team has extensively analyzed the methodology presented in CN101148450A and possesses the expertise to implement this high-yield process effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Entecavir intermediate we produce meets the highest international standards. We understand that in the pharmaceutical industry, quality is not just a metric but a mandate, and our commitment to excellence is reflected in our advanced analytical capabilities and process control systems.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of this novel synthetic pathway can drive value, reduce risk, and secure your position in the global antiviral market.
