Scalable Production of High-Purity Atorvastatin Side Chain Intermediate via Optimized Nitro-Alkane Route
Scalable Production of High-Purity Atorvastatin Side Chain Intermediate via Optimized Nitro-Alkane Route
The global demand for high-quality statin intermediates continues to surge as cardiovascular therapies remain a cornerstone of modern medicine. Patent CN102391243B introduces a robust and economically viable preparation method for the critical atorvastatin intermediate, (3R,5R)-7-amino-3,5-O-isopropylidene-3,5-dihydroxyheptanoic acid tert-butyl ester. This technical breakthrough addresses long-standing inefficiencies in chiral synthesis by utilizing a streamlined seven-step sequence starting from readily available (R)-epichlorohydrin. Unlike legacy processes that rely on costly chiral resolution or cryogenic conditions, this novel approach leverages efficient nitro-alkane chemistry and zinc-mediated coupling to achieve yields between 80% and 86% with exceptional purity levels exceeding 99.0%. For pharmaceutical manufacturers seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process intensification and cost-effective manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key atorvastatin side chain has been plagued by complex multi-step sequences that hinder industrial scalability. Prior art, such as the route described in patent CN 200910061164.4A, relies on Salen(Co) catalysts for chiral resolution followed by tedious ring-opening and esterification steps. As illustrated in the reaction scheme below, these conventional pathways often necessitate up to eight distinct synthetic operations, each introducing potential yield losses and impurity profiles that complicate downstream purification.
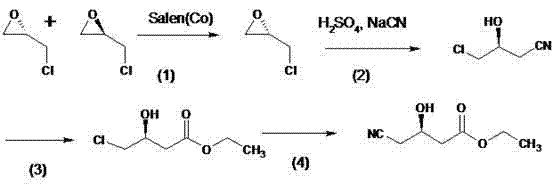
Furthermore, alternative methods reported in documents like US20090216029A1 frequently depend on the use of noble metal lithium compounds and require extreme cryogenic conditions, specifically temperatures as low as -80°C. These harsh operational parameters not only drive up energy costs drastically but also pose significant safety challenges in a large-scale reactor environment. The reliance on such specialized reagents and equipment creates bottlenecks in the supply chain, making cost reduction in API manufacturing difficult to achieve while maintaining consistent quality standards.
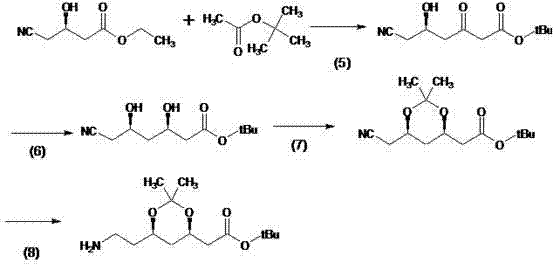
The Novel Approach
In stark contrast, the methodology outlined in CN102391243B circumvents these obstacles by bypassing the synthesis of the inefficient intermediate (R)-(-)-4-cyano-3-hydroxybutyric acid ethyl ester entirely. By initiating the sequence with a direct ring-opening of (R)-epichlorohydrin with nitromethane, the process establishes the carbon backbone more efficiently. This strategic shift reduces the total number of unit operations and eliminates the need for ultra-low temperature reactors, thereby simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. The result is a more linear, predictable, and economically attractive production flow that aligns perfectly with the needs of modern generic drug developers.
Mechanistic Insights into Zinc-Mediated Coupling and Stereoselective Reduction
The core chemical innovation of this process lies in the precise control of stereochemistry during the carbon-carbon bond formation and subsequent reduction steps. In step three and four, the generation of the tert-butyl bromoacetate zinc reagent serves as a pivotal Reformatsky-type coupling agent. This organozinc species reacts with the nitrile-containing precursor under mild conditions (0-50°C) to extend the carbon chain while preserving the chiral integrity established in the initial epoxide opening. The use of zinc powder activated with cuprous bromide ensures high reactivity without the pyrophoric hazards associated with other organometallic reagents, facilitating a safer reaction environment for operators.
Following the coupling, the stereoselective reduction of the ketone moiety in step five is critical for establishing the correct 3R,5R configuration required for biological activity. The protocol employs sodium borohydride in the presence of diethylmethoxyborane at controlled low temperatures (-50°C to -80°C), which is significantly warmer and easier to manage than the -80°C extremes of prior art. This specific reagent combination directs the hydride attack to the desired face of the carbonyl group, minimizing the formation of diastereomeric impurities. Subsequent acetonide protection locks this stereochemistry in place, ensuring that the final hydrogenation step yields the target amine with the high-purity pharmaceutical intermediate specifications demanded by regulatory bodies.
How to Synthesize (3R,5R)-7-Amino-3,5-O-isopropylidene-3,5-dihydroxyheptanoic Acid Tert-Butyl Ester Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control, particularly during the exothermic cyanation and reduction phases. The detailed standardized synthesis steps provided in the patent offer a clear roadmap for replicating these results in a pilot or production plant setting. Operators must ensure rigorous exclusion of moisture during the organozinc formation and maintain precise pH control during the workup phases to maximize recovery. For a comprehensive breakdown of the specific operating parameters, please refer to the technical guide below.
- Ring-opening of (R)-epichlorohydrin with nitromethane using BF3 or HCl catalyst to form 1-chloro-4-nitro-(R)-2-butanol.
- Cyanation reaction with sodium cyanide in ethanol/water to yield (R)-3-hydroxy-5-nitro-valeronitrile.
- Reformatsky-type reaction with tert-butyl bromoacetate zinc reagent followed by stereoselective reduction and acetonide protection.
- Final catalytic hydrogenation using Raney nickel to reduce the nitro group to the primary amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented route offers substantial strategic benefits by decoupling production from volatile specialty reagent markets. The substitution of expensive lithium reagents with common zinc powder and the elimination of ultra-low temperature requirements directly translate to lower operational expenditures. This shift allows manufacturing teams to utilize standard stainless steel reactors rather than specialized cryogenic vessels, significantly reducing capital expenditure barriers for new production lines. Consequently, this leads to significant cost savings in the overall manufacturing budget without compromising the quality of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of noble metal lithium compounds and the avoidance of energy-intensive cryogenic cooling systems drastically reduce both raw material and utility costs. By operating at milder temperatures ranging from 0°C to 50°C for most steps, the process minimizes the load on refrigeration infrastructure, allowing for continuous operation with lower energy consumption. Furthermore, the high overall yield of 80% to 86% means less raw material is wasted per kilogram of product, optimizing the cost of goods sold and improving margin potential for the final drug product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as (R)-epichlorohydrin, nitromethane, and zinc powder ensures a stable and resilient supply chain. Unlike processes dependent on custom-synthesized chiral catalysts or scarce organolithium reagents, this method utilizes materials that are widely available from multiple global vendors. This diversification of the supplier base mitigates the risk of production stoppages due to single-source failures, ensuring reducing lead time for high-purity pharmaceutical intermediates and guaranteeing consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The simplified workflow with fewer purification steps and the use of recyclable solvents like ethyl acetate and toluene enhance the environmental profile of the synthesis. The avoidance of heavy metal catalysts in the final steps, utilizing instead Raney nickel which can be filtered and recovered, simplifies waste treatment protocols. This alignment with green chemistry principles facilitates easier regulatory approval for new drug master files and supports the long-term sustainability goals of modern pharmaceutical enterprises seeking to minimize their ecological footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process robustness and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this process improve upon traditional lithium-based synthesis routes?
A: This method eliminates the need for expensive noble metal lithium compounds and avoids ultra-low temperature conditions (-80°C) required in prior art, significantly simplifying industrial scalability and reducing energy consumption.
Q: What is the expected purity and yield of the final intermediate?
A: According to patent CN102391243B, the optimized process achieves an overall yield ranging from 80% to 86% with a final product purity of ≥99.0%, meeting stringent pharmaceutical standards.
Q: Are the raw materials readily available for large-scale production?
A: Yes, the synthesis utilizes commercially abundant starting materials such as (R)-epichlorohydrin and nitromethane, ensuring a stable supply chain and mitigating risks associated with specialty reagent shortages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R,5R)-7-Amino-3,5-O-isopropylidene-3,5-dihydroxyheptanoic Acid Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this optimized route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets the ≥99.0% purity benchmark required for global regulatory submissions.
We invite you to collaborate with us to leverage this advanced synthesis technology for your atorvastatin supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's efficiency and profitability.
