Revolutionizing Atorvastatin Intermediate Production with Safe Catalytic Asymmetric Synthesis
The global demand for lipid-lowering agents continues to drive innovation in the synthesis of key pharmaceutical building blocks, particularly those required for HMG-CoA reductase inhibitors. Patent CN102827030B introduces a groundbreaking preparation method for (3R,5R)-3,5-dihydroxy-6-cyanohexanoate, a critical chiral intermediate in the production of Atorvastatin calcium. This technology addresses long-standing challenges in the industry by offering a route that avoids the use of highly toxic inorganic cyanides, which have traditionally posed severe environmental and safety hazards during large-scale manufacturing. By leveraging asymmetric catalytic alcoholysis and controlled condensation reactions, this method achieves high stereoselectivity and yield under mild reaction conditions. The structural integrity of the target molecule is paramount for downstream biological activity, and this novel approach ensures precise control over the chiral centers at the 3 and 5 positions. 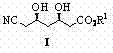
For procurement managers and supply chain directors, the shift away from hazardous reagents translates directly into reduced regulatory burden and lower operational risk. The ability to source a reliable pharmaceutical intermediate supplier who utilizes such environmentally friendly protocols is increasingly becoming a prerequisite for partnerships with top-tier multinational pharmaceutical companies. This synthesis pathway not only enhances the safety profile of the manufacturing process but also simplifies the purification workflow, leading to substantial cost savings in waste management and worker protection. Furthermore, the scalability of this method has been validated through multiple embodiments, demonstrating its robustness from laboratory bench to potential commercial production lines. As the industry moves towards greener chemistry standards, adopting this technology positions stakeholders at the forefront of sustainable pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of statin intermediates like (3R,5R)-3,5-dihydroxy-6-cyanohexanoate has relied heavily on routes involving the ring-opening of chiral epoxides using alkali metal cyanides such as potassium cyanide or sodium cyanide. These traditional methods, while chemically effective, introduce significant safety liabilities due to the extreme toxicity of cyanide salts, requiring specialized equipment and rigorous containment protocols to prevent exposure. Additionally, the handling of these reagents generates hazardous waste streams that are costly and complex to treat, often necessitating dedicated destruction facilities that increase the overall cost reduction in pharmaceutical intermediate manufacturing. The reaction conditions for these cyanide-based openings can also be harsh, sometimes requiring cryogenic temperatures or strong bases that limit the choice of protecting groups and solvents. Moreover, the stereochemical outcome in some older routes depends on enzymatic resolutions which can suffer from limited substrate scope and difficulties in enzyme sourcing, creating bottlenecks in the supply chain continuity. These factors collectively contribute to higher production costs and longer lead times for high-purity pharmaceutical intermediates, making them less attractive for modern, efficiency-driven supply chains.
The Novel Approach
In stark contrast, the method disclosed in CN102827030B utilizes a strategic condensation with cyanoacetate rather than direct cyanide substitution, effectively bypassing the need for toxic inorganic cyanides entirely. This innovative route begins with the asymmetric alcoholysis of 3-silyloxycyclopentanoic anhydride, a transformation catalyzed by chiral quinine derivatives that set the initial stereochemistry with high precision. The subsequent steps involve mild decarboxylation and desilylation processes that proceed under controllable thermal conditions, avoiding the extreme pH swings often seen in legacy methods. By integrating a chiral auxiliary during the final reduction step, the process ensures the formation of the desired (3R,5R) diol configuration without the need for difficult chromatographic separations of diastereomers. This streamlined approach not only improves the overall safety profile of the facility but also enhances the atom economy of the synthesis, aligning with the principles of green chemistry. For a reliable agrochemical intermediate supplier or pharma partner, this means a more resilient production capability that is less susceptible to regulatory shutdowns or raw material shortages associated with controlled substances.
Mechanistic Insights into Quinine-Catalyzed Asymmetric Alcoholysis
The core of this technological advancement lies in the enantioselective desymmetrization of the meso-anhydride starting material using a chiral organocatalyst. Specifically, the use of quinine thiourea or sulfonamide derivatives facilitates a highly selective nucleophilic attack by the alcohol on one of the carbonyl groups of the 3-silyloxycyclopentanoic anhydride. This catalytic cycle operates through a dual-activation mechanism where the catalyst simultaneously activates the electrophilic anhydride and the nucleophilic alcohol via hydrogen bonding networks, lowering the activation energy for the desired enantiomer. The reaction proceeds efficiently at temperatures ranging from -78°C to 50°C, depending on the specific solvent system employed, such as toluene or methyl tert-butyl ether. The stereochemical information introduced in this first step is crucial as it dictates the configuration of the downstream intermediates, ensuring that the final product maintains the required optical purity for biological efficacy. Understanding this mechanistic nuance is vital for R&D directors evaluating the robustness of the process, as it demonstrates a deep control over molecular architecture that surpasses simple stoichiometric transformations. 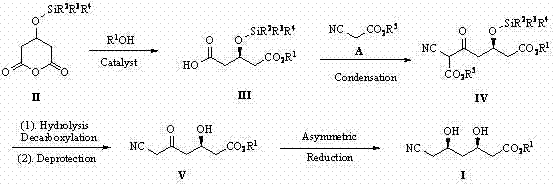
Following the initial chiral induction, the impurity profile is tightly managed through the selection of specific condensing agents and reducing systems. The condensation with cyanoacetate is performed under alkaline conditions using bases like triethylamine or DBU, which promote the formation of the beta-keto ester while minimizing side reactions such as self-condensation of the ester. Subsequent decarboxylation and desilylation are carefully orchestrated to remove protecting groups without racemizing the sensitive chiral centers. The final asymmetric reduction employs boron-based reagents like diethyl methoxyborane combined with sodium borohydride or lithium borohydride, which deliver hydride to the ketone face selectively. This multi-layered approach to impurity control ensures that by-products are kept to a minimum, simplifying the workup and isolation procedures. For technical teams, this level of detail confirms that the process is not merely a theoretical possibility but a practically viable pathway that has been optimized for reproducibility and purity specifications.
How to Synthesize (3R,5R)-3,5-Dihydroxy-6-cyanohexanoate Efficiently
The implementation of this synthesis requires precise adherence to the molar ratios and reaction times specified in the patent embodiments to achieve optimal results. The process is divided into four distinct stages, beginning with the catalytic alcoholysis which sets the foundation for the entire sequence. Operators must ensure that the chiral catalyst is properly activated and that moisture is excluded from the reaction vessel to prevent hydrolysis of the anhydride before the desired alcoholysis occurs. Following the isolation of the chiral monoester, the condensation step demands careful temperature control to manage the exotherm associated with the activation of cyanoacetate.
- Perform asymmetric alcoholysis of 3-silyloxycyclopentanoic anhydride using a chiral quinine thiourea catalyst to generate the chiral monoester.
- Conduct condensation with cyanoacetate under alkaline conditions to form the beta-keto ester intermediate with high stereocontrol.
- Execute decarboxylation and desilylation followed by asymmetric reduction using boron reagents to achieve the final diol configuration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for organizations focused on cost reduction in fine chemical manufacturing and supply chain resilience. The elimination of highly toxic cyanide salts removes a major regulatory hurdle, allowing for faster permitting and reduced insurance premiums associated with hazardous material storage. This shift significantly lowers the barrier to entry for scaling production, as facilities do not require the specialized infrastructure needed to handle acute toxins, thereby accelerating time-to-market for new drug filings. Furthermore, the quantitative recovery of the chiral catalyst represents a direct material cost saving, as expensive organocatalysts do not need to be purchased in stoichiometric quantities for every batch. This efficiency translates into a more stable pricing model for buyers, shielding them from volatility in the raw material market. For supply chain heads, the use of common organic solvents and readily available reagents like cyanoacetate ensures that production is not held hostage by the supply constraints of niche or controlled chemicals.
- Cost Reduction in Manufacturing: The process design inherently reduces operational expenditures by simplifying the waste treatment workflow, as the effluent does not contain lethal cyanide residues that require complex chemical destruction before discharge. The ability to run reactions under milder conditions also reduces energy consumption related to heating and cooling, contributing to a lower carbon footprint and utility costs. Additionally, the high yield reported in the patent embodiments minimizes the loss of valuable starting materials, maximizing the output per unit of input. These factors combine to create a leaner manufacturing process that delivers substantial cost savings without compromising on the quality of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: By relying on stable, non-controlled reagents, manufacturers can maintain continuous production schedules without the risk of regulatory interruptions often associated with cyanide procurement. The robustness of the catalytic system allows for flexible batch sizing, enabling suppliers to respond quickly to fluctuating demand from downstream drug manufacturers. This agility is critical in the pharmaceutical sector where just-in-time delivery is often required to support clinical trial timelines and commercial launches. A partner utilizing this technology can guarantee consistent availability of high-purity intermediates, reducing the risk of stockouts that could delay critical medication production.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scale-up in mind, utilizing unit operations that are standard in modern chemical plants such as filtration, extraction, and distillation. The environmental benefits extend beyond safety to include reduced solvent usage and improved atom economy, aligning with global sustainability goals and corporate social responsibility mandates. This compliance advantage makes the resulting product more attractive to environmentally conscious buyers and helps pharmaceutical companies meet their own green chemistry targets. Ultimately, the ease of scale-up ensures that the transition from pilot plant to multi-ton production is smooth and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the intellectual property documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply networks.
Q: How does this method improve safety compared to traditional cyanide routes?
A: This process eliminates the use of highly toxic alkali metal cyanides (KCN/NaCN) typically used for epoxide ring-opening, replacing them with safer cyanoacetate condensation, significantly reducing EHS risks and waste treatment costs.
Q: What is the stereoselectivity of the catalytic system?
A: The method utilizes chiral quinine-derived thiourea or sulfonamide catalysts which provide high stereoselectivity during the initial alcoholysis and subsequent asymmetric reduction steps, ensuring consistent optical purity.
Q: Is the chiral catalyst recoverable for industrial scale-up?
A: Yes, the patent specifies that the chiral catalyst is used in small amounts and can be quantitatively recovered, which drastically lowers material costs and supports sustainable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R,5R)-3,5-Dihydroxy-6-cyanohexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a supply chain built on scientific excellence and operational integrity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of (3R,5R)-3,5-dihydroxy-6-cyanohexanoate meets the highest international standards. Our infrastructure is designed to support the complex requirements of chiral synthesis, providing a secure environment for the manufacture of sensitive pharmaceutical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific application. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this safer, more efficient method. We are ready to provide specific COA data and route feasibility assessments to support your validation processes. Partner with us to leverage cutting-edge chemistry that drives value, safety, and performance in your pharmaceutical supply chain.
