Advanced Manufacturing of Bilastine Intermediate: A Novel Three-Step Catalytic Route for Global Supply
Advanced Manufacturing of Bilastine Intermediate: A Novel Three-Step Catalytic Route for Global Supply
The pharmaceutical industry constantly seeks robust, scalable, and safe synthetic routes for critical active pharmaceutical ingredient (API) precursors. A significant breakthrough in this domain is detailed in patent CN110734375B, which discloses a highly efficient preparation method for a key bilastine intermediate, specifically the compound of Formula I. Bilastine, a second-generation histamine H1 receptor antagonist, is widely utilized for treating allergic rhinoconjunctivitis and urticaria due to its favorable safety profile and lack of cardiotoxicity. The intermediate described herein serves as a pivotal building block in the assembly of this valuable therapeutic agent. This report analyzes the technical merits of this novel three-step sequence, highlighting its potential to redefine supply chain reliability for global manufacturers seeking a reliable bilastine intermediate supplier.
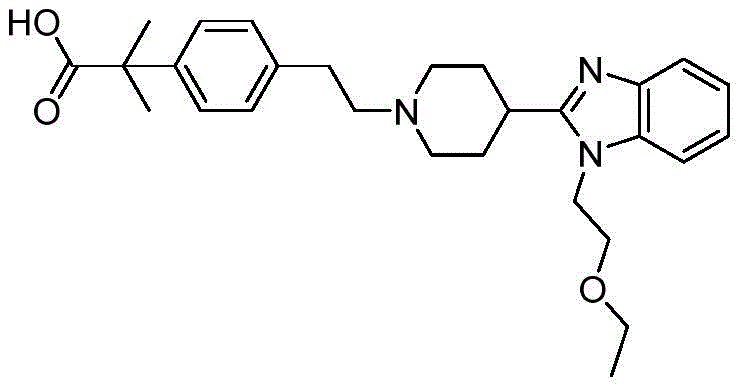
The core innovation lies in the strategic selection of starting materials and reaction conditions that prioritize operational safety and yield maximization. Unlike traditional pathways that often rely on hazardous oxidants or unstable organometallic species, this method leverages a streamlined sequence starting from 2-bromoethyl anisole. The process involves an initial elimination reaction to generate a vinyl ether, followed by a palladium-catalyzed cross-coupling, and concludes with a selective hydrogenation. This logical progression not only simplifies the synthetic tree but also aligns with modern green chemistry principles by reducing waste generation and avoiding extreme reaction parameters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 4-(2-hydroxyethyl)-phenyl tert-butyl methyl ester have been plagued by significant operational hazards and inefficiencies that hinder large-scale adoption. For instance, the route reported by Collier et al. utilizes borane dimethyl sulfide for reduction, a reagent notorious for its pungent odor and potential for air pollution, alongside hydrogen peroxide oxidation which introduces inherent safety risks due to its explosive nature. Furthermore, other patented approaches, such as those disclosed in WO2009/102155 and US2011/9636, necessitate low-temperature reactions that are energy-intensive and difficult to control in bulk manufacturing. These legacy methods often produce intermediates that are hypersensitive to moisture, leading to hydrolysis issues that compromise product stability and storage, thereby creating bottlenecks in the supply chain for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
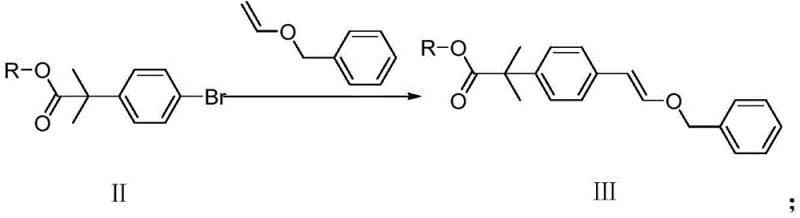
In stark contrast, the methodology outlined in CN110734375B offers a paradigm shift towards safer and more manageable chemistry. By initiating the synthesis with 2-bromoethyl anisole (Formula IV), the process bypasses the need for dangerous oxidants entirely. The first step employs DBU (1,8-diazabicycloundec-7-ene) to effect a clean elimination, generating the vinyl anisole (Formula V) in high yield. Subsequent coupling with the aryl bromide (Formula II) is achieved under relatively mild thermal conditions (90-110°C) using standard palladium catalysts. This approach eliminates the moisture sensitivity issues associated with prior organometallic routes, ensuring that the intermediate remains stable throughout the workflow. The final hydrogenation step is conducted at ambient temperature and pressure, further underscoring the operational simplicity and safety profile of this novel pathway.
Mechanistic Insights into Pd-Catalyzed Coupling and Hydrogenation
The heart of this synthetic strategy is the palladium-catalyzed coupling reaction between the vinyl ether (Formula V) and the aryl bromide (Formula II). This transformation likely proceeds via a mechanism analogous to the Heck reaction or a specialized Pd-catalyzed vinylation, where the palladium center facilitates the formation of a carbon-carbon bond between the aromatic ring and the vinyl group. The choice of ligand and catalyst precursor, such as tetrakis(triphenylphosphine)palladium(0) or palladium acetate, is critical for maintaining catalytic turnover and minimizing side reactions like homocoupling. The presence of a base, such as potassium carbonate or triethylamine, serves to neutralize the hydrogen bromide byproduct, driving the equilibrium towards the desired product (Formula III). This step is pivotal as it constructs the core carbon skeleton required for the final API structure.
Following the coupling, the final step involves the catalytic hydrogenation of the double bond in Formula III to yield the saturated ethyl chain in Formula I. This reduction is typically performed using heterogeneous catalysts like palladium on carbon (Pd/C) or palladium hydroxide. The mechanism involves the adsorption of hydrogen gas and the substrate onto the metal surface, followed by syn-addition of hydrogen across the alkene. Crucially, the conditions are mild enough (15-30°C) to prevent the hydrogenolysis of the benzyl ether protecting group or the ester functionality, ensuring high chemoselectivity. This precise control over reactivity is essential for maintaining the high-purity bilastine intermediate specifications required by regulatory bodies, effectively minimizing the formation of de-protected or over-reduced impurities.
How to Synthesize 4-(2-hydroxyethyl)-phenyl tert-butyl methyl ester Efficiently
The execution of this synthesis requires careful attention to solvent selection and stoichiometry to maximize efficiency. The process begins with the elimination of 2-bromoethyl anisole in a polar aprotic solvent like tetrahydrofuran (THF) or DMF, heated to facilitate the removal of HBr. The resulting vinyl ether is then coupled with the aryl bromide in a solvent system such as 1,4-dioxane, which supports the solubility of both organic partners and the inorganic base. Finally, the crude coupling product is subjected to hydrogenation in an alcoholic solvent like methanol or ethanol. Detailed standardized operating procedures for each stage, including specific work-up protocols to isolate the pure product, are essential for reproducibility.
- React 2-bromoethyl anisole (Formula IV) with DBU in a solvent like THF at 60-75°C to form vinyl anisole (Formula V).
- Couple Formula V with methyl 3-bromophenyl tert-butyrate (Formula II) using a Pd catalyst and base in 1,4-dioxane at 90-110°C to yield Formula III.
- Hydrogenate Formula III using a second Pd catalyst (e.g., Pd/C) in methanol or ethanol at 15-30°C to obtain the final intermediate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of hazardous reagents like borane complexes and concentrated hydrogen peroxide significantly reduces the regulatory burden and safety infrastructure costs associated with manufacturing. This simplification of the hazard profile allows for production in a wider range of facilities, enhancing supply chain resilience. Moreover, the use of commodity chemicals such as 2-bromoethyl anisole and common palladium catalysts ensures that raw material sourcing remains stable and less susceptible to geopolitical fluctuations, directly supporting commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The new process drastically lowers operational expenditures by removing the need for cryogenic cooling systems required by prior low-temperature methods. The ability to run reactions at moderate temperatures (60-110°C) reduces energy consumption significantly. Furthermore, the high yields reported in the patent examples (e.g., 97% for step 1, 96.7% for step 2) imply a substantial reduction in raw material waste and downstream purification costs. By avoiding expensive and dangerous reagents, the overall cost of goods sold (COGS) is optimized without compromising quality.
- Enhanced Supply Chain Reliability: Stability is a key factor in supply chain management. The intermediates generated in this route, particularly the vinyl ether and the coupled product, do not exhibit the extreme moisture sensitivity seen in alternative Grignard or organolithium-based routes. This robustness allows for longer storage times and more flexible logistics planning, reducing lead time for high-purity bilastine intermediates. Suppliers can maintain strategic stockpiles without the risk of rapid degradation, ensuring continuous availability for downstream API manufacturers even during market disruptions.
- Scalability and Environmental Compliance: From an environmental perspective, this route generates fewer hazardous wastes, aligning with increasingly stringent global environmental regulations. The solvents used, such as THF, dioxane, and alcohols, are well-understood and can be efficiently recovered and recycled in a closed-loop system. The absence of heavy metal contaminants from exotic reagents simplifies the purification process, reducing the load on wastewater treatment facilities. This eco-friendly profile not only mitigates compliance risks but also enhances the brand value of the final pharmaceutical product by adhering to green chemistry standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical implementation and advantages of this patent-protected method. These insights are derived directly from the experimental data and claims presented in the intellectual property documentation, providing a factual basis for decision-making.
Q: What are the primary safety advantages of this new synthesis route compared to prior art?
A: The novel route eliminates the use of hazardous reagents such as borane dimethyl sulfide (which is malodorous and polluting) and hydrogen peroxide (which poses explosion risks) found in previous methods like the Collier route. Instead, it utilizes mild elimination and catalytic hydrogenation steps.
Q: Which palladium catalysts are suitable for the coupling step in this process?
A: The patent specifies a range of effective catalysts including palladium on carbon, tetrakis(triphenylphosphine)palladium(0), palladium acetate, and bis(dibenzylideneacetone)palladium, offering flexibility for process optimization.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial scalability. It operates under mild temperatures (60-110°C), uses common solvents like THF and dioxane, and avoids moisture-sensitive intermediates that complicate storage and handling in large reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology described in CN110734375B and possesses the technical capability to implement this three-step process at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of bilastine intermediate delivered meets the highest international standards.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
