Scalable Production of Bilastine Key Intermediate via Novel Nickel-Catalyzed Route
Scalable Production of Bilastine Key Intermediate via Novel Nickel-Catalyzed Route
The pharmaceutical industry continuously seeks robust and safe manufacturing processes for high-volume active pharmaceutical ingredients (APIs), and Bilastine stands out as a critical second-generation antihistamine. The synthesis of Bilastine presents significant challenges, primarily centered around the efficient construction of the quaternary carbon structure found in its key intermediate, 2-(4-(2-substituted ethyl) phenyl)-2-methylpropionic acid. Patent CN112552157B introduces a groundbreaking preparation method that addresses these historical bottlenecks by utilizing a nickel-catalyzed coupling strategy. This innovation not only streamlines the synthetic pathway but also drastically mitigates the safety risks associated with traditional reagents like ethylene oxide. For global supply chains, this represents a pivotal shift towards more sustainable and reliable sourcing of high-purity pharmaceutical intermediates.
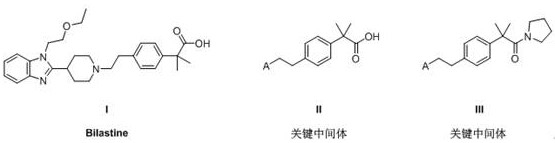
The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for the final drug substance. By optimizing the construction of this core structure, manufacturers can achieve better control over impurity profiles and overall yield. The patent details a sequence that begins with commercially available boronic acids and proceeds through a series of high-efficiency transformations. This approach aligns perfectly with the needs of R&D directors who require reproducible chemistry and procurement managers who demand cost-effective raw material strategies. The elimination of hazardous steps ensures that the process is not only chemically elegant but also operationally superior for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
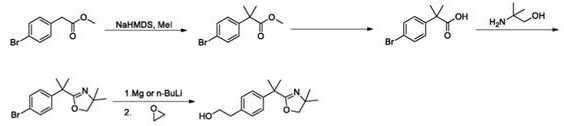
Historically, the synthesis of the Bilastine intermediate has been plagued by severe safety and efficiency drawbacks. The original route reported by FAES Company, for instance, relies heavily on the use of ethylene oxide to extend the carbon chain. While chemically effective, ethylene oxide is a known carcinogen and poses extreme explosion hazards, requiring specialized equipment and rigorous safety protocols that drive up capital and operational expenditures. Furthermore, alternative academic routes have explored the use of organotin reagents and noble metal catalysts, which introduce significant environmental toxicity and high material costs. These conventional methods often suffer from poor atom economy and generate substantial hazardous waste, complicating the purification process and limiting their viability for large-scale commercial production.
The Novel Approach
In stark contrast, the method disclosed in CN112552157B circumvents these issues by employing a nickel-catalyzed cross-coupling reaction to install the ethyl side chain. This strategy replaces the dangerous ethylene oxide expansion with a safer alkylation of a boronic acid derivative. The subsequent steps utilize a Corey-Fuchs type transformation followed by a methylation sequence to construct the critical quaternary carbon center. This route is characterized by milder reaction conditions, avoiding the cryogenic temperatures and pyrophoric reagents often required for lithiation-based approaches. By shifting to base-metal catalysis and safer alkylating agents, the new method offers a pathway that is inherently more stable, easier to control, and significantly less hazardous to personnel and the environment.
Mechanistic Insights into Nickel-Catalyzed Coupling and Quaternary Carbon Construction
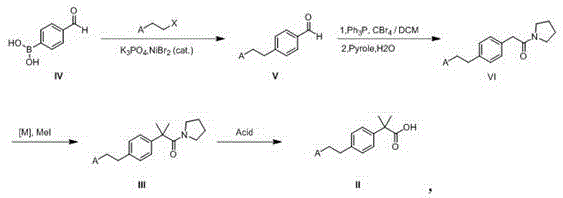
The heart of this innovative synthesis lies in the initial coupling step, where a nickel catalyst facilitates the bond formation between the aromatic ring and the alkyl chain. Unlike palladium, which is prone to beta-hydride elimination with alkyl halides, nickel catalysts are uniquely suited for sp3-sp2 cross-couplings, allowing for the efficient attachment of the substituted ethyl group. The mechanism likely involves the oxidative addition of the alkyl halide to the nickel center, followed by transmetallation with the boronic acid species and reductive elimination to release the coupled product. This step is crucial as it sets the stage for the rest of the synthesis, ensuring that the side chain is installed with high regioselectivity and minimal formation of homocoupling byproducts.
Following the coupling, the construction of the quaternary carbon is achieved through a sequential methylation strategy. The intermediate aldehyde is first converted into a reactive species, potentially a dibromoalkene or an activated amide, which then undergoes deprotonation and alkylation with methyl iodide. This step effectively installs the two methyl groups on the alpha-carbon, creating the sterically hindered quaternary center that defines the Bilastine structure. The use of strong bases like sodium hydroxide or sodium hydride in polar aprotic solvents ensures complete enolization and subsequent methylation. This mechanistic pathway is robust and minimizes the formation of mono-methylated impurities, thereby simplifying the downstream purification and enhancing the overall purity of the final acid intermediate.
How to Synthesize 2-(4-(2-substituted ethyl) phenyl)-2-methylpropionic acid Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly in the coupling and methylation stages. The process begins with the preparation of the coupled aldehyde, followed by transformation into the amide precursor. The critical methylation step must be monitored closely to ensure double alkylation occurs without degrading the sensitive functional groups. Finally, the hydrolysis step releases the free carboxylic acid, which precipitates upon acidification, allowing for easy isolation. The detailed standardized synthetic steps for this process are outlined below to guide technical teams in replicating this high-efficiency route.
- Perform a nickel-catalyzed coupling reaction between 4-formylphenylboronic acid and a halogenated alkane to form the aldehyde intermediate.
- Convert the aldehyde to a dibromoalkene using triphenylphosphine and carbon tetrabromide, followed by treatment with tetrahydropyrrole.
- Construct the quaternary carbon center by reacting the intermediate with a metal reagent and methyl iodide.
- Hydrolyze the final ester or amide precursor in an acidic solution to obtain the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic reduction of operational risk. By eliminating ethylene oxide and organotin reagents, facilities can lower their insurance premiums, reduce the need for specialized containment infrastructure, and minimize the regulatory burden associated with handling extremely hazardous substances. This translates directly into a more resilient supply chain that is less susceptible to shutdowns caused by safety incidents or regulatory compliance issues. Furthermore, the use of nickel instead of palladium represents a significant cost saving, as nickel is an earth-abundant metal with a fraction of the price volatility of precious metals.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with nickel systems fundamentally alters the cost structure of the intermediate. Additionally, the avoidance of cryogenic conditions reduces energy consumption for cooling, while the simplified workup procedures decrease solvent usage and waste disposal costs. The high yields reported in the patent examples suggest that raw material utilization is optimized, further driving down the cost per kilogram of the final product. These factors combine to create a highly competitive cost profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as boronic acids, alkyl halides, and common bases ensures that the supply chain is not dependent on niche or single-source suppliers. This diversification of raw material sources mitigates the risk of shortages and price spikes. Moreover, the robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with greater ease, ensuring continuity of supply even if one facility faces disruptions. This reliability is paramount for pharmaceutical companies managing long-term contracts for API production.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing solvents and reagents that are manageable in large reactors. The absence of highly toxic byproducts simplifies effluent treatment, making it easier to meet stringent environmental regulations. The ability to run reactions at moderate temperatures and pressures reduces the engineering complexity required for scale-up, allowing for faster technology transfer from the lab to the pilot plant and eventually to commercial production. This scalability ensures that the method can meet the growing global demand for Bilastine without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility of the route for potential partners and licensees. Understanding these details is essential for evaluating the fit of this technology within existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional Bilastine synthesis?
A: Traditional routes often rely on ethylene oxide, which is highly toxic, explosive, and difficult to handle industrially. This novel method eliminates the need for ethylene oxide and avoids toxic organotin reagents, significantly reducing operational risks and environmental hazards.
Q: What are the cost advantages of using nickel catalysis over palladium in this process?
A: Nickel catalysts are substantially less expensive than noble metal catalysts like palladium. Additionally, the use of earth-abundant nickel reduces the burden of heavy metal removal and recovery, leading to lower overall production costs and simplified downstream processing.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It utilizes robust reaction conditions, avoids cryogenic temperatures required by some alternative lithiation methods, and employs readily available raw materials, making it highly viable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain. Our team of expert chemists has extensively evaluated the route described in CN112552157B and confirmed its potential for delivering high-purity Bilastine intermediates at a commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch conforms to the highest industry standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for Bilastine production.
