Scalable Synthesis of Bilastine Intermediate via Safe Vinyl Sulfate Chemistry for Global API Manufacturing
Introduction to Advanced Bilastine Intermediate Synthesis
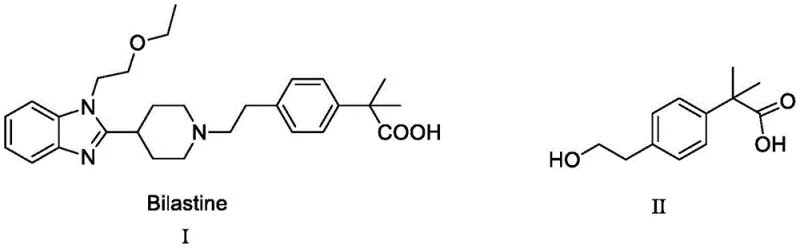
The global demand for second-generation antihistamines continues to drive innovation in the manufacturing of key pharmaceutical building blocks. Patent CN110803985A introduces a groundbreaking preparation method for Compound II, a critical quaternary carbon-containing intermediate essential for the synthesis of Bilastine. This technical disclosure addresses long-standing challenges in the industry, specifically focusing on the construction of the sterically hindered quaternary center adjacent to the carboxylic acid functionality. By leveraging the unique reactivity of vinyl sulfate, this novel process circumvents the use of hazardous gaseous reagents and expensive transition metal catalysts that have historically plagued the supply chain. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more cost-effective, and environmentally compliant manufacturing protocols for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
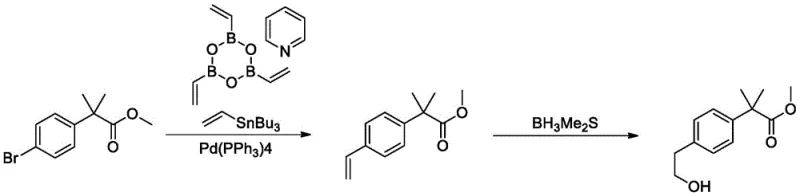
Historically, the synthesis of the Bilastine core structure has been fraught with significant technical and safety hurdles. Early routes, such as those reported by FAES, relied heavily on the use of ethylene oxide to extend the carbon chain. While chemically feasible, ethylene oxide is a carcinogenic, explosive gas that requires specialized high-pressure equipment and rigorous safety protocols, making it ill-suited for large-scale production in standard multipurpose plants. Furthermore, alternative strategies employing noble metal catalysis, such as the palladium-catalyzed coupling of silyl enol ethers described in WO2009/102155A2, introduce prohibitive costs due to the price of palladium and the difficulty of removing trace metal residues to meet pharmaceutical purity standards. Other academic approaches, like the Still coupling followed by hydroboration shown in literature, utilize organotin reagents which are notoriously toxic and difficult to separate from the final product, creating massive environmental liabilities and purification bottlenecks.
The Novel Approach
The methodology disclosed in CN110803985A offers a transformative solution by replacing these hazardous and expensive reagents with solid vinyl sulfate. This approach fundamentally redesigns the synthetic logic: instead of building the chain onto a pre-formed acid or ester using dangerous gases, it constructs the quaternary carbon and the side chain simultaneously through a nucleophilic attack on a solid electrophile. This not only eliminates the need for high-pressure reactors but also streamlines the downstream processing. The route is designed to be telescoped, meaning the intermediate formed after the alkylation step does not require isolation before the final deprotection. This consolidation of steps significantly reduces solvent consumption, labor hours, and overall production time, offering a robust alternative for reliable API intermediate suppliers seeking to optimize their manufacturing footprint.
Mechanistic Insights into Lithium-Halogen Exchange and Vinyl Sulfate Alkylation
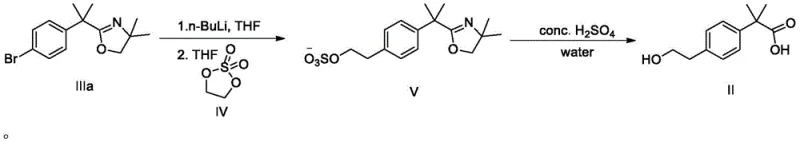
The core of this innovation lies in the precise control of organolithium chemistry combined with the specific reactivity of cyclic vinyl sulfates. The process initiates with a lithium-halogen exchange on a halogenated precursor (Compound III, where X is Br, Cl, or I). This step is highly sensitive and must be conducted at cryogenic temperatures, specifically between -80°C and -50°C, using reagents like n-butyllithium or sec-butyllithium in solvents such as tetrahydrofuran (THF) or methyl tert-butyl ether (MTBE). Maintaining this low temperature window is critical to suppress competing side reactions, such as nucleophilic attack on the oxazoline protecting group or Wurtz-type coupling, ensuring the formation of the desired aryl-lithium species with high fidelity. Once generated, this highly reactive nucleophile attacks the vinyl sulfate (Compound IV) in situ.
The subsequent transformation involves a cascade of events triggered by acid treatment. The intermediate Compound V contains both an oxazoline ring (acting as a masked carboxylic acid) and a sulfate ester. Upon exposure to strong mineral acids like concentrated sulfuric acid or hydrochloric acid at elevated temperatures (60°C to 100°C), a dual deprotection occurs. The acidic conditions hydrolyze the oxazoline ring to reveal the free carboxylic acid while simultaneously facilitating the elimination of the sulfate group to generate the terminal hydroxyl functionality. This "two-in-one" deprotection strategy is mechanistically elegant, as it avoids the need for separate hydrolysis and reduction steps, thereby minimizing impurity formation and maximizing the overall yield of the target quaternary carbon structure.
How to Synthesize Bilastine Intermediate Efficiently
Implementing this synthesis requires strict adherence to the thermal profiles and stoichiometry outlined in the patent examples to ensure reproducibility and safety. The process is amenable to batch processing in standard glass-lined or stainless steel reactors equipped with efficient cooling capabilities for the cryogenic steps. Operators must ensure that the vinyl sulfate is added slowly to the cold lithiated mixture to manage the exotherm and prevent thermal runaway. Following the alkylation, the reaction mixture can be warmed to room temperature before quenching, after which the aqueous phase containing the sulfate intermediate is directly subjected to acid hydrolysis without intermediate isolation. For a detailed, step-by-step breakdown of the reagent quantities, specific addition rates, and workup procedures validated at the kilogram scale, please refer to the standardized protocol below.
- Perform lithium-halogen exchange on compound III (where X is Br, Cl, or I) using n-butyllithium or sec-butyllithium at temperatures between -80°C and -50°C in solvents like THF or MTBE.
- React the resulting phenyllithium species in situ with solid vinyl sulfate (Compound IV) at temperatures ranging from -80°C to -20°C to construct the quaternary carbon and introduce the ethyl sulfate moiety.
- Treat the intermediate Compound V with strong acid (e.g., concentrated sulfuric acid or hydrochloric acid) at 60°C to 100°C to simultaneously hydrolyze the oxazoline ring and eliminate the sulfate group, yielding the final carboxylic acid alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary driver for cost reduction in pharmaceutical manufacturing here is the complete elimination of noble metal catalysts. By avoiding palladium, manufacturers remove the need for expensive scavenging resins and complex analytical testing for residual metals, which are significant cost centers in API production. Additionally, the substitution of gaseous ethylene oxide with solid vinyl sulfate drastically lowers the barrier to entry for production facilities, as it removes the requirement for specialized high-pressure containment systems and reduces insurance premiums associated with handling explosive gases. This shift translates to substantial capital expenditure savings and lower operating costs per kilogram of produced intermediate.
Enhanced supply chain reliability is another critical benefit derived from this chemistry. The raw materials required—halogenated aromatics, butyllithium, and vinyl sulfate—are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Unlike organotin reagents which face increasing regulatory scrutiny and supply constraints due to their toxicity, vinyl sulfate is a stable, manageable solid that simplifies logistics and warehousing. This stability ensures consistent lead times and reduces the risk of production stoppages caused by hazardous material shipping delays. Furthermore, the simplified purification process, which relies on standard acid-base extractions rather than chromatography or distillation of sensitive organometallics, ensures a more predictable and continuous output suitable for just-in-time manufacturing models.
Scalability and environmental compliance are inherently built into this route, making it ideal for commercial scale-up of complex pharmaceutical intermediates. The absence of heavy metals like tin and palladium means that waste streams are significantly less hazardous, lowering the cost of waste disposal and easing the burden on environmental health and safety (EHS) departments. The process generates fewer byproducts and utilizes common solvents like MTBE and ethyl acetate, which are easily recovered and recycled. This green chemistry profile not only aligns with modern sustainability goals but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term viability and uninterrupted supply for downstream API manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational parameters and strategic benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the integration of this method into existing production lines.
Q: Why is vinyl sulfate preferred over ethylene oxide for constructing the quaternary carbon in Bilastine intermediates?
A: Vinyl sulfate is a solid reagent with significantly higher operational safety compared to ethylene oxide, which is a toxic, explosive gas. Furthermore, the vinyl sulfate route allows for a one-pot deprotection strategy that simplifies the workflow.
Q: How does this novel route address the environmental concerns of previous methods like the Still coupling?
A: Unlike the Still coupling route which utilizes highly toxic organotin reagents and borane dimethylsulfide, this method avoids heavy metals and toxic volatile reagents entirely, drastically reducing hazardous waste generation and purification complexity.
Q: What are the critical temperature controls required for the lithium-halogen exchange step?
A: To prevent side reactions and ensure high selectivity during the formation of the phenyllithium species, the reaction temperature must be strictly maintained between -80°C and -50°C before the addition of the vinyl sulfate electrophile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust intermediate supply chains for the global antihistamine market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with the necessary cryogenic capabilities and corrosion-resistant reactors required to execute the lithium-halogen exchange and acid hydrolysis steps safely. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Bilastine intermediate meets the highest quality standards, free from the heavy metal impurities associated with older synthetic routes.
We invite procurement leaders and R&D teams to collaborate with us to leverage this advanced technology for your supply needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this vinyl sulfate route can optimize your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and secure a reliable supply of high-purity intermediates for your next commercial campaign.
