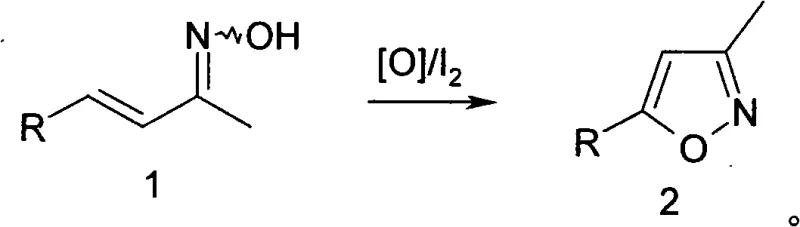
Advanced Iodine-Catalyzed Oxidation Strategy for Scalable Isoxazole Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for constructing heterocyclic scaffolds, particularly isoxazoles, which serve as critical building blocks in numerous active pharmaceutical ingredients. A pivotal advancement in this domain is disclosed in patent CN101274915A, which outlines a highly efficient method for synthesizing isoxazole derivatives through an oxidative cyclization strategy. This technology represents a significant departure from legacy protocols by replacing costly stoichiometric reagents with a catalytic system driven by inexpensive iodine and common inorganic oxidants. The process utilizes alpha,beta-unsaturated oximes as the primary starting materials, subjecting them to mild oxidative conditions that facilitate ring closure with exceptional selectivity. By operating at temperatures between 10°C and 100°C and employing readily available bases such as sodium bicarbonate, this method addresses the longstanding economic and operational bottlenecks associated with heterocycle production. For procurement managers and R&D directors alike, this patent offers a compelling blueprint for cost reduction in fine chemical manufacturing, transforming a traditionally expensive transformation into a commodity-scale operation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoxazole rings has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional routes often rely on the condensation of 1,3-dicarbonyl compounds with hydroxylamine, a process fraught with selectivity issues that frequently result in complex mixtures requiring arduous purification steps to isolate the desired single product. Alternative pathways involving alpha-acetylenic ketones or aldehydes suffer from the inherent instability and poor availability of these precursors, making them unsuitable for reliable industrial supply chains. Furthermore, methods utilizing halogenated alpha,beta-unsaturated carbonyl compounds are similarly constrained by the difficulty in sourcing raw materials and generally low overall yields. Perhaps most critically, prior art methods that employ IKI (iodine-potassium iodide) complexes for oxidation require massive excesses of reagents, typically 3 to 4 equivalents, alongside large volumes of organic solvents like tetrahydrofuran. This stoichiometric dependence not only drives up raw material costs exponentially but also generates substantial waste streams, creating environmental compliance challenges and inflating the total cost of ownership for the final API intermediate.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in CN101274915A introduces a streamlined catalytic cycle that fundamentally alters the economics of isoxazole production. By shifting from stoichiometric IKI complexes to a catalytic system utilizing merely 1% to 10% by weight of iodine, the process drastically reduces the consumption of expensive halogen sources. This novel approach leverages cheap, abundant inorganic oxidants such as hydrogen peroxide, sodium hypochlorite, or persulfates to drive the oxidative cyclization of alpha,beta-unsaturated oximes directly to the isoxazole core. The reaction conditions are remarkably mild, typically proceeding between 40°C and 80°C, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive functional groups. Moreover, the versatility of the solvent system allows for operation in either homogeneous or heterogeneous phases, providing process engineers with the flexibility to optimize workup procedures based on specific substrate properties. This direct conversion eliminates the need for isolating unstable epoxy intermediates, thereby simplifying the workflow and enhancing the overall throughput of the manufacturing line.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the efficient activation of the alpha,beta-unsaturated oxime substrate through a catalytic iodine-mediated oxidation mechanism. In this system, iodine acts not as a consumable reagent but as a redox mediator that facilitates the transfer of oxygen from the inorganic oxidant to the organic substrate. The presence of an inorganic base, such as sodium bicarbonate or potassium carbonate, is crucial for deprotonating the oxime hydroxyl group, thereby increasing its nucleophilicity and preparing it for the subsequent cyclization event. As the temperature is gradually increased to the optimal range of 10°C to 100°C, the oxidant—whether it be hydrogen peroxide, chlorine dioxide, or a metal oxide like manganese dioxide—regenerates the active iodine species, sustaining the catalytic cycle without the need for continuous replenishment of the halogen source. This mechanistic efficiency ensures that the reaction proceeds with high atom economy, minimizing the formation of halogenated byproducts that often complicate downstream purification in traditional halogenation-cyclization sequences.
Furthermore, the control of impurities in this process is inherently superior due to the specificity of the oxidative cyclization. Unlike multi-step routes that generate unstable epoxy intermediates prone to decomposition, this one-pot transformation drives the reaction directly to the thermodynamically stable isoxazole ring. The use of mild oxidants prevents over-oxidation of sensitive substituents on the R-group, which can range from simple C1-C6 alkyl chains to complex aryl and heteroaryl moieties like furyl or methoxyphenyl groups. The reaction mixture, upon completion, is treated with an inorganic reducing agent such as sodium bisulfite to quench any residual oxidant, ensuring a clean organic layer ready for extraction. This precise control over the reaction environment results in a crude product profile that is amenable to simple vacuum fractionation, yielding materials with purity exceeding 95% without the need for resource-intensive chromatography. For R&D teams, this implies a significantly reduced impurity burden, facilitating faster regulatory filing and more robust quality control protocols.
How to Synthesize Isoxazole Efficiently
Implementing this synthesis route in a pilot or production setting requires careful attention to the phase behavior of the reaction mixture and the sequential addition of reagents to maintain catalytic efficiency. The process begins by dissolving the alpha,beta-unsaturated oxime precursor in a suitable organic solvent, with the patent specifying a wide range of options including ethanol, acetone, THF, or toluene, depending on whether a homogeneous or heterogeneous system is desired. Water is then introduced to the system, followed by the base and the catalytic iodine load. If a heterogeneous system is chosen to facilitate product separation, a phase transfer catalyst such as tetrabutylammonium bromide is added to ensure effective mass transfer between the aqueous and organic phases. The detailed standardized synthesis steps, including specific molar ratios, temperature ramping profiles, and quenching procedures, are outlined in the guide below to ensure reproducible high-yield outcomes.
- Dissolve alpha,beta-unsaturated oxime in an organic solvent (1-20x weight) and add water (0.25-4x volume) to form a homogeneous or heterogeneous system.
- Add inorganic base (0.3-2 mol ratio) and catalytic iodine (1-10% weight), optionally adding a phase transfer catalyst for heterogeneous systems.
- Heat to 10-100°C, add oxidant (e.g., hydrogen peroxide), react for 8-20 hours, quench excess oxidant, extract, and purify via vacuum fractionation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed methodology translates into tangible strategic advantages that extend far beyond simple chemistry. The most immediate impact is seen in the raw material cost structure, where the replacement of stoichiometric IKI complexes with catalytic iodine and commodity oxidants leads to a substantial reduction in bill of materials expenses. Since iodine is used in minute quantities and regenerated in situ, the dependency on volatile halogen markets is minimized, stabilizing the cost basis for long-term production contracts. Additionally, the ability to use a diverse array of inexpensive oxidants, including hydrogen peroxide and persulfates, provides procurement teams with the flexibility to source chemicals based on regional availability and spot pricing, further insulating the supply chain from market fluctuations. This flexibility is critical for maintaining margin integrity in the competitive landscape of generic pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the simplification of the workup process contribute to a leaner manufacturing cost model. By avoiding the use of 3 to 4 equivalents of IKI complex, the process significantly lowers the chemical waste disposal costs associated with halogen-rich effluent streams. Furthermore, the ability to purify the final product via simple vacuum distillation rather than complex chromatography reduces solvent consumption and processing time, leading to lower utility and labor costs per kilogram of output. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity specifications required by downstream API manufacturers.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as alpha,beta-unsaturated carbonyl compounds and hydroxylamine, ensures a robust and continuous supply of precursors. Unlike specialized reagents that may have single-source suppliers or long lead times, the inputs for this process are commodity chemicals produced globally in vast quantities. This ubiquity reduces the risk of supply disruptions and allows for the maintenance of strategic inventory buffers with minimal capital tie-up. Moreover, the mild reaction conditions reduce the stress on reactor equipment, extending asset life and minimizing unplanned downtime due to maintenance, thereby guaranteeing consistent delivery schedules to customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative to traditional methods by reducing the load of hazardous halogenated waste. The use of water as a co-solvent and the generation of benign inorganic salts as byproducts simplify wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations. The scalability of the reaction is proven by its tolerance to a wide range of substrates and its operation under atmospheric pressure and moderate temperatures, making it suitable for scale-up from laboratory benchtop to multi-ton commercial production without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of this iodine-catalyzed method over traditional IKI complex oxidation?
A: The primary advantage is the drastic reduction in reagent cost. Traditional methods require stoichiometric amounts (3-4 equivalents) of expensive IKI complexes, whereas this novel method utilizes only catalytic amounts (1-10% by weight) of inexpensive iodine alongside cheap inorganic oxidants like hydrogen peroxide or persulfates.
Q: Can this synthesis method be adapted for both homogeneous and heterogeneous reaction systems?
A: Yes, the process is highly versatile. It can operate in homogeneous phases using water-miscible solvents like THF or ethanol, or in heterogeneous phases using immiscible solvents like toluene or ethyl acetate, provided a phase transfer catalyst such as tetrabutylammonium bromide is employed.
Q: What is the typical purity and yield range achievable with this oxidative cyclization route?
A: According to the patent data, the method consistently achieves yields ranging from 60% to over 90% depending on the substrate. The resulting isoxazole products typically exhibit purity levels exceeding 95% after simple vacuum fractionation, eliminating the need for complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in the lab are faithfully reproduced on an industrial scale. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of isoxazole intermediate meets the exacting standards of the global pharmaceutical industry. Our commitment to quality assurance means that we can reliably supply high-purity intermediates that facilitate smoother regulatory approvals for your final drug products.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this catalytic route for your specific molecule. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the performance of our materials against your internal benchmarks before committing to larger volumes. Let us collaborate to optimize your supply chain and drive innovation in your drug development pipeline.
