Advanced Purification Technology for Pharmaceutical Grade Vitamin H Intermediates
Advanced Purification Technology for Pharmaceutical Grade Vitamin H Intermediates
The global demand for high-purity vitamins, particularly Vitamin H (D-Biotin), continues to surge across the pharmaceutical and nutraceutical sectors. However, achieving pharmaceutical-grade compliance has historically been hindered by persistent impurity profiles in synthetic intermediates. Patent CN113121558A introduces a transformative purification methodology specifically targeting the critical intermediate, Compound 2, in the Vitamin H synthesis pathway. This innovation addresses the long-standing bottleneck where crude intermediates typically possess purities ranging from 80% to 95%, inevitably forcing manufacturers to accept feed-grade final products or incur prohibitive costs on final refining. By implementing a rigorous acid-base extraction coupled with controlled anti-solvent precipitation, this technology enables the production of Intermediate 2 with purities exceeding 99%. For R&D directors and supply chain leaders, this represents a pivotal shift towards more efficient, compliant, and cost-effective manufacturing of essential vitamins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Vitamin H generally involve the hydrogenation of a urea-containing precursor (Compound 1) followed by debenzylation and cyclization. As illustrated in the reaction scheme below, the conventional pathway often suffers from incomplete reactions and the formation of stubborn by-products that co-elute with the desired intermediate. When crude Compound 2 is carried forward without rigorous purification, these impurities propagate through the subsequent HBr debenzylation and triphosgene cyclization steps. The result is a final Vitamin H product that fails to meet the stringent impurity thresholds of the European Pharmacopoeia, specifically regarding single impurities which often exceed 0.1%. Consequently, manufacturers are forced to implement complex, low-yield recrystallization processes on the final API, drastically reducing overall throughput and increasing solvent waste.
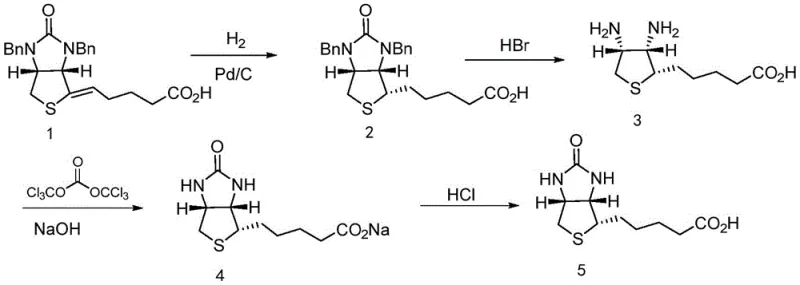
The Novel Approach
The patented method fundamentally alters the purification landscape by intervening at the Intermediate 2 stage rather than attempting to salvage the final product. Instead of relying solely on chromatography or simple recrystallization, the process employs a chemically selective workup. The crude material is first dissolved in an alkaline solution, converting the carboxylic acid moiety into a water-soluble salt while leaving neutral organic impurities behind or allowing for their separation. Subsequent acidification regenerates the free acid, which is then extracted into an organic phase. The true breakthrough lies in the final precipitation step using n-heptane as an anti-solvent. By carefully controlling the temperature and solvent ratios, the pure Compound 2 crystallizes out while remaining impurities stay in solution. This approach not only simplifies the operational workflow but also ensures that the starting material for the final cyclization is of exceptional quality, thereby guaranteeing a pharmaceutical-grade end product.
Mechanistic Insights into Acid-Base Extraction and Precipitation
The efficacy of this purification strategy relies on the precise manipulation of solubility parameters driven by pH changes and solvent polarity. In the initial alkaline treatment, typically using sodium hydroxide at a concentration of 1-3 mol/L, the carboxylic acid group of Compound 2 is deprotonated. This ionic form exhibits high affinity for the aqueous phase, effectively partitioning the target molecule away from non-polar organic contaminants that may have originated from the hydrogenation step. The patent specifies maintaining a pH between 10 and 14 to ensure complete salt formation. Following this, the addition of strong acid, such as 4-6 mol/L HCl, lowers the pH to between 1 and 3. This reprotonation converts the salt back into its neutral, less water-soluble acid form, facilitating its transfer into the ethyl acetate organic layer during extraction. This liquid-liquid extraction acts as a primary filter, removing inorganic salts and highly polar degradation products.
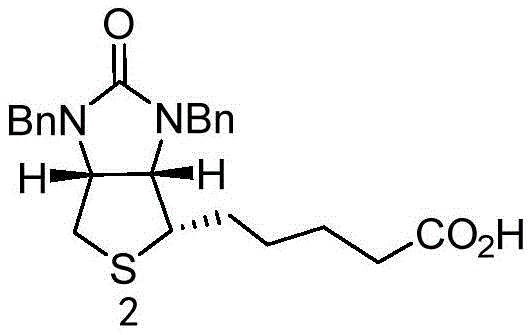
The secondary purification mechanism involves anti-solvent crystallization, a technique highly valued for its scalability and ability to reject structurally similar impurities. Once the Compound 2 is isolated in ethyl acetate, the solution is heated to 40-60°C to ensure full dissolution and kinetic energy for molecular arrangement. The addition of n-heptane, a non-polar anti-solvent, drastically reduces the solubility of the target compound. As the mixture is cooled to 5-10°C, the supersaturation level drives the nucleation and growth of pure crystals. The slow cooling rate and specific solvent ratios (organic phase to n-heptane ratio of 1:0.5-2) are critical; they prevent the occlusion of mother liquor containing impurities within the crystal lattice. This results in a solid product with purity levels consistently above 99%, effectively breaking the cycle of impurity accumulation that plagues conventional Vitamin H synthesis.
How to Synthesize Vitamin H Intermediate Efficiently
Implementing this purification protocol requires strict adherence to the specified physicochemical parameters to maximize yield and purity. The process begins with the catalytic hydrogenation of Compound 1 using Pd/C in ethanol, followed by the critical purification sequence detailed in the patent. Operators must monitor pH levels precisely during the acid-base transitions and maintain tight temperature control during the precipitation phase to ensure consistent crystal morphology. The following guide outlines the standardized operational procedure derived from the patent examples, designed for seamless integration into pilot and commercial-scale reactors.
- Dissolve the crude compound of formula 2 in an alkali solution (e.g., NaOH) to obtain a mixed solution with pH 10-14.
- Add acid (e.g., HCl) to the mixture to adjust pH to 1-3, followed by extraction with ethyl acetate to isolate the organic phase.
- Heat the organic phase to 40-60°C, add n-heptane as an anti-solvent, and cool to 5-10°C to precipitate the purified solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible strategic advantages beyond mere technical compliance. By shifting the purification burden to the intermediate stage, the overall manufacturing timeline is significantly compressed. The elimination of final API recrystallization steps reduces the consumption of high-grade solvents and minimizes the hold-up time in production vessels. This streamlining effect enhances the agility of the supply chain, allowing for faster response times to market demands for Vitamin H. Furthermore, the robustness of the acid-base extraction method means that variations in crude feedstock quality can be effectively mitigated, ensuring a consistent supply of high-quality intermediate regardless of upstream fluctuations.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the drastic simplification of the downstream processing. By achieving >99% purity at the intermediate stage, the necessity for expensive and yield-loss-prone purification of the final Vitamin H API is removed. This eliminates the costs associated with additional solvent cycles, filtration equipment usage, and drying times. Moreover, the reagents used—sodium hydroxide, hydrochloric acid, ethyl acetate, and n-heptane—are commodity chemicals with stable pricing, avoiding the volatility associated with specialized chromatography resins or exotic solvents. The cumulative effect is a substantial reduction in the cost of goods sold (COGS) for pharmaceutical grade Vitamin H.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical vitamins is paramount for pharmaceutical manufacturers. This method enhances reliability by decoupling the purity of the final product from the variability of the initial hydrogenation step. Even if the crude hydrogenation yield fluctuates, the purification protocol acts as a robust filter, consistently delivering specification-compliant Intermediate 2. This consistency reduces the risk of batch failures at the final API stage, which are notoriously costly and disruptive. Additionally, the use of common, widely available solvents ensures that production is not vulnerable to supply shortages of niche reagents, securing the continuity of manufacturing operations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is exceptionally favorable. The avoidance of column chromatography on a multi-ton scale significantly reduces silica waste and solvent volume, aligning with green chemistry principles. The precipitation step is inherently scalable; the thermodynamics of crystallization behave predictably when moving from liters to cubic meters, unlike some complex extraction techniques. The reduction in solvent usage per kilogram of product lowers the environmental footprint, simplifying waste treatment protocols and reducing disposal costs. This makes the technology not only economically viable but also sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and regulatory compliance.
Q: Why is purification of Intermediate 2 critical for Pharmaceutical Grade Vitamin H?
A: Conventional synthesis routes often result in intermediates with 80-95% purity, leading to final Vitamin H products that only meet feed-grade standards. Purifying Intermediate 2 to >99% purity ensures the final API meets European Pharmacopoeia (EP) standards without requiring additional final purification steps.
Q: What is the key mechanism behind the novel purification method?
A: The method utilizes a selective acid-base extraction followed by anti-solvent precipitation. By converting the intermediate to a water-soluble salt and then re-acidifying, water-soluble impurities are removed. Subsequent precipitation with n-heptane removes organic soluble impurities, achieving high purity.
Q: Does this method eliminate the need for final Vitamin H purification?
A: Yes. By controlling the purity at the Intermediate 2 stage (>99%), the subsequent debenzylation and cyclization steps yield Vitamin H with >99.8% purity and single impurities <0.1%, complying with EP standards directly after acid precipitation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin H Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification protocols described in Patent CN113121558A can be executed with precision at any volume. We maintain stringent purity specifications across all our vitamin intermediate lines, supported by rigorous QC labs equipped with state-of-the-art HPLC and GC-MS instrumentation. Our commitment to quality ensures that every batch of Vitamin H intermediate we supply meets the exacting standards required for pharmaceutical applications, providing you with a foundation for success in your final API synthesis.
We invite you to leverage our technical capabilities to optimize your Vitamin H supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are prepared to provide specific COA data from recent batches and comprehensive route feasibility assessments to demonstrate how our advanced purification methods can enhance your product quality while reducing overall manufacturing costs. Let us collaborate to secure a reliable, high-purity supply of Vitamin H intermediates for your global operations.
