Advanced Purification Technology for Vitamin H Intermediates: Enhancing Purity and Commercial Scalability
Advanced Purification Technology for Vitamin H Intermediates: Enhancing Purity and Commercial Scalability
The global demand for high-quality vitamins continues to surge, driven by increasing health consciousness and the expanding nutraceutical sector. Within this landscape, Vitamin H, commonly known as D-Biotin or Coenzyme R, stands out as a critical water-soluble vitamin essential for fat and protein metabolism. The industrial synthesis of Vitamin H relies heavily on the efficiency and purity of its key intermediates. A recent technological breakthrough, documented in patent CN115215802A, introduces a refined method for purifying the cyclic acid intermediate of Vitamin H. This innovation addresses long-standing challenges in impurity control and yield optimization, offering a robust pathway for manufacturers aiming to produce high-purity pharmaceutical intermediates. By integrating a specialized recrystallization protocol involving isopropanol and water, this method elevates the purity profile significantly above traditional standards, ensuring a superior starting material for the final API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
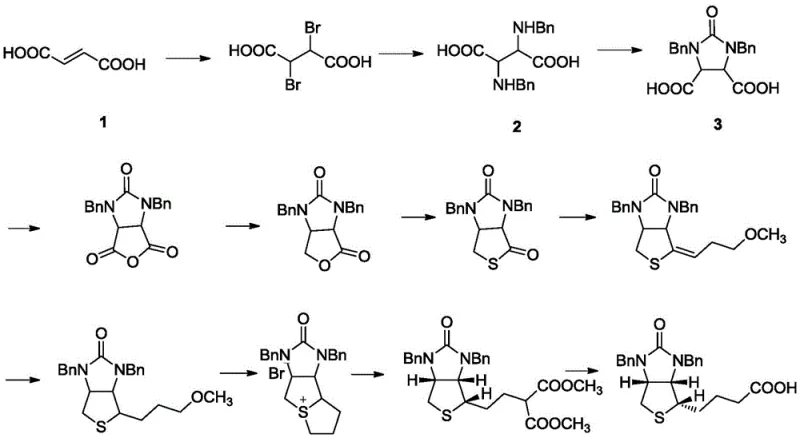
Historically, the industrial production of the Vitamin H cyclic acid intermediate has relied on a straightforward but limited workup procedure. In the traditional Hoffmann-La Roche route, the precursor Compound 2 undergoes cyclization using triphosgene and potassium hydroxide. Following the reaction, the standard practice involves simply adjusting the pH to acidic conditions, filtering the precipitate, washing, and drying. While operationally simple, this conventional approach suffers from significant drawbacks regarding product quality. The resulting crude product typically exhibits an HPLC purity ranging only between 95.5% and 96.5%. For modern pharmaceutical applications, where impurity thresholds are becoming increasingly stringent, this level of purity is often insufficient. Residual impurities at this stage can propagate through downstream synthesis steps, complicating the final purification of Vitamin H and potentially affecting the safety profile of the finished drug product. Furthermore, the lack of a dedicated purification step limits the ability to remove specific structural analogs or unreacted starting materials effectively.
The Novel Approach
The methodology disclosed in patent CN115215802A represents a paradigm shift in how this critical intermediate is processed. Instead of accepting the crude precipitate directly, the novel approach introduces a sophisticated recrystallization and acidification sequence. After the initial cyclization and isolation of the crude Compound 3, the process incorporates a dissolution step using a specific mixture of isopropanol and water. The mixture is heated to ensure complete clarity, followed by a controlled dropwise addition of hydrochloric acid. This precise manipulation of solvent composition and pH induces a highly selective crystallization event. The result is a dramatic improvement in product quality, with HPLC purity consistently reaching between 98.5% and 99.5%. This enhancement is not merely incremental; it fundamentally alters the quality baseline for the supply chain, reducing the burden on downstream processing and ensuring a more reliable feedstock for the production of high-purity Vitamin H.
Mechanistic Insights into Triphosgene-Mediated Cyclization and Recrystallization
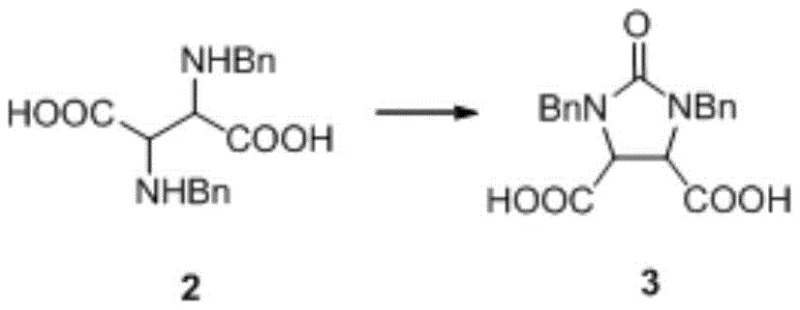
The core chemical transformation involves the cyclization of the dibenzyl-protected diamino dicarboxylic acid derivative (Compound 2) into the cyclic urea structure (Compound 3). This reaction utilizes triphosgene as a safe and effective source of phosgene equivalents. In the presence of potassium hydroxide, triphosgene decomposes to generate reactive carbonyl species that attack the amine groups of Compound 2. The mechanism proceeds through the formation of an isocyanate intermediate, which subsequently undergoes intramolecular nucleophilic attack by the adjacent amine to close the five-membered imidazolidinone ring. Maintaining the pH between 8 and 9 is mechanistically crucial; it ensures that the amine nucleophiles remain sufficiently deprotonated to react while preventing the rapid hydrolysis of the triphosgene or the newly formed cyclic urea. The temperature control between 10-30°C further moderates the reaction kinetics, preventing thermal degradation and minimizing the formation of polymeric byproducts.
The purification mechanism relies on the differential solubility properties of the target cyclic acid versus its impurities in an aqueous isopropanol system. Upon heating the crude solid in the solvent mixture to approximately 37-38°C, the target compound and soluble impurities dissolve. The subsequent dropwise addition of hydrochloric acid serves a dual purpose: it protonates basic impurities, keeping them in the aqueous phase, and it adjusts the ionization state of the cyclic acid to favor precipitation. As the solution is cooled to 0-10°C, the solubility of the target molecule drops sharply, leading to nucleation and crystal growth. Because the crystal lattice formation is highly selective, impurities are excluded from the growing crystal structure, remaining in the mother liquor. This thermodynamic control over crystallization is what drives the purity from the mid-90s to nearly 99%, effectively acting as a molecular sieve without the need for expensive chromatography.
How to Synthesize Vitamin H Intermediate Cyclic Acid Efficiently
Implementing this purification technology requires careful attention to the specific parameters outlined in the patent data to ensure reproducibility and maximum yield. The process begins with the preparation of the reaction mixture under strictly controlled thermal conditions to manage the exotherm associated with triphosgene addition. Following the isolation of the crude material, the critical recrystallization step must be executed with precision regarding solvent ratios and cooling rates. The following guide summarizes the operational workflow derived from the patent examples, providing a roadmap for technical teams to adopt this high-efficiency protocol. For the detailed standardized synthesis steps, please refer to the guide below.
- Cyclization Reaction: Dissolve Compound 2 in potassium hydroxide solution, cool to 10-15°C, and simultaneously add triphosgene/toluene solution and additional KOH while maintaining pH 8-9 and temperature 10-30°C.
- Crude Isolation: After reaction completion, adjust the pH to 1-2 using acid, filter the precipitate, and wash to obtain the crude Compound 3.
- Recrystallization Purification: Dissolve the crude product in a mixture of isopropanol and water at 37-38°C, add hydrochloric acid dropwise at 20-28°C, cool to 0-10°C, and filter to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced purification method translates into tangible strategic benefits beyond mere technical specifications. The primary advantage lies in the substantial enhancement of supply reliability and cost efficiency throughout the manufacturing value chain. By securing an intermediate with near-99% purity, downstream processing becomes significantly more predictable and less resource-intensive. This reduces the risk of batch failures in the final API production, which is a major cost driver in pharmaceutical manufacturing. Furthermore, the process utilizes common, commercially available solvents like isopropanol and hydrochloric acid, avoiding the need for exotic or hazardous reagents that could disrupt supply continuity.
- Cost Reduction in Manufacturing: The implementation of this purification route offers significant potential for cost optimization without compromising quality. By eliminating the need for complex chromatographic separations or multiple recrystallization cycles that are often required to upgrade lower-purity crude materials, the overall processing time and solvent consumption are drastically reduced. The high mass yield reported in the patent examples, consistently hovering around 97-98%, indicates that very little material is lost during the purification upgrade. This high recovery rate directly correlates to improved material efficiency and lower cost per kilogram of the final active ingredient. Additionally, the simplicity of the filtration and washing steps reduces labor and energy costs associated with extended drying or complex workup procedures.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often a bottleneck in the supply chain for vitamins and fine chemicals. This method enhances reliability by simplifying the production workflow, making it less susceptible to variability. The use of robust, scalable unit operations such as filtration and crystallization ensures that the process can be easily transferred between different manufacturing sites or scaled up to meet surging demand. The consistent purity profile of 98.5-99.5% reduces the need for extensive incoming quality control testing and renegotiation of specifications with downstream partners. This stability allows for longer-term supply contracts and better inventory planning, mitigating the risks associated with market volatility.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with modern green chemistry principles. The avoidance of heavy metal catalysts or chlorinated solvents (beyond the toluene/triphosgene system which is managed in the earlier step) simplifies waste treatment. The aqueous isopropanol mother liquor can be more easily treated or recycled compared to waste streams containing complex organic mixtures. The process generates minimal hazardous waste, facilitating easier compliance with increasingly strict environmental regulations. The scalability is further supported by the use of standard reactor equipment capable of handling slurry filtration and temperature-controlled crystallization, making the transition from pilot scale to multi-ton commercial production seamless and low-risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Vitamin H intermediates. These answers are derived directly from the experimental data and claims presented in patent CN115215802A, providing clarity on the operational parameters and expected outcomes of this novel method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: What is the purity improvement achieved by this new purification method compared to traditional methods?
A: Traditional methods typically yield a purity of 95.5% to 96.5%. The novel purification method described in patent CN115215802A utilizes a specific isopropanol-water recrystallization technique that consistently achieves HPLC purity levels between 98.5% and 99.5%, significantly reducing impurity profiles for downstream API synthesis.
Q: Why is the control of temperature and pH critical during the triphosgene addition step?
A: Maintaining the reaction temperature between 10-30°C and pH between 8-9 is essential to control the exothermic nature of the triphosgene reaction and prevent side reactions or decomposition of the sensitive diamine intermediate. Strict thermal control ensures high conversion rates and minimizes the formation of chlorinated byproducts.
Q: How does the isopropanol-water solvent system enhance the crystallization process?
A: The specific ratio of isopropanol to water creates an optimal solubility gradient for the cyclic acid intermediate. Heating to 37-38°C ensures complete dissolution of impurities, while the subsequent controlled acidification and cooling to 0-10°C forces the selective precipitation of the target compound in a highly crystalline form, facilitating easy filtration and drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin H Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final pharmaceutical product is inextricably linked to the purity of its precursors. Our technical team has thoroughly analyzed the advancements in Vitamin H intermediate synthesis, including the purification protocols described in recent patents. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of order volume. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cyclic acid intermediate meets the demanding requirements of the global nutraceutical and pharmaceutical markets.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in process chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to deliver high-purity Vitamin H intermediates that drive efficiency and excellence in your final products.
