Transforming Waste into Value: A Scalable Route for 3,4,5-Trifluorobromobenzene Production
Transforming Waste into Value: A Scalable Route for 3,4,5-Trifluorobromobenzene Production
In the highly competitive landscape of fine chemical manufacturing, the ability to convert low-value byproducts into high-performance intermediates represents a significant strategic advantage. The patent CN112010733B, published in August 2022, introduces a groundbreaking methodology for the preparation of 3,4,5-trifluorobromobenzene, a critical building block for advanced pharmaceuticals and agrochemicals. Traditionally, the production of chlorinated nitrobenzenes generates substantial amounts of 3,4,5-trichloronitrobenzene as an unwanted byproduct, which is often discarded due to the high cost and difficulty of purification. This new technical disclosure solves that inefficiency by establishing a direct, high-yield pathway that utilizes this specific byproduct as the primary raw material. By integrating fluorination, reduction, and deamination reactions, the process not only valorizes waste but also ensures a consistent supply of high-purity fluorinated aromatics. For R&D directors and procurement specialists, this represents a dual opportunity: securing a reliable source of complex intermediates while simultaneously driving down the cost base through intelligent feedstock selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of poly-fluorinated benzene derivatives has relied on starting materials that are either prohibitively expensive or require extensive purification sequences to remove isomeric impurities. In the specific context of chloronitrobenzene chlorination, the formation of 3,4,5-trichloronitrobenzene is inevitable, yet its utility has been severely limited. Conventional wisdom dictated that separating this solid byproduct from the desired 3,4-dichloronitrobenzene was economically unviable due to their similar solubilities and physical properties. Consequently, manufacturers were forced to treat this material as waste, incurring disposal costs and suffering from poor atom economy. Furthermore, alternative synthetic routes to 3,4,5-trifluorobromobenzene often involve multiple protection-deprotection steps or the use of exotic reagents that do not translate well to large-scale manufacturing. These traditional pathways frequently result in lower overall yields and generate significant hazardous waste streams, creating bottlenecks for supply chain continuity and environmental compliance.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by embracing the impurity rather than fighting it. Instead of attempting difficult solid-state separations, the novel approach immediately subjects the crude 3,4,5-trichloronitrobenzene to a selective fluorination reaction. This strategic pivot converts the solid, hard-to-purify nitro compound into liquid intermediates, such as 3,5-dichloro-4-fluoroaniline, which can be easily purified via distillation. This phase change from solid to liquid is the key enabler for high-purity output without expensive chromatography. The process flows through a logical sequence of functional group transformations—nitro reduction, diazotization, and subsequent re-fluorination—each step optimized to maximize yield and minimize side reactions. By the time the final bromination and deamination occur, the molecular architecture is precisely tuned, delivering 3,4,5-trifluorobromobenzene with exceptional purity. This approach effectively turns a waste management problem into a streamlined production asset.
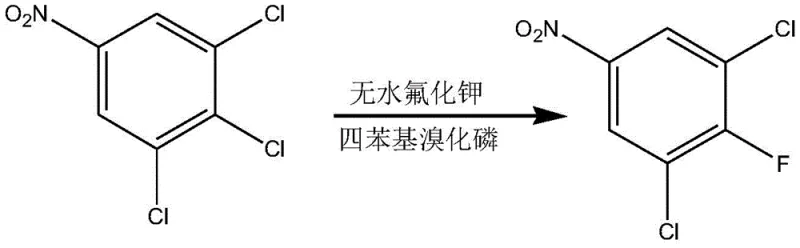
Mechanistic Insights into Sequential Fluorination and Deamination
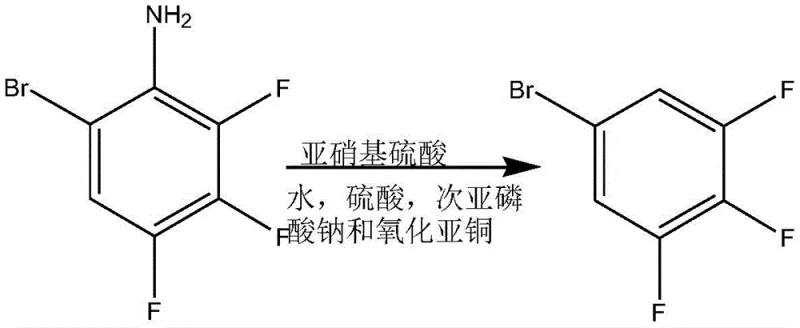
The core of this synthesis lies in the mastery of nucleophilic aromatic substitution (SnAr) and diazonium chemistry. The initial step involves the displacement of a chlorine atom on the nitrobenzene ring by fluoride ions derived from anhydrous potassium fluoride. This reaction is facilitated by a phase transfer catalyst, such as tetraphenylphosphonium bromide, which enhances the nucleophilicity of the fluoride ion in the organic phase. The presence of the strong electron-withdrawing nitro group ortho and para to the leaving chlorine atom activates the ring sufficiently for this substitution to proceed at elevated temperatures (175-180°C). Following this, the nitro group is reduced to an amine using a skeletal nickel catalyst under hydrogen pressure. This reduction is critical not only for changing the electronic nature of the ring but also for enabling the subsequent deamination step. The conversion of the amino group into a diazonium salt using nitroso sulfuric acid allows for the replacement of the nitrogen functionality with a hydrogen atom, effectively removing the directing group and simplifying the substitution pattern for future steps.
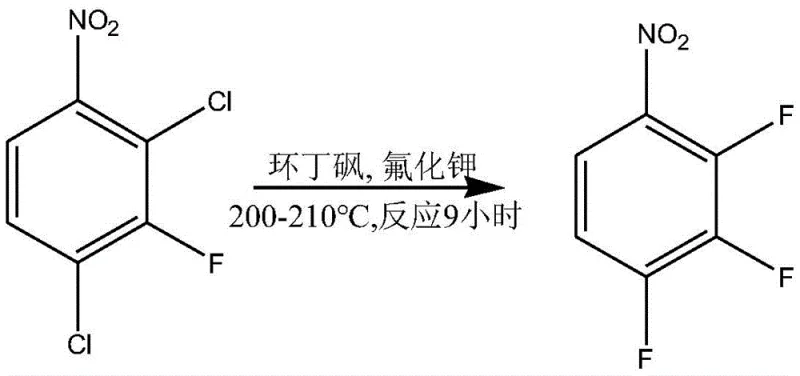
Further down the synthetic tree, the process employs a second, more aggressive fluorination to replace the remaining chlorine atoms. This step utilizes sulfolane as a high-boiling polar aprotic solvent, allowing the reaction to reach temperatures of 200-210°C, which is necessary to overcome the higher activation energy of displacing chlorine atoms from a less activated ring. The final stages involve a precise bromination of the trifluoroaniline intermediate followed by another deamination. The use of bromine and hydrogen peroxide in an acidic medium ensures regioselective bromination, while the final diazotization-reduction sequence cleanly removes the amino group to yield the target 3,4,5-trifluorobromobenzene. Throughout this cascade, the control of impurities is managed through physical separation techniques like steam distillation, which leverages the volatility differences between the desired fluorinated products and non-volatile tarry byproducts.
How to Synthesize 3,4,5-Trifluorobromobenzene Efficiently
Executing this synthesis requires careful attention to reaction conditions, particularly temperature control and moisture exclusion during the fluorination steps. The process begins with the dehydration of the starting material to prevent hydrolysis of the fluoride source. Subsequent steps involve standard hydrogenation equipment and acid-resistant reactors for the diazotization phases. The integration of steam distillation is a crucial operational detail that allows for the purification of intermediates without the need for complex solvent extraction workflows. For a detailed breakdown of the specific molar ratios, reaction times, and workup procedures required to achieve the reported yields of over 80% in key steps, please refer to the standardized protocol below.
- Perform nucleophilic fluorination on 3,4,5-trichloronitrobenzene using anhydrous potassium fluoride and a phase transfer catalyst to obtain 3,5-dichloro-4-fluoronitrobenzene.
- Reduce the nitro group to an amine using skeletal nickel catalyst and hydrogen to form 3,5-dichloro-4-fluoroaniline.
- Execute a deamination reaction via diazotization followed by reduction with sodium hypophosphite to yield 2,6-dichlorofluorobenzene.
- Nitrate the 2,6-dichlorofluorobenzene using mixed acid to introduce a nitro group, forming 2,4-dichloro-3-fluoronitrobenzene.
- Conduct a second high-temperature fluorination using sulfolane and potassium fluoride to replace remaining chlorine atoms, yielding 2,3,4-trifluoronitrobenzene.
- Reduce the nitro compound to 2,3,4-trifluoroaniline using catalytic hydrogenation.
- Brominate the aniline derivative followed by a final deamination step to produce the target 3,4,5-trifluorobromobenzene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers a compelling value proposition for organizations seeking to optimize their supply chains for fluorinated intermediates. The primary driver of cost efficiency is the utilization of 3,4,5-trichloronitrobenzene, a material that is typically available at a fraction of the cost of purpose-synthesized fine chemicals. By sourcing this feedstock from existing chlorination processes, manufacturers can significantly decouple their production costs from the volatility of the broader fluorine chemical market. Furthermore, the elimination of complex purification steps for the starting material reduces both capital expenditure on separation equipment and operational expenditure on solvents and energy. The robustness of the reaction conditions, which rely on commodity chemicals like potassium fluoride and sulfuric acid rather than precious metal catalysts, further insulates the production process from supply chain disruptions associated with rare earth or noble metal shortages.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it monetizes a waste stream. By avoiding the purchase of high-purity specialty starting materials and instead upgrading a low-cost byproduct, the overall cost of goods sold (COGS) is drastically reduced. Additionally, the recovery and reuse of solvents like sulfolane and methanol, as described in the patent examples, contribute to a circular economy within the plant, minimizing raw material consumption and waste disposal fees. The high yields reported in the experimental data indicate that very little material is lost to side reactions, maximizing the throughput of the facility.
- Enhanced Supply Chain Reliability: Dependence on a single source for complex intermediates is a major risk for pharmaceutical and agrochemical companies. This synthesis route diversifies the supply base by enabling production from a widely available chlorination byproduct. Since 3,4-dichloronitrobenzene is produced in large volumes globally, the feedstock for this process is abundant and geographically diverse. This abundance ensures that production schedules can be maintained even when specific fine chemical suppliers face capacity constraints. The simplicity of the unit operations also means that the technology can be transferred to multiple manufacturing sites with relative ease, further securing the supply chain against localized disruptions.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are compatible with standard stainless steel and glass-lined reactors. The absence of cryogenic conditions or ultra-high-pressure requirements simplifies the engineering controls needed for commercialization. From an environmental standpoint, the valorization of a chemical byproduct aligns perfectly with green chemistry principles, reducing the overall carbon footprint of the manufacturing operation. The use of steam distillation reduces the volume of organic wastewater generated compared to extensive aqueous washing protocols, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific product pipeline. The answers are derived directly from the experimental data and technical disclosures found in the patent literature, ensuring accuracy and relevance for process development teams.
Q: How does this process handle impurities in the starting byproduct?
A: The process is designed to tolerate low-purity starting materials. Impurities present in the crude 3,4,5-trichloronitrobenzene are effectively removed during the intermediate purification steps, particularly through steam distillation and rectification of liquid intermediates like 3,5-dichloro-4-fluoroaniline, eliminating the need for expensive pre-purification of the solid byproduct.
Q: What are the key advantages of using 3,4,5-trichloronitrobenzene as a feedstock?
A: Utilizing 3,4,5-trichloronitrobenzene, which is typically a waste byproduct of 3,4-dichloronitrobenzene production, significantly lowers raw material costs. This approach transforms a disposal liability into a high-value asset, improving overall process economics and reducing environmental waste associated with chlorination processes.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route utilizes standard unit operations such as vacuum rectification, catalytic hydrogenation, and steam distillation. The reaction conditions, including temperatures up to 210°C for fluorination, are achievable in standard industrial reactors, ensuring robust commercial scale-up potential from kilogram to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluorobromobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory to the marketplace. Our facilities are equipped with the specialized reactors and safety systems required to handle high-temperature fluorination and hazardous reagents like bromine and mixed acids safely. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,4,5-trifluorobromobenzene meets the exacting standards required for API and advanced agrochemical synthesis.
We invite you to leverage our technical proficiency to optimize your supply chain and reduce your manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you transform the potential of this innovative synthesis route into a tangible competitive advantage for your business.
