Scalable One-Pot Synthesis of 3,4,5-Trifluorobromobenzene for High-Volume Agrochemical Manufacturing
Introduction to Advanced Fluorinated Intermediate Synthesis
The escalating global demand for high-performance succinate dehydrogenase inhibitor (SDHI) fungicides, particularly fluxapyroxad, has necessitated the development of more efficient synthetic routes for key building blocks like 3,4,5-trifluorobromobenzene. Patent CN116253612A introduces a transformative one-pot methodology that converts readily available 2,6-difluoroaniline into this critical intermediate through a seamless sequence of bromination, diazotization, and fluorination. This technological leap addresses long-standing bottlenecks in agrochemical intermediate manufacturing by significantly enhancing regioselectivity and operational safety while minimizing waste generation. For R&D directors and supply chain leaders, this patent represents a viable pathway to secure a stable, cost-effective supply of high-purity precursors essential for next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4,5-trifluorobromobenzene has relied on routes starting from 1,2,3-trichlorobenzene, which involve harsh fluorination and bromination steps that frequently result in poor selectivity. These conventional pathways often generate a complex mixture of positional isomers due to the similar reactivity of the chlorine substituents, necessitating energy-intensive and yield-depleting separation processes to isolate the desired product. Furthermore, alternative strategies involving nitration and reduction sequences to introduce amino groups for selectivity improvement invariably increase the number of synthetic steps, thereby compounding material costs and expanding the environmental footprint through increased solvent usage and waste discharge. The cumulative effect of these inefficiencies is a fragile supply chain vulnerable to yield fluctuations and elevated production costs that ultimately impact the profitability of the final fungicide.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the intrinsic electronic and steric properties of 2,6-difluoroaniline to achieve superior control over the reaction trajectory. By utilizing the amino group as a powerful directing handle and the fluorine atoms as blocking groups, the process ensures that bromination occurs exclusively at the para-position, effectively eliminating the formation of unwanted ortho-isomers. This strategic design allows for the consolidation of multiple reaction steps into a single vessel, streamlining the workflow and reducing the need for intermediate isolation. The result is a robust, high-yielding process that not only simplifies operations but also aligns with modern green chemistry principles by reducing the overall consumption of resources and hazardous reagents.
Mechanistic Insights into Regioselective Bromination and Fluorination
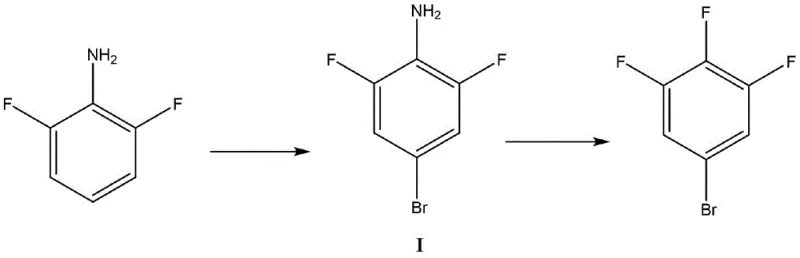
The core of this synthesis lies in the precise manipulation of electrophilic aromatic substitution and diazonium chemistry to install the bromine and fluorine atoms with atomic precision. In the initial bromination phase, the amino group of 2,6-difluoroaniline activates the aromatic ring towards electrophilic attack, while the fluorine atoms at the 2 and 6 positions sterically hinder the ortho-sites, forcing the brominating species—whether generated from NBS, Br2, or NaBr/H2O2—to attack the para-position exclusively. This inherent regiocontrol is further enhanced by the choice of catalysts such as ferric chloride or radical initiators like AIBN, which optimize the reaction kinetics to ensure complete conversion of the starting material without over-bromination or side reactions. The resulting 2,6-difluoro-4-bromoaniline intermediate is formed with exceptional purity, setting the stage for the subsequent transformation.
Following bromination, the process transitions seamlessly into diazotization and fluorination, where the amino group is converted into a diazonium salt using reagents like tert-butyl nitrite or sodium nitrite under controlled acidic conditions. This unstable intermediate is immediately subjected to nucleophilic attack by fluoride ions sourced from complexes such as HF-triethylamine or potassium fluoride, facilitating the replacement of the diazonium group with a fluorine atom via a Balz-Schiemann-type mechanism. This one-pot telescoping of steps prevents the accumulation of hazardous diazonium salts and minimizes exposure risks, while the careful selection of solvents like acetonitrile or dichloroethane ensures optimal solubility and reaction rates. The mechanistic elegance of this route guarantees that the final 3,4,5-trifluorobromobenzene product is obtained with minimal impurities, reducing the burden on downstream purification units.
How to Synthesize 3,4,5-Trifluorobromobenzene Efficiently
Implementing this synthesis requires careful attention to reaction parameters such as temperature control and reagent stoichiometry to maximize the efficiency of the one-pot sequence. The patent outlines a standardized protocol where 2,6-difluoroaniline is first treated with a brominating agent and catalyst at moderate temperatures (30-60°C) until conversion is complete, followed by the direct addition of diazotizing and fluorinating agents to the same reactor. This operational simplicity reduces equipment requirements and operator intervention, making it highly attractive for facilities aiming to optimize their batch cycles. For detailed procedural specifics regarding reagent ratios, solvent choices, and workup procedures, please refer to the standardized synthesis guide below.
- Brominate 2,6-difluoroaniline using NBS or Br2 with a catalyst like AIBN or FeCl3 in an organic solvent at 30-60°C to form 2,6-difluoro-4-bromoaniline.
- Without isolating the intermediate, add a diazotizing agent (e.g., tert-butyl nitrite) and a fluorinating agent (e.g., HF-Et3N complex) to the reaction mixture.
- Heat the mixture to facilitate diazotization and subsequent fluorination, then purify via extraction and distillation to obtain high-purity 3,4,5-trifluorobromobenzene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic advantages by addressing key pain points related to cost, scalability, and regulatory compliance in the agrochemical sector. The ability to source raw materials like 2,6-difluoroaniline from established supply chains ensures consistent availability, while the high atom economy of the one-pot design translates directly into reduced raw material consumption per kilogram of product. By eliminating the need for multiple isolation steps and the associated solvent swaps, the process significantly lowers utility costs and waste disposal fees, providing a clear path to margin improvement for manufacturers. Furthermore, the demonstrated success of kilogram-scale trials confirms the technical feasibility of scaling this route to multi-ton production levels without compromising quality or safety standards.
- Cost Reduction in Manufacturing: The consolidation of three distinct chemical transformations into a single reactor vessel drastically reduces the operational overhead associated with heating, cooling, and transferring materials between steps. By avoiding the isolation of the brominated intermediate, the process saves significant amounts of solvent and energy that would otherwise be consumed during filtration, drying, and redissolution phases. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the cost and complexity of final purification and increases the overall yield of saleable product. These cumulative efficiencies result in a leaner manufacturing process that is inherently more cost-competitive than traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2,6-difluoroaniline, NBS, and common fluorinating agents mitigates the risk of supply disruptions often associated with exotic or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, provides flexibility in sourcing and allows for adaptation to local supply chain constraints without sacrificing performance. This resilience ensures a steady flow of high-purity intermediates to downstream formulation plants, safeguarding production schedules against raw material volatility. Consequently, procurement teams can negotiate more favorable terms and secure long-term supply agreements with greater confidence.
- Scalability and Environmental Compliance: The reduction in solvent usage and waste generation inherent to the one-pot design aligns perfectly with increasingly stringent environmental regulations governing chemical manufacturing. By minimizing the volume of hazardous waste requiring treatment or disposal, facilities can lower their environmental compliance costs and reduce their carbon footprint. The process has already been validated at the kilogram scale, demonstrating that heat transfer and mixing issues can be effectively managed in larger reactors. This proven scalability assures supply chain heads that the technology is ready for immediate commercial deployment, supporting the rapid expansion of capacity to meet growing market demand for fluxapyroxad.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for adoption. Understanding these nuances is critical for making informed decisions about process integration and supplier qualification.
Q: What are the primary advantages of this one-pot method over traditional trichlorobenzene routes?
A: Traditional routes starting from 1,2,3-trichlorobenzene often suffer from poor bromination selectivity, generating difficult-to-separate isomers that lower overall yield. This novel method utilizes 2,6-difluoroaniline, where the fluorine atoms inherently block ortho-positions, ensuring exclusive para-bromination and significantly higher regioselectivity.
Q: How does this process impact environmental compliance and waste generation?
A: By combining bromination, diazotization, and fluorination into a one-pot sequence, the process eliminates multiple isolation and purification steps required in conventional multi-step syntheses. This consolidation drastically reduces solvent consumption and the generation of hazardous 'three wastes,' aligning with stricter environmental regulations for agrochemical manufacturing.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates successful kilogram-level scale-up (Example 4), achieving high purity (>96%) and robust yields. The use of commercially available raw materials like 2,6-difluoroaniline and standard reagents ensures the supply chain reliability necessary for ton-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluorobromobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern agrochemical formulations, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every shipment of 3,4,5-trifluorobromobenzene meets the exacting standards required for pharmaceutical and agrochemical applications. Our dedication to technical excellence allows us to navigate complex regulatory landscapes and deliver products that support your innovation pipeline.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific manufacturing needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain structure. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency and reduce your total cost of ownership. Let us be your partner in driving value and reliability in your agrochemical intermediate supply chain.
