Advanced Manufacturing of 3,4,5-Trifluorobromobenzene for Global Agrochemical Supply Chains
The global demand for high-performance agrochemical intermediates continues to surge, driven by the need for more efficient crop protection solutions. A pivotal development in this sector is documented in patent CN112174770A, which outlines a robust production process for 3,4,5-trifluorobromobenzene. This compound serves as a critical building block for synthesizing succinic dehydrogenase inhibitor (SDHI) fungicides, such as fluxapyroxad, and is also utilized in advanced liquid crystal materials. The patented methodology addresses longstanding challenges in fluorinated aromatic synthesis, specifically targeting the optimization of yield and the minimization of environmental waste. By leveraging a novel diazotization reagent, this process offers a compelling value proposition for a reliable agrochemical intermediate supplier seeking to enhance their portfolio with sustainable manufacturing capabilities. The technical breakthroughs detailed herein provide a foundation for significant cost reduction in pesticide intermediate manufacturing while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
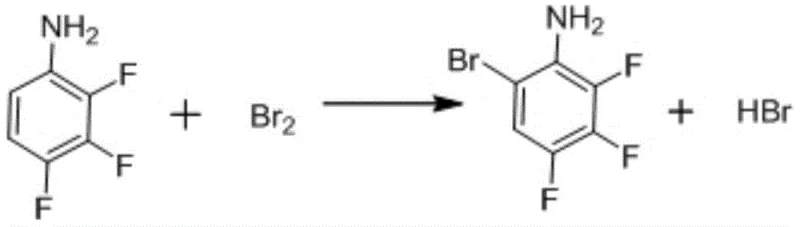
Traditionally, the synthesis of 3,4,5-trifluorobromobenzene from 2,3,4-trifluoroaniline has relied on sodium nitrite as the primary diazotization reagent. This conventional approach, while chemically feasible, presents substantial drawbacks in an industrial setting. The use of sodium nitrite often leads to the generation of significant amounts of nitrogen oxides and saline waste streams, complicating the three wastes treatment process and increasing operational expenditures. Furthermore, conventional methods frequently struggle to maintain consistent yield and purity levels, often resulting by-products that require extensive and costly purification steps. The instability of reaction conditions in older protocols can also pose safety risks, particularly when handling large volumes of reactive intermediates. These inefficiencies create bottlenecks for supply chain heads who require predictable output and minimal environmental liability. Consequently, the industry has long sought a method that mitigates these waste issues without compromising the structural integrity of the final fluorinated product.
The Novel Approach
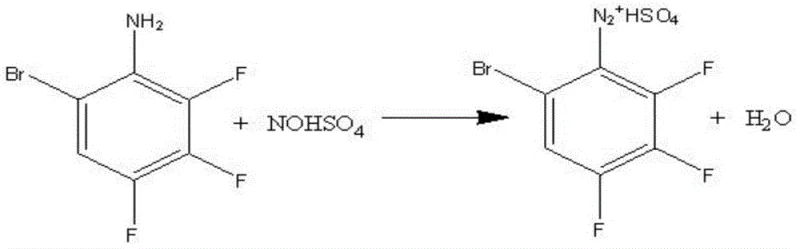
The innovative process described in the patent data introduces nitrosyl sulfuric acid as a superior diazotization reagent, fundamentally altering the reaction landscape. This strategic substitution eliminates the need for sodium nitrite, thereby drastically reducing the volume of hazardous waste generated during the diazotization phase. The new approach ensures that the acid utilized in the reaction can be effectively recycled, contributing to a more circular and efficient manufacturing loop. By optimizing the molar ratios of reactants, specifically maintaining a precise balance between the aniline derivative and the diazotization agent, the process achieves a remarkable yield of 85% with a purity of 99%. This level of precision not only enhances the economic viability of the production run but also simplifies the downstream processing requirements. For procurement managers, this translates to a more stable supply of high-purity 3,4,5-trifluorobromobenzene, reducing the risk of batch failures and ensuring continuity in the production of downstream fungicides.
Mechanistic Insights into Nitrosyl Sulfuric Acid Diazotization
The core of this technological advancement lies in the mechanistic efficiency of the diazotization step. In the patented process, nitrosyl sulfuric acid acts as a potent nitrosating agent that reacts smoothly with the brominated aniline intermediate. The reaction is conducted at temperatures below 10°C, ensuring thermal stability and preventing the decomposition of the diazonium salt, which is a common failure point in less controlled environments. The use of concentrated sulfuric acid as a solvent medium further stabilizes the reaction mixture, allowing for a prolonged reaction time of approximately 15 hours followed by a heat preservation period. This extended duration ensures complete conversion of the starting material, minimizing the presence of unreacted amines that could act as impurities in the final product. The mechanistic pathway avoids the formation of unstable by-products often associated with aqueous nitrite systems, thereby streamlining the purification workflow.
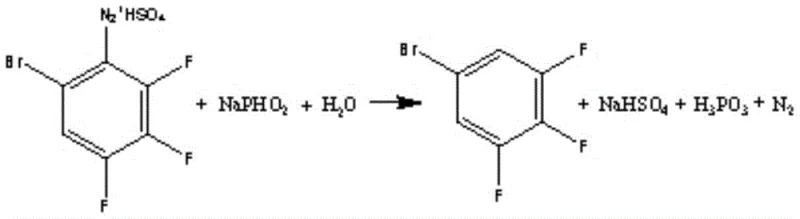
Impurity control is further enhanced during the subsequent deamination step, where sodium hypophosphite is employed as the reducing agent. The reaction is carefully managed at temperatures below 50°C to prevent thermal runaway while ensuring the efficient removal of the diazo group. The stoichiometry is tightly controlled, with a weight ratio of sodium hypophosphite to the initial aniline derivative maintained between 1:1.3 and 1:1.6. This precise dosing prevents the accumulation of phosphorus-containing by-products that could contaminate the final distillate. Following the reaction, the crude product undergoes alkali washing and water washing to remove acidic residues, followed by rectification. This multi-stage purification protocol guarantees that the final 3,4,5-trifluorobromobenzene meets the stringent purity specifications required for sensitive agrochemical applications, ensuring that the impurity profile remains well within acceptable limits for downstream synthesis.
How to Synthesize 3,4,5-Trifluorobromobenzene Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters outlined in the patent data. The process begins with the bromination of 2,3,4-trifluoroaniline, where temperature control below 15°C is critical to prevent poly-bromination. Following this, the diazotization step must be executed with high-purity nitrosyl sulfuric acid to ensure the reaction proceeds without generating excessive nitrogen oxide gases. The final deamination step utilizes sodium hypophosphite under controlled heating to release nitrogen gas safely while forming the carbon-bromine bond. Detailed standardized synthesis steps see the guide below.
- Conduct bromination of 2,3,4-trifluoroaniline below 15°C using bromine and hydrogen peroxide.
- Perform diazotization using nitrosyl sulfuric acid at temperatures below 10°C to minimize waste.
- Execute deamination with sodium hypophosphite below 50°C followed by distillation to achieve 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers tangible strategic benefits beyond mere chemical yield. The elimination of sodium nitrite and the reduction of three wastes directly correlate to lower disposal costs and reduced regulatory burden, which are significant factors in the total cost of ownership for chemical manufacturing. The ability to recycle acid within the process further diminishes the consumption of raw materials, leading to substantial cost savings over the lifecycle of the production campaign. Moreover, the robustness of the reaction conditions enhances supply chain reliability by minimizing the risk of batch-to-batch variability. This consistency is crucial for maintaining just-in-time delivery schedules for downstream pharmaceutical and agrochemical clients who depend on a steady flow of high-quality intermediates.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the reduction of waste treatment expenses and the efficient recycling of acidic reagents. By avoiding the use of sodium nitrite, the facility eliminates the need for complex scrubbing systems required to handle nitrogen oxide emissions, thereby lowering capital and operational expenditures. The high yield of 85% ensures that raw material utilization is maximized, reducing the cost per kilogram of the final product. Additionally, the simplified purification sequence reduces energy consumption associated with distillation and drying, contributing to a leaner manufacturing footprint. These factors combined create a compelling economic case for switching to this novel synthetic route.
- Enhanced Supply Chain Reliability: The stability of the reaction conditions, particularly the use of non-flammable and stable oxidants like hydrogen peroxide, ensures that production schedules are not disrupted by safety incidents or unplanned shutdowns. The process is designed to be scalable, allowing for seamless transition from pilot batches to commercial scale-up of complex fluorinated intermediates. This scalability ensures that suppliers can meet fluctuating market demands without compromising on quality or lead times. The high purity of 99% reduces the need for re-processing or rejection of batches, ensuring a consistent flow of goods to the customer. This reliability is a key differentiator for a reliable agrochemical intermediate supplier in a competitive global market.
- Scalability and Environmental Compliance: The process is inherently designed with environmental compliance in mind, generating significantly less hazardous waste compared to traditional methods. The absence of heavy metal catalysts and the use of recyclable acids simplify the waste management protocol, making it easier to adhere to strict environmental regulations. The safety profile of the reaction, which avoids the generation of explosive gas phases, facilitates easier permitting and operation in regulated jurisdictions. This environmental and safety advantage future-proofs the manufacturing asset against tightening global regulations on chemical emissions. It allows the manufacturer to position itself as a sustainable partner for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,4,5-trifluorobromobenzene based on the patented methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. They are intended to provide clarity on the process capabilities and its alignment with industrial standards for quality and safety.
Q: What is the primary advantage of using nitrosyl sulfuric acid in this synthesis?
A: Using nitrosyl sulfuric acid instead of traditional sodium nitrite significantly reduces the generation of three wastes during the diazotization process, leading to a cleaner production profile and simplified downstream purification.
Q: What purity levels can be achieved with this patented process?
A: The process consistently yields 3,4,5-trifluorobromobenzene with a purity of 99% and a yield of approximately 85%, meeting the stringent specifications required for high-performance pesticide intermediates.
Q: Is the bromination step considered safe for industrial scale-up?
A: Yes, the reaction environment is acidic and stable, with hydrogen peroxide acting as a safe oxidant. The process avoids the generation of flammable gas phases, ensuring safety during commercial scale-up of complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluorobromobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of 3,4,5-trifluorobromobenzene meets the exacting standards required for SDHI fungicide synthesis. Our infrastructure is designed to handle complex fluorinated chemistries safely and efficiently, providing our partners with a secure and dependable source of supply.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with us, you gain access to a wealth of chemical expertise and a production capacity that is ready to support your growth in the global agrochemical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
