Advanced Purification Technology for High-Purity Sugammadex Sodium: Overcoming Oxidation and Scalability Challenges
Advanced Purification Technology for High-Purity Sugammadex Sodium: Overcoming Oxidation and Scalability Challenges
The pharmaceutical landscape for neuromuscular blocking reversal agents has been significantly advanced by the development of robust purification methodologies for Sugammadex Sodium, as detailed in patent CN109021147B. This intellectual property represents a paradigm shift from traditional, resource-intensive purification techniques to a streamlined, chemically elegant process that ensures exceptional product quality. The core innovation lies in the utilization of a specialized organic acid-organic base-reducing agent mixed system, which effectively dissociates crude Sugammadex Sodium into its acid form under remarkably mild conditions. This approach not only preserves the delicate cyclodextrin architecture but also integrates antioxidant protection directly into the purification workflow, addressing the critical issue of thioether oxidation that has plagued previous manufacturing attempts. For global pharmaceutical manufacturers, this technology offers a viable pathway to producing high-purity active pharmaceutical ingredients (APIs) that meet stringent regulatory standards while optimizing production efficiency.
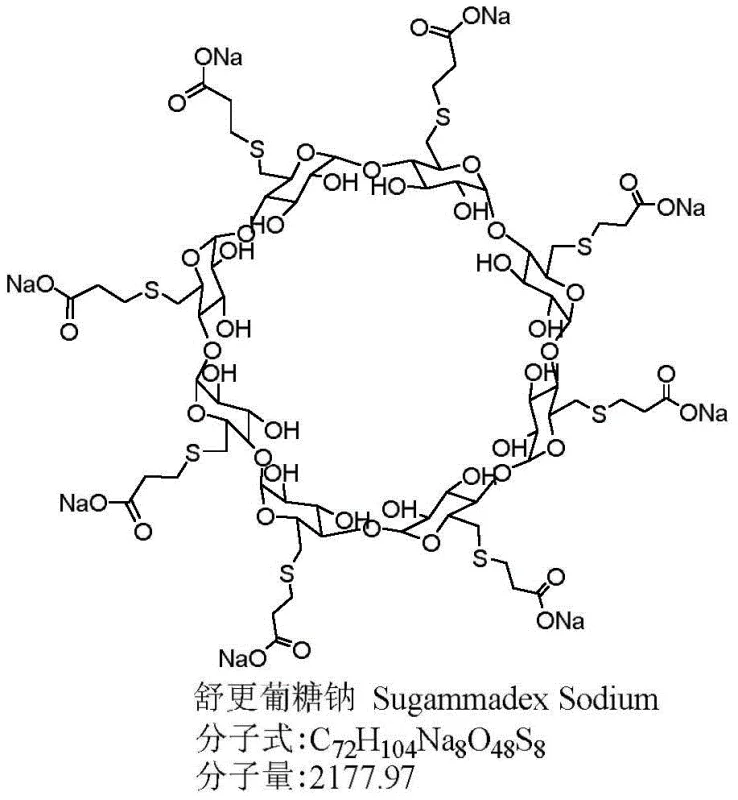
The structural complexity of Sugammadex Sodium, characterized by a gamma-cyclodextrin backbone modified with eight carboxyethyl thio groups, necessitates a purification strategy that is both selective and gentle. As illustrated in the molecular diagram, the presence of multiple sulfur atoms renders the molecule susceptible to oxidation, which can compromise patient safety if not rigorously controlled. The patented method addresses this by replacing harsh inorganic acid treatments with a buffered organic system, typically employing acetic acid and triethylamine to maintain a pH between 3 and 4. This precise control over the reaction environment prevents the hydrolysis of glycosidic bonds, a common failure mode in earlier processes that resulted in low yields and difficult-to-handle viscous solids. By stabilizing the substrate during the critical free acid conversion step, the process lays the foundation for high-efficiency downstream purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Sugammadex Sodium has been hindered by reliance on inefficient and environmentally burdensome technologies such as prolonged dialysis and preparative high-performance liquid chromatography (HPLC). Prior art methods, including those disclosed in US6670340, often require dialysis periods exceeding 36 hours, consuming vast quantities of water and generating substantial volumes of wastewater that pose significant environmental disposal challenges. Furthermore, the final product obtained from dialysis remains in an aqueous state, complicating subsequent isolation steps due to the high water solubility of the target molecule. Alternative approaches utilizing silica gel columns or nanofiltration membranes have shown limited success in removing structurally similar impurities, often failing to achieve the purity levels required for injectable formulations. Additionally, the use of strong inorganic acids for dissociation in older protocols frequently leads to substrate degradation, resulting in sticky, amorphous solids that are difficult to filter and dry, thereby drastically reducing overall process yield and operational throughput.
The Novel Approach
In stark contrast to these legacy techniques, the novel purification method introduced in patent CN109021147B leverages a sophisticated organic solvent system combined with reductive chemistry to achieve superior purification outcomes. By employing a mixture of organic acids and bases, the process creates a homogeneous reaction medium that facilitates the complete dissolution of the crude material without compromising its structural integrity. The integration of a reducing agent, such as tris(p-methylphenyl)phosphine or zinc powder, directly into the dissociation and recrystallization steps provides a continuous shield against oxidative degradation. This strategic addition allows for the effective removal of related substances and ensures that the final recrystallized acid possesses a purity exceeding 98% before salt formation. The subsequent conversion to the sodium salt is performed under controlled alkaline conditions using sodium hydroxide or carbonate, yielding a free-flowing crystalline powder with a total purity greater than 99.5%, effectively bypassing the need for chromatographic separation or membrane dialysis.
Mechanistic Insights into Reductive Stabilization and Impurity Control
The chemical stability of Sugammadex Sodium is intrinsically linked to the oxidation state of its eight thioether linkages, which are prone to forming sulfoxide derivatives upon exposure to oxygen or oxidizing agents. These sulfoxide impurities, specifically identified as Formula II and Formula III in the patent literature, are not only difficult to separate due to their structural similarity but also pose potential safety risks in intravenous applications. The mechanistic breakthrough of this purification technology is the proactive suppression of these oxidative pathways through the introduction of stoichiometric amounts of reducing agents throughout the process. During the initial dissociation phase, the reducing agent scavenges dissolved oxygen and reactive oxygen species within the organic acid-base matrix, preventing the initial formation of sulfoxides. This protective effect is maintained during the critical recrystallization step, where the solubility differences between the target acid and impurities are exploited in a reducing environment.
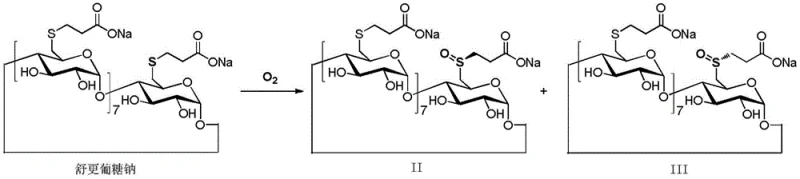
As depicted in the reaction scheme, the transformation of the stable thioether moiety into sulfoxide impurities is a thermodynamically favorable process in the presence of oxygen, leading to a mixture of regioisomers that complicate purification. The patented method effectively interrupts this pathway by maintaining a low redox potential within the reaction vessel. Experimental data from the patent examples demonstrates that when this reductive protocol is followed, the total content of oxidation impurities (the sum of sulfoxide impurities II and III) is consistently controlled to less than 0.1%, with individual single impurities also remaining below the 0.1% threshold. This level of impurity control is achieved without the need for expensive chiral columns or repeated recrystallizations, highlighting the efficiency of the chemical design. Furthermore, the use of mild organic acids prevents the acid-catalyzed hydrolysis of the cyclodextrin ring, ensuring that the primary degradation pathway is blocked, thereby maximizing the recovery of the pharmacologically active octamercapto-substituted species.
How to Synthesize Sugammadex Sodium Efficiently
The implementation of this purification protocol requires precise control over solvent ratios, temperature profiles, and reagent addition rates to ensure consistent crystal morphology and purity. The process begins with the preparation of the organic acid-organic base mixture, followed by the careful addition of the reducing agent and crude substrate under an inert atmosphere to minimize oxygen ingress. Detailed standard operating procedures regarding the specific weight-to-volume ratios of solvents like DMF and acetonitrile, as well as the molar equivalents of sodium base required for salt formation, are critical for reproducibility. For a comprehensive guide on the exact experimental parameters and step-by-step instructions validated by the patent examples, please refer to the standardized synthesis protocol below.
- Dissociate crude Sugammadex Sodium into Sugammadex acid using a mixed system of organic acid (e.g., acetic acid), organic base (e.g., triethylamine), and a reducing agent at 20-40°C.
- Recrystallize the resulting Sugammadex acid in a solvent system containing a reducing agent (e.g., DMF/Acetonitrile) to remove structural impurities and prevent oxidation.
- Convert the purified acid to Sugammadex Sodium by reacting with a sodium-containing base (e.g., NaOH) in an aqueous alcohol solution, followed by crystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification technology offers transformative benefits for procurement managers and supply chain directors seeking to optimize the cost structure and reliability of their API supply. The elimination of preparative HPLC and long-duration dialysis steps translates directly into a drastic reduction in processing time and utility consumption, particularly water and electricity. By replacing these bottleneck operations with standard crystallization and filtration units, manufacturers can significantly increase batch throughput and reduce the capital expenditure associated with specialized purification equipment. The simplicity of the reagent profile, utilizing commonly available organic acids, bases, and reducing agents, further mitigates supply chain risks associated with sourcing exotic or highly regulated chemicals. This operational simplicity ensures a more robust and resilient supply chain capable of meeting fluctuating market demands without the delays inherent in complex multi-step purification workflows.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive consumables and energy-intensive unit operations. Traditional methods relying on large quantities of activated carbon or silica gel generate significant solid waste disposal costs, while dialysis requires extensive water treatment infrastructure. By shifting to a solvent-based recrystallization process, the manufacturing footprint is reduced, and the cost of goods sold (COGS) is lowered through improved yield and reduced waste generation. The avoidance of strong inorganic acids also reduces corrosion-related maintenance costs for reactor vessels and piping, contributing to long-term operational savings. Furthermore, the higher purity of the intermediate acid form reduces the load on the final salt formation step, minimizing the loss of valuable material during the final isolation.
- Enhanced Supply Chain Reliability: Supply continuity is greatly enhanced by the robustness of the chemical process, which is less sensitive to minor variations in raw material quality compared to enzymatic or biological purification methods. The use of stable, shelf-stable reagents ensures that production schedules are not disrupted by the degradation of sensitive catalysts or enzymes. Additionally, the ability to produce a free-flowing crystalline solid rather than a hygroscopic or viscous mass simplifies packaging, storage, and transportation logistics. This physical stability reduces the risk of product degradation during transit and allows for more flexible inventory management strategies. The scalability of the process from kilogram to metric-ton scales ensures that suppliers can rapidly ramp up production to accommodate large volume contracts without requiring extensive process re-engineering.
- Scalability and Environmental Compliance: Environmental sustainability is a key driver for modern pharmaceutical manufacturing, and this method aligns perfectly with green chemistry principles by minimizing solvent usage and waste generation. The absence of heavy metal catalysts or toxic dialysis effluents simplifies wastewater treatment and ensures compliance with increasingly stringent environmental regulations. The process is designed for seamless scale-up, utilizing standard stirred-tank reactors and filtration equipment found in most multipurpose API manufacturing facilities. This compatibility with existing infrastructure allows for rapid technology transfer and validation, reducing the time to market for generic or biosimilar versions of the drug. The high purity profile also reduces the regulatory burden associated with impurity qualification, facilitating faster approval times for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear understanding of the process advantages over legacy methods. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this purification method control sulfoxide impurities in Sugammadex Sodium?
A: The method introduces a reducing agent (such as tris(p-methylphenyl)phosphine or zinc powder) during both the dissociation and recrystallization steps. This creates an antioxidant environment that prevents the oxidation of the critical thioether bonds into sulfoxide impurities (Formula II and III), keeping total oxidation impurities below 0.1%.
Q: Why is the organic acid-organic base system superior to traditional inorganic acid dissociation?
A: Traditional methods using strong inorganic acids often degrade the cyclodextrin substrate, leading to sticky, viscous products and glycosidic bond cleavage. The patented organic acid-organic base system operates under mild conditions (pH 3-4), maintaining the structural integrity of the cyclodextrin cage and significantly improving yield and filterability.
Q: Is this purification process suitable for large-scale industrial production?
A: Yes, unlike prior art methods relying on time-consuming dialysis (36+ hours) or expensive preparative HPLC, this method utilizes standard crystallization and filtration unit operations. It eliminates the need for large volumes of water and specialized membranes, making it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process reliability in the production of complex pharmaceutical intermediates like Sugammadex Sodium. Our technical team has extensively analyzed the advancements presented in patent CN109021147B and integrated similar high-efficiency purification strategies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality material regardless of order volume. Our facility is equipped with rigorous QC labs and stringent purity specifications that exceed industry standards, guaranteeing that every batch of Sugammadex Sodium meets the necessary criteria for safety and efficacy in neuromuscular blockade reversal applications.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your supply chain needs. By leveraging our expertise in process optimization and impurity control, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent, data-driven insights into our production capabilities and quality assurance protocols.
