Advanced Synthetic Route for Empagliflozin Intermediates: Scalability and Purity Optimization
Advanced Synthetic Route for Empagliflozin Intermediates: Scalability and Purity Optimization
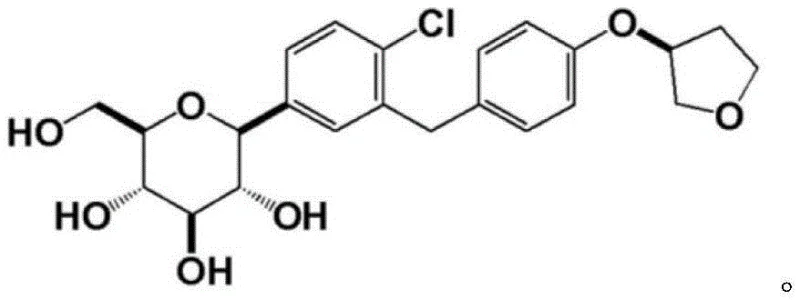
The global demand for SGLT2 inhibitors continues to surge as type 2 diabetes prevalence rises, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients (APIs) efficiently. Patent CN114213365A, published in March 2022, introduces a transformative synthetic methodology for producing the critical empagliflozin intermediate, (S)-3-(4-(5-iodo-2-chlorobenzyl)phenoxy)tetrahydrofuran. This innovation addresses long-standing bottlenecks in organic synthesis by replacing hazardous reagents and complex multi-step sequences with a streamlined, three-step protocol that operates under remarkably mild conditions. By leveraging specific catalytic systems such as BINAP/MTAD for etherification and zinc chloride/sodium borohydride for reduction, this route achieves exceptional purity levels exceeding 99% while maintaining high yields. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and economically viable production models, ensuring a reliable supply of this high-value pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
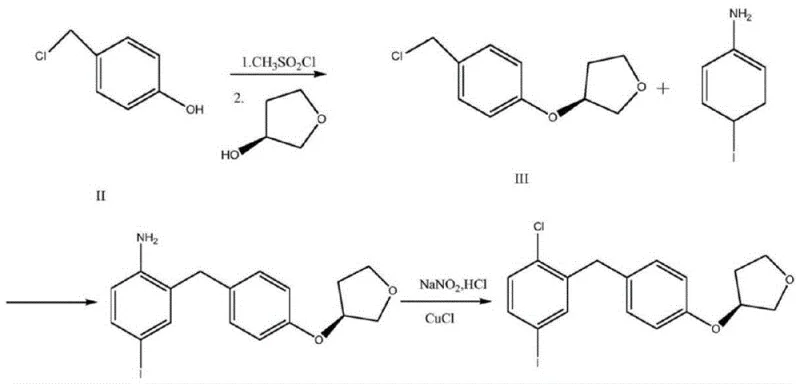
Historically, the synthesis of empagliflozin intermediates has been plagued by inefficient pathways that hinder large-scale commercialization. Prior art, such as the methods disclosed in CN107311962A and CN108178751A, often relies on starting materials like 4-fluorotoluene or 4-hydroxybenzyl chloride, which necessitate rigorous reaction conditions and cumbersome purification steps. For instance, traditional routes frequently involve diazotization reactions using sodium nitrite and hydrochloric acid followed by cuprous chloride substitution, processes that generate significant hazardous waste and pose safety risks due to the instability of diazonium salts. Furthermore, these legacy methods often suffer from low overall yields and poor stereochemical control, leading to expensive downstream purification requirements to remove regioisomers and byproducts. The reliance on harsh reagents and multiple isolation steps not only inflates production costs but also extends lead times, creating vulnerabilities in the supply chain for critical antidiabetic medications.
The Novel Approach
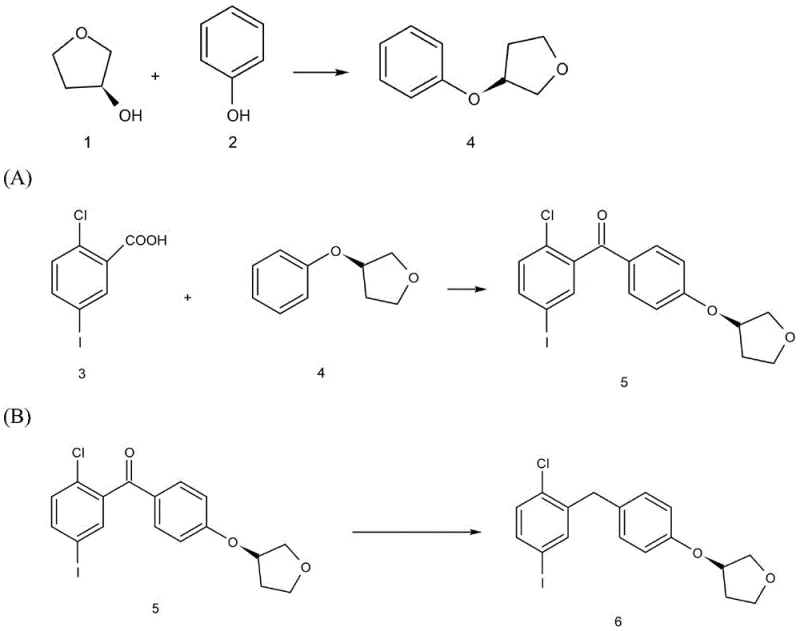
In stark contrast, the methodology outlined in patent CN114213365A offers a streamlined alternative that fundamentally reimagines the construction of the molecular scaffold. This novel approach initiates with a direct etherification between phenol and (S)-3-hydroxytetrahydrofuran, bypassing the need for halogenated precursors and toxic heavy metal catalysts typically required in nucleophilic substitutions. The subsequent acylation step utilizes p-toluenesulfonyl chloride (TsCl) as an activating agent in a mixed solvent system, facilitating a clean coupling with 2-chloro-5-iodobenzoic acid. Finally, the reduction of the ketone intermediate is achieved using a mild zinc chloride and sodium borohydride system, eliminating the need for high-pressure hydrogenation equipment. This strategic redesign of the synthetic tree results in a shorter reaction sequence with fewer unit operations, directly translating to enhanced process robustness and operational simplicity for industrial partners seeking a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into BINAP/MTAD Catalyzed Etherification and Reduction
The cornerstone of this improved synthesis lies in the sophisticated selection of catalytic systems that drive high conversion rates under ambient conditions. In the initial etherification step, the combination of BINAP (a bulky chiral bisphosphine ligand) and MTAD (an azodicarboxylate derivative) facilitates a Mitsunobu-type coupling mechanism. This interaction activates the hydroxyl group of the tetrahydrofuran ring, allowing for a stereospecific nucleophilic attack by the phenol oxygen. The steric bulk of the BINAP ligand helps to minimize side reactions and ensures the retention of the (S)-configuration, which is critical for the biological activity of the final API. Following this, the acylation step leverages the electrophilic activation of the carboxylic acid by TsCl, forming a reactive mixed anhydride or acyl sulfonate intermediate that is highly susceptible to nucleophilic attack by the phenoxy species, thereby driving the formation of the ketone linkage with high efficiency.
Furthermore, the final reduction step demonstrates a masterful application of chemoselective reducing agents to achieve the target benzyl alcohol without affecting other sensitive functional groups like the aryl iodide. The use of zinc chloride acts as a Lewis acid catalyst, coordinating with the carbonyl oxygen to increase its electrophilicity, while sodium borohydride serves as the hydride source. This synergistic effect allows the reduction to proceed rapidly at room temperature in dioxane, a solvent chosen for its ability to solubilize both organic substrates and inorganic salts. This mechanistic pathway effectively suppresses the formation of dehalogenated byproducts, a common issue in catalytic hydrogenation of aryl halides, thus ensuring the integrity of the iodine handle required for subsequent glycosylation steps in the full empagliflozin synthesis.
How to Synthesize (S)-3-(4-(5-iodo-2-chlorobenzyl)phenoxy)tetrahydrofuran Efficiently
Implementing this patented protocol requires precise control over stoichiometry and solvent quality to replicate the high yields reported in the examples. The process begins with the preparation of the ether backbone, followed by the installation of the chloro-iodo-benzoyl moiety, and concludes with the reduction to the methylene bridge. Operators must ensure that the (S)-3-hydroxytetrahydrofuran starting material is of high optical purity to maintain the stereochemical integrity throughout the sequence. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures, are provided in the guide below to assist process chemists in technology transfer.
- Perform etherification of phenol and (S)-3-hydroxytetrahydrofuran using BINAP and MTAD catalysts in THF at room temperature.
- Conduct acylation with 2-chloro-5-iodobenzoic acid and TsCl in a THF/DMSO mixed solvent system with EDTA.
- Execute carbonyl reduction using zinc chloride and sodium borohydride in dry dioxane to obtain the final benzyl intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. By eliminating the need for high-pressure hydrogenation and toxic diazotization reagents, the process significantly reduces the capital expenditure required for specialized reactor infrastructure and waste treatment facilities. The use of readily available starting materials like phenol and 2-chloro-5-iodobenzoic acid mitigates the risk of raw material shortages, ensuring a more stable and predictable supply chain for long-term production planning. Additionally, the simplified post-treatment procedures, which primarily involve filtration and solvent recovery rather than complex chromatographic separations, drastically lower the operational costs associated with labor and consumables.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in reaction steps lead to substantial cost savings in the overall manufacturing process. By operating at room temperature, the method also minimizes energy consumption for heating and cooling, further enhancing the economic viability of large-scale production. The high yields achieved in each step reduce the amount of raw material wasted, optimizing the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents like THF and dioxane ensures that the supply chain is resilient to market fluctuations affecting exotic reagents. The robustness of the reaction conditions means that production can be scaled up with minimal risk of batch failure, guaranteeing consistent delivery schedules to downstream API manufacturers. This reliability is crucial for maintaining uninterrupted production of life-saving diabetes medications.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal contaminants simplify the regulatory approval process for new drug filings, as the impurity profile is cleaner and easier to characterize. The process generates less hazardous waste, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. This compliance advantage facilitates faster regulatory clearance and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What are the key advantages of the new synthesis method in CN114213365A?
A: The method features short reaction steps, mild room temperature conditions, simple post-treatment procedures, and significantly higher product yields compared to prior art routes.
Q: What catalysts are used in the etherification step?
A: The process utilizes BINAP (binaphthyldiphenylphosphine) and MTAD (N-methyl-1,2,4-triazoline-3,5-dione) to facilitate the coupling of phenol and (S)-3-hydroxytetrahydrofuran.
Q: How is the carbonyl group reduced in the final step?
A: The reduction is achieved using zinc chloride and sodium borohydride in dry dioxane at room temperature, avoiding harsh hydrogenation conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Empagliflozin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of antidiabetic drug manufacturing. Our team of expert process chemists has extensively analyzed the methodology described in CN114213365A and possesses the technical capability to scale this pathway from laboratory benchtop to industrial tonnage. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency in your supply chain.
