Advanced Synthesis of Bromhexine Hydrochloride: Enhancing Purity and Scalability for Global Markets
Advanced Synthesis of Bromhexine Hydrochloride: Enhancing Purity and Scalability for Global Markets
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for established expectorants like bromhexine hydrochloride. Patent CN103333074A introduces a transformative production method that addresses long-standing challenges in yield optimization and product stability. By integrating a specialized condensation reaction catalyzed by glacial acetic acid with an innovative azeotropic dehydration system, this technology achieves a significant leap in quality control. The core breakthrough lies in the immediate removal of reaction-generated water using a toluene-based azeotropic agent within a custom-designed reflux apparatus. This approach not only drives the chemical equilibrium toward the desired product but also minimizes the degradation of sensitive intermediates. For global supply chains, this represents a critical advancement, offering a reliable pharmaceutical intermediates supplier pathway that consistently delivers bulk drug substances with purity exceeding 99.9%.
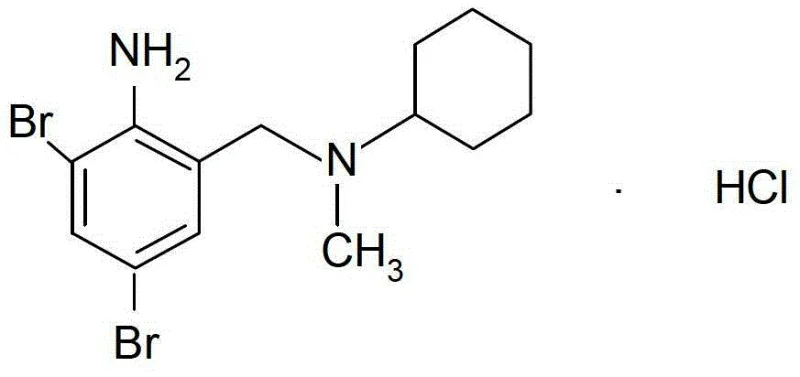
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for bromhexine hydrochloride have historically suffered from inefficiencies that compromise both cost-effectiveness and final product quality. Conventional processes often rely on multiple recrystallization steps to meet pharmacopoeia standards, leading to substantial losses in overall yield and increased consumption of energy and solvents. Furthermore, older methods frequently struggle with product stability; the active constituent content can degrade rapidly during storage due to the presence of unstable free base residues or hydrolytic impurities. The reliance on complex solvent systems, such as mixed alcohols or acetonitrile, complicates solvent recovery and increases environmental waste. These factors collectively result in a fragmented production workflow where achieving the stringent impurity limits required by modern regulatory bodies, such as the 2010 Chinese Pharmacopoeia, becomes an arduous and resource-intensive task.
The Novel Approach
The patented methodology revolutionizes this landscape by streamlining the synthesis into a highly efficient, continuous-flow-compatible sequence. The introduction of a "distillation and reflux secondary division box" allows for the real-time separation of water from the reaction mixture during the condensation phase. By maintaining a water-free environment, the reaction is pushed to completion with significantly higher conversion rates, reportedly improving yields from typical ranges of 75-80% to over 85-90%. Crucially, this method enables the production of high-quality crude products that require only a single refining step to achieve ultra-high purity. The elimination of repetitive crystallization cycles not only reduces processing time but also drastically lowers the operational expenditure associated with labor, heating, and solvent management, presenting a compelling case for cost reduction in API manufacturing.
Mechanistic Insights into Azeotropic Dehydration Condensation
The chemical elegance of this process centers on the manipulation of reaction thermodynamics through azeotropic distillation. In the condensation step, 3,5-dibromo-2-aminobenzyl alcohol reacts with N-methylcyclohexylamine in the presence of glacial acetic acid. Under standard conditions, the water produced would accumulate, potentially reversing the reaction or promoting hydrolysis. However, the addition of toluene creates a low-boiling azeotrope with water. As the mixture refluxes, the vapor rises into the specialized fractionation column where the water-rich phase is condensed and separated, while the dry toluene and amine return to the reactor. This continuous removal of the byproduct effectively locks the equilibrium in favor of the N-methyl-N-cyclohexyl-2-amino-3,5-dibromobenzylamine intermediate. This mechanistic control is vital for suppressing side reactions that typically generate colored impurities or polymeric byproducts, ensuring a cleaner reaction profile from the outset.
Following condensation, the purification mechanism relies on precise pH control and selective solubility. The crude base is salified with hydrochloric acid in an alcoholic medium, where the pH is rigorously adjusted to between 2 and 3. This specific acidity range is critical; it ensures the complete protonation of the amine groups, converting any residual free base—which is thermally unstable—into the stable hydrochloride salt. Subsequent decolorization with activated carbon in ethanol removes trace organic impurities without dissolving the product excessively. The synergy between the initial high-purity condensation and this targeted acid-base refinement results in a final product with exceptional stability, as evidenced by accelerated testing showing negligible impurity growth over 24 months. This level of control is essential for producing high-purity bromhexine hydrochloride suitable for sensitive respiratory formulations.
How to Synthesize Bromhexine Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for industrial implementation, emphasizing the importance of equipment configuration alongside chemical reagents. The process begins with the charging of N-methylcyclohexylamine and the dibromo-alcohol precursor into a heated reactor, followed by the controlled addition of the acetic acid catalyst and toluene entrainer. The reaction is maintained at reflux temperatures between 170°C and 180°C until water evolution ceases, indicating completion. The subsequent salification and purification steps utilize standard unit operations but are optimized by the high quality of the incoming crude material. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios required for GMP-compliant production, please refer to the standardized technical guide below.
- Conduct condensation between 3,5-dibromo-2-aminobenzyl alcohol and N-methylcyclohexylamine using glacial acetic acid and toluene for azeotropic water removal.
- Perform salification of the resulting base with hydrochloric acid in an alcoholic solvent system under controlled pH conditions.
- Execute a single-step decolorization and refinement in ethanol with activated carbon to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical specifications. The primary advantage lies in the drastic simplification of the downstream processing workflow. By achieving high purity after a single refinement step, manufacturers can significantly reduce the cycle time per batch, thereby increasing overall plant throughput without requiring additional capital investment in new reactors. This efficiency gain translates directly into improved supply continuity, allowing suppliers to respond more agilely to market demand fluctuations. Furthermore, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, mitigating the risk of production stoppages due to compliance issues and ensuring a more sustainable sourcing model for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization cycles represents a major driver for operational cost savings. Traditional methods often lose substantial material mass during repeated washing and filtering; this novel approach retains a much higher percentage of the theoretical yield. Additionally, the ability to recycle the toluene azeotropic agent and the alcoholic solvents used in purification further lowers the variable cost per kilogram. By minimizing the consumption of auxiliary materials like activated carbon and reducing energy loads associated with extended heating and drying times, the overall cost of goods sold (COGS) is optimized, providing a competitive pricing structure for bulk purchasers.
- Enhanced Supply Chain Reliability: Consistency is the cornerstone of a reliable pharmaceutical intermediates supplier. The robust nature of this process, which tolerates minor variations better than fragile multi-step syntheses, ensures batch-to-batch uniformity. The stability data indicates that the product maintains its potency and purity profile over extended storage periods, reducing the risk of inventory write-offs due to expiration or degradation. This reliability allows downstream formulators to plan their production schedules with greater confidence, knowing that the quality of the incoming active ingredient will remain stable throughout the shelf life of their finished respiratory medications.
- Scalability and Environmental Compliance: The patent explicitly describes embodiments using 300L to 1000L reactors, demonstrating that the chemistry is not limited to laboratory scales but is ready for commercial scale-up of complex expectorants. The closed-loop nature of the azeotropic distillation system minimizes volatile organic compound (VOC) emissions, addressing a key concern in modern chemical manufacturing. The reduced generation of mother liquor waste simplifies effluent treatment requirements, lowering the environmental footprint of the facility. This scalability ensures that suppliers can ramp up production volumes to meet global demand surges without compromising on quality or regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of bromhexine hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains and formulation pipelines.
Q: How does the new production method improve the stability of Bromhexine Hydrochloride?
A: The method utilizes a specialized distillation setup to remove water continuously during condensation, preventing hydrolysis and side reactions. Additionally, strict pH control (2-3) during purification ensures complete salt formation, eliminating unstable free base residues.
Q: What are the advantages of the 'distillation and reflux secondary division box' equipment?
A: This specialized reactor design allows for the efficient separation of the water-toluene azeotrope while returning dry organic solvents to the reaction vessel. This drives the equilibrium forward, significantly increasing yield and reducing the formation of polymeric impurities common in traditional batch processes.
Q: Can this synthesis route be scaled for industrial API manufacturing?
A: Yes, the patent explicitly details embodiments using 300L to 1000L reactors, demonstrating industrial viability. The elimination of multiple recrystallization steps simplifies the workflow, making it highly suitable for large-scale commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromhexine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and rigorous quality oversight. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this advanced synthesis method are fully realized in every batch we deliver. Our facilities are equipped with state-of-the-art stainless steel reactors and precision distillation columns capable of replicating the azeotropic dehydration conditions described in the patent. We maintain stringent purity specifications and operate rigorous QC labs to verify that every shipment meets the highest international standards for pharmaceutical intermediates, guaranteeing safety and efficacy for your final drug products.
We invite you to collaborate with us to optimize your supply chain for respiratory therapeutics. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient synthesis route can improve your margins. We encourage potential partners to contact our technical procurement team to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a stable, high-quality supply of bromhexine hydrochloride that supports your commitment to patient health and operational excellence.
