Advanced Reductive Amination Strategy for High-Purity Bromhexine Hydrochloride Manufacturing
Advanced Reductive Amination Strategy for High-Purity Bromhexine Hydrochloride Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high yield with operational safety, particularly for mucolytic agents like Bromhexine Hydrochloride. A pivotal advancement in this domain is detailed in patent CN112194585A, which discloses a novel synthetic method utilizing a palladium-catalyzed reductive amination strategy. This approach fundamentally shifts the paradigm from traditional, hazardous chlorination chemistries to a more sustainable and efficient coupling of 2-amino-3,5-dibromobenzaldehyde and N-methylcyclohexylamine. By leveraging formic acid as a hydrogen donor in the presence of a Pd/C catalyst, this technology achieves exceptional conversion rates while circumventing the generation of toxic acid gases. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for this critical respiratory therapeutic intermediate, ensuring both cost efficiency and regulatory compliance in API manufacturing.
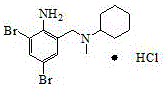
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bromhexine Hydrochloride has relied heavily on multi-step sequences involving aggressive halogenating agents such as thionyl chloride or triphosgene. As highlighted in prior art like CN 102531922B and CN 102617359A, these conventional pathways typically necessitate the reduction of the starting aldehyde to a benzyl alcohol, followed by chlorination to activate the benzylic position for nucleophilic substitution. This reliance on thionyl chloride introduces severe operational challenges, including the evolution of large volumes of corrosive hydrogen chloride and sulfur dioxide gases, which demand specialized scrubbing systems and accelerate the degradation of reactor linings. Furthermore, these legacy processes often suffer from mediocre overall yields, frequently hovering around 62% to 80%, due to the formation of difficult-to-remove impurities during the harsh chlorination and subsequent amination steps. The economic burden of waste treatment and equipment replacement renders these methods increasingly untenable in a modern, cost-sensitive manufacturing environment.
The Novel Approach
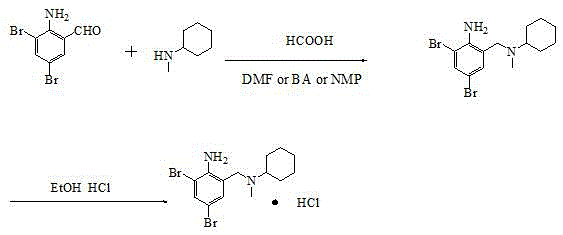
In stark contrast, the methodology presented in CN112194585A streamlines the synthesis into a direct reductive amination, effectively bypassing the isolation of unstable alcohol or chloride intermediates. By reacting 2-amino-3,5-dibromobenzaldehyde directly with N-methylcyclohexylamine in solvents such as butyl acetate, DMF, or NMP, the process forms an imine intermediate in situ which is immediately reduced by formic acid. This "one-pot" philosophy not only drastically reduces solvent consumption and processing time but also elevates the isolated yield to an impressive 91% to 92%. The elimination of gaseous byproducts like HCl during the main reaction phase significantly lowers the environmental footprint and simplifies the engineering controls required for production. This transition from a stepwise, hazard-intensive protocol to a convergent, catalytic process exemplifies the kind of process intensification that drives down the cost of goods sold (COGS) for high-volume pharmaceutical intermediates.
Mechanistic Insights into Pd/C-Catalyzed Reductive Amination
The core of this technological breakthrough lies in the efficient transfer hydrogenation mechanism facilitated by the palladium on carbon (Pd/C) catalyst. In this system, the primary amine group of the starting material and the secondary amine (N-methylcyclohexylamine) coexist, yet the reaction exhibits remarkable chemoselectivity for the aldehyde functionality. Upon heating to 100-110°C, the aldehyde condenses with the secondary amine to generate an iminium ion or Schiff base intermediate. Formic acid then serves as the hydride source, decomposing on the surface of the palladium catalyst to release reactive hydrogen species that reduce the C=N double bond to a stable C-N single bond. This catalytic cycle avoids the need for high-pressure hydrogen gas cylinders, thereby enhancing plant safety profiles while maintaining rapid reaction kinetics. The choice of solvent, particularly polar aprotic options like NMP or DMF, further stabilizes the transition states, ensuring complete conversion of the starting materials within a 5-hour window.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic substitution routes. Traditional methods involving benzyl chlorides are prone to dialkylation side reactions or elimination pathways that generate olefinic impurities, which are notoriously difficult to separate from the final API. The reductive amination pathway, however, is inherently self-limiting; once the tertiary amine is formed, it cannot undergo further alkylation under these conditions. Additionally, the mild acidic environment provided by formic acid helps suppress the oxidation of the sensitive aniline moiety, preserving the integrity of the 3,5-dibromo substitution pattern. The result is a crude product profile that is exceptionally clean, allowing for a straightforward recrystallization from methanol-acetone mixtures to achieve HPLC purities exceeding 99.1%. This level of purity is critical for meeting the stringent specifications required by global pharmacopoeias for respiratory drug substances.
How to Synthesize Bromhexine Hydrochloride Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the catalytic system. The process begins with the charging of stoichiometric amounts of 2-amino-3,5-dibromobenzaldehyde and a slight excess of N-methylcyclohexylamine into a reactor containing the chosen solvent, typically butyl acetate for ease of recovery. The addition of the Pd/C catalyst (5-10% loading) initiates the cycle upon heating, followed by the controlled dropwise addition of anhydrous formic acid to manage the exotherm and gas evolution. Following the reaction completion, the catalyst is removed via filtration, and the filtrate is concentrated to remove the solvent before the crucial salification step. While the general workflow is robust, adherence to specific temperature gradients and addition rates is essential for reproducibility. For the detailed standardized operating procedures and specific quality control checkpoints, please refer to the technical guide below.
- Charge 2-amino-3,5-dibromobenzaldehyde, N-methylcyclohexylamine, and Pd/C catalyst into a reactor with solvent (e.g., butyl acetate) and heat to 100°C.
- Dropwise add anhydrous formic acid over 1.5 hours, maintaining temperature at 100-110°C for 5 hours to complete the reductive amination.
- Filter off catalyst, concentrate filtrate, treat with 30% HCl in ethanol for salt formation, and recrystallize from methanol-acetone to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this reductive amination technology translates into tangible strategic benefits beyond mere chemical elegance. The most immediate impact is observed in the reduction of raw material volatility and equipment depreciation costs associated with corrosive reagents. By removing thionyl chloride and triphosgene from the bill of materials, manufacturers can significantly extend the lifecycle of their stainless steel reactors and reduce the frequency of costly maintenance shutdowns. Furthermore, the simplified workup procedure, which avoids complex aqueous extractions and multiple intermediate isolations, leads to a substantial reduction in cycle time. This efficiency gain allows for increased batch turnover rates, effectively enhancing the overall capacity of existing production facilities without the need for capital-intensive expansion projects.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, driven primarily by the dramatic improvement in mass efficiency. Achieving yields in the range of 91% compared to the historical baseline of roughly 62% means that nearly one-third more product is generated from the same amount of starting material. This increase in atom economy directly lowers the variable cost per kilogram of the active pharmaceutical ingredient. Additionally, the use of formic acid as a reducing agent is considerably more economical than purchasing specialized reducing agents or managing the disposal costs of heavy metal waste streams often associated with alternative reduction methods. The cumulative effect is a significantly leaner cost structure that provides a competitive edge in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely available from multiple global vendors. Unlike specialized chlorinating agents that may face logistical bottlenecks or regulatory shipping restrictions due to their hazardous classification, formic acid and Pd/C catalysts are standard inventory items for most fine chemical plants. This ubiquity reduces the risk of production stoppages caused by raw material shortages. Moreover, the robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—minimizes the risk of batch failures due to equipment malfunction. This reliability ensures consistent delivery schedules to downstream API manufacturers, fostering stronger long-term partnerships and reducing the need for excessive safety stock holdings.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this process aligns perfectly with modern green chemistry principles. The absence of sulfur-containing byproducts eliminates the need for complex caustic scrubbing systems required to neutralize sulfur dioxide emissions. The solvent systems employed, such as butyl acetate, are relatively benign and can be efficiently recovered and recycled through standard distillation trains, minimizing liquid waste discharge. This reduced environmental burden simplifies the permitting process for scale-up and ensures compliance with increasingly stringent international environmental regulations. Consequently, the technology is highly scalable, capable of being transferred from pilot kilolab scales to multi-ton commercial production with minimal process re-engineering, ensuring a smooth path to market for new drug filings.
Frequently Asked Questions (FAQ)
Understanding the nuances of this synthesis method is vital for stakeholders evaluating its integration into their supply networks. The following questions address common technical and commercial inquiries regarding the feasibility and performance of this reductive amination route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities. By clarifying these points, we aim to facilitate informed decision-making for R&D and operations teams considering the adoption of this advanced manufacturing process.
Q: What are the key advantages of the Pd/C catalyzed route over traditional thionyl chloride methods?
A: The Pd/C catalyzed reductive amination eliminates the use of highly corrosive thionyl chloride and triphosgene, significantly reducing equipment maintenance costs and environmental hazards while improving yields from ~62% to over 91%.
Q: How does this synthesis method impact impurity profiles?
A: By avoiding harsh chlorinating agents and utilizing a selective catalytic hydrogen transfer, the process minimizes side reactions, achieving HPLC purity levels exceeding 99.1% without complex purification steps.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method uses common solvents like butyl acetate or DMF and operates at moderate temperatures (100-110°C), making it highly scalable with standard stainless steel reactors and reducing lead times for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromhexine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Pd/C catalyzed reductive amination are fully realized in a GMP-compliant setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Bromhexine Hydrochloride meets the highest global standards. Our commitment to quality assurance extends beyond the final product, encompassing a thorough validation of the entire supply chain to mitigate risks and ensure continuity of supply for our partners.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their Bromhexine supply chain through this innovative technology. By leveraging our process development capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive efficiency and sustainability in the production of essential respiratory therapeutics, securing a competitive advantage in the global marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
