Advanced One-Pot Synthesis of Alpha-Cyano Nitro Phenylacrylic Acid Derivatives for Commercial API Production
Advanced One-Pot Synthesis of Alpha-Cyano Nitro Phenylacrylic Acid Derivatives for Commercial API Production
The development of efficient synthetic routes for complex pharmaceutical intermediates is a critical driver in reducing the overall cost of goods for oncology therapeutics. Patent CN102558098A discloses a robust and economically viable preparation method for α-cyano-3-nitro-4-substituted aminophenylacrylic acid derivatives, which serve as pivotal building blocks for the synthesis of Epidermal Growth Factor Receptor inhibitors (EGFRis) and Histone Deacetylase inhibitors (HDACis). These compound classes are essential in modern cancer treatment regimens, necessitating reliable supply chains capable of delivering high-purity materials. The disclosed technology represents a significant departure from legacy methodologies by employing a tandem one-pot strategy that merges nucleophilic aromatic substitution with Knoevenagel condensation. This approach not only streamlines the operational workflow but also mitigates the reliance on precious metal catalysts, thereby addressing key pain points for procurement and supply chain stakeholders in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these valuable intermediates has been plagued by inefficient multi-step protocols that impose substantial burdens on manufacturing costs and environmental compliance. Traditional literature methods, such as those cited in Chinese Chemical Letters (2010), typically involve a discrete two-stage process. The initial stage requires the coupling of a secondary amine with 3-nitro-4-chlorobenzaldehyde using expensive Palladium catalysts and bipyridine ligands to generate the amino-aldehyde intermediate. As illustrated in the prior art reaction scheme below, this step often suffers from moderate yields, reported around 54%, and necessitates rigorous purification to remove trace heavy metals which are strictly regulated in pharmaceutical products.
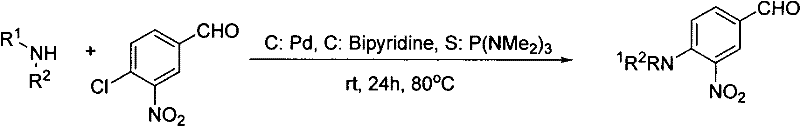
Furthermore, alternative conventional routes utilizing strong inorganic bases like Cesium Carbonate in polar aprotic solvents such as DMSO have demonstrated even lower efficiency, with yields dropping to approximately 43% under heated conditions. These legacy processes are characterized by long reaction times, the generation of significant hazardous waste streams, and the instability of the isolated intermediate aldehydes, which can degrade before the subsequent condensation step. For a procurement manager, these factors translate into volatile pricing, extended lead times, and complex quality control requirements to ensure the final Active Pharmaceutical Ingredient (API) meets stringent impurity specifications regarding residual solvents and metal content.
The Novel Approach
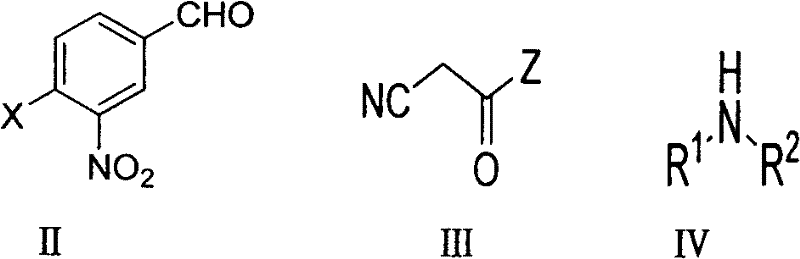
In stark contrast, the methodology outlined in patent CN102558098A introduces a convergent one-pot synthesis that dramatically simplifies the production landscape. By reacting 3-nitro-4-halobenzaldehyde directly with a secondary amine and a cyanoacetate or cyanoacetamide derivative in the presence of a base, the process achieves both the substitution of the halogen and the formation of the acrylic double bond in a single operational unit. This telescoped approach eliminates the need to isolate the unstable amino-aldehyde intermediate, thereby preventing degradation losses and reducing the total number of processing steps. The reaction proceeds smoothly in common organic solvents like ethanol or even green ionic liquids at moderate temperatures ranging from 50°C to 100°C. This innovation offers a reliable pharmaceutical intermediate supplier with the ability to offer a more stable and cost-competitive product, as the elimination of the Palladium catalyst removes a major cost driver and supply chain bottleneck associated with precious metal availability.
Mechanistic Insights into Tandem SNAr and Knoevenagel Condensation
The success of this one-pot transformation relies on the precise orchestration of electronic effects within the substrate and the dual role of the amine reagent. The reaction initiates with a Nucleophilic Aromatic Substitution (SNAr) where the secondary amine attacks the electron-deficient aromatic ring of the 3-nitro-4-halobenzaldehyde. The presence of the strongly electron-withdrawing nitro group at the ortho-position relative to the leaving halogen significantly activates the ring towards nucleophilic attack, facilitating the displacement of the chloride or bromide ion without the need for transition metal catalysis. Simultaneously or subsequently, the basic nature of the secondary amine, or an added base, deprotonates the active methylene group of the cyanoacetate component. This generates a nucleophilic carbanion that undergoes a Knoevenagel condensation with the aldehyde moiety of the same molecule or the newly formed amino-aldehyde species in situ.
From an impurity control perspective, this mechanism is advantageous because it minimizes the residence time of reactive intermediates that could otherwise participate in side reactions such as polymerization or oxidation. The thermodynamic drive towards the formation of the conjugated E-isomer ensures high stereochemical purity, which is critical for downstream biological activity. The process tolerates a wide range of substituents on the amine, including cyclic structures like piperidine and morpholine, as well as acyclic chains, demonstrating remarkable chemoselectivity. For R&D directors, understanding this mechanism confirms that the process is robust against minor variations in raw material quality, provided the stoichiometry is maintained, ensuring consistent batch-to-batch reproducibility essential for regulatory filings and commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize E-Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to maximize yield and purity. The general protocol involves dissolving the halogenated benzaldehyde starting material in a solvent such as ethanol, followed by the sequential addition of the cyanoacetate ester and the chosen secondary amine. The reaction mixture is then heated to reflux or maintained at a controlled temperature between 50°C and 100°C depending on the solvent boiling point and reactivity of the specific amine. Progress is typically monitored via Thin Layer Chromatography (TLC) until the starting aldehyde is fully consumed. Upon completion, the product often precipitates directly from the reaction mixture upon cooling or concentration, allowing for isolation via simple filtration and recrystallization, bypassing the need for resource-intensive column chromatography in most cases.
- Dissolve 3-nitro-4-halobenzaldehyde in a suitable organic solvent such as ethanol or DMF within a reaction vessel.
- Add cyanoacetate or cyanoacetamide and a secondary amine (such as piperidine or morpholine) to the mixture under alkaline conditions.
- Heat the reaction mixture to 50-100°C until completion, monitored by TLC, followed by crystallization or filtration to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the sourcing of critical raw materials, the adoption of this patented synthesis method offers transformative benefits that extend beyond mere chemical yield. The shift away from Palladium-catalyzed processes fundamentally alters the cost structure of manufacturing these intermediates. By removing the requirement for expensive noble metals and specialized ligands, the direct material costs are significantly reduced. Furthermore, the simplification of the workup procedure—often requiring only recrystallization instead of complex chromatographic purification—drastically lowers the consumption of silica gel and elution solvents. This reduction in auxiliary materials contributes to substantial cost savings in manufacturing overhead and waste disposal fees, aligning with modern green chemistry initiatives and sustainability goals that are increasingly important to corporate procurement strategies.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts represents a direct saving on high-value input costs, while the one-pot nature of the reaction reduces energy consumption by cutting down on heating and cooling cycles associated with multi-step isolations. The ability to use commodity solvents like ethanol instead of specialized anhydrous or degassed solvents further enhances the economic viability of the process. Additionally, the higher overall yields reported in the patent examples, reaching up to 85% for certain morpholine derivatives compared to the 43-54% of prior art, mean that less raw material is required to produce the same amount of finished goods, effectively lowering the cost per kilogram of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as substituted benzaldehydes and common cyclic amines mitigates the risk of supply disruptions often associated with specialized catalysts or reagents. The robustness of the reaction conditions, which do not require inert atmosphere protection (nitrogen or argon), simplifies the equipment requirements and allows for production in standard glass-lined or stainless steel reactors. This operational simplicity ensures that production schedules are less prone to delays caused by equipment downtime or the need for specialized maintenance, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a steady flow of materials to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable and the absence of hazardous reagents that pose significant safety risks at large volumes. The reduction in solvent usage and the avoidance of heavy metal waste streams simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. For supply chain heads, this means easier validation of contract manufacturing organizations (CMOs) and a lower risk of regulatory non-compliance. The ability to recycle solvents like ethanol further enhances the environmental profile, making the supply chain more resilient to tightening environmental regulations and carbon footprint targets imposed by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects detailed in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or production partnerships. Understanding these details is crucial for assessing the feasibility of integrating this method into existing manufacturing portfolios.
Q: What are the primary advantages of this one-pot method over traditional multi-step synthesis?
A: The primary advantage is the elimination of expensive transition metal catalysts like Palladium and the reduction of unit operations. By combining the nucleophilic substitution and Knoevenagel condensation into a single pot, the process significantly reduces solvent usage, processing time, and waste generation compared to the stepwise approaches described in prior art.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the method is highly scalable. It utilizes common organic solvents like ethanol and avoids sensitive reagents that require inert gas protection. The workup typically involves simple recrystallization rather than complex column chromatography, making it ideal for large-scale commercial manufacturing of pharmaceutical intermediates.
Q: What types of amines are compatible with this reaction pathway?
A: The process demonstrates broad substrate scope, accommodating both acyclic secondary amines (like diethylamine) and various cyclic amines including piperidine, morpholine, and methylpiperazine. This flexibility allows for the rapid generation of diverse derivative libraries for drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyano Nitro Phenylacrylic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive landscape of oncology drug development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with international standards. We are committed to delivering high-purity compounds that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against comprehensive analytical profiles. Our infrastructure is designed to support the complex chemical transformations required for advanced intermediates, guaranteeing supply continuity for your critical drug programs.
We invite you to engage with our technical procurement team to discuss how this innovative one-pot synthesis can be leveraged to optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how adopting this route can impact your overall project economics. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique molecular requirements, ensuring that your development timelines are met with the highest quality materials available in the market.
