Advanced One-Pot Synthesis of Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives for Commercial Scale
Introduction to Next-Generation Intermediate Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for critical oncology intermediates, particularly those serving as precursors for Epidermal Growth Factor Receptor (EGFR) and Histone Deacetylase (HDAC) inhibitors. A significant technological breakthrough in this domain is detailed in patent CN102558098A, which discloses a highly efficient preparation method for α-cyano-3-nitro-4-substituted aminophenylacrylic acid derivatives. This innovation addresses long-standing inefficiencies in the production of E-α-cyano-3-nitro-4-aminocinnamic acid ethyl esters and related amides. By transitioning from traditional multi-step protocols involving precious metal catalysis to a streamlined one-pot condensation and substitution strategy, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks. The method leverages readily available 3-nitro-4-halobenzaldehydes, secondary amines, and cyanoacetates to achieve high yields under mild alkaline conditions, fundamentally altering the cost and complexity landscape for these high-value chemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these critical nitro-substituted cinnamic acid derivatives has been plagued by economic and operational inefficiencies. Prior art methodologies often necessitated a stepwise approach, beginning with the nucleophilic aromatic substitution of the halogen on the benzaldehyde ring using expensive palladium catalysts and bipyridine ligands. As illustrated in the reaction scheme below, this traditional pathway not only incurred high raw material costs due to the reliance on precious metals but also suffered from moderate yields, typically hovering around 54%. Furthermore, alternative methods utilizing cesium carbonate in DMSO yielded even lower conversion rates, approximately 43%, while requiring harsh thermal conditions. These legacy processes introduced significant supply chain vulnerabilities, including the need for rigorous heavy metal removal steps to meet stringent pharmaceutical purity specifications, thereby extending production lead times and increasing waste generation.
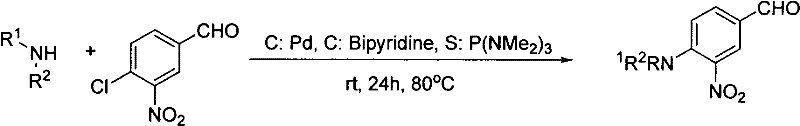
The Novel Approach
In stark contrast, the novel methodology described in the patent data eliminates the dependency on transition metal catalysts entirely. By employing a direct one-pot reaction where the nucleophilic substitution and Knoevenagel condensation occur concurrently or sequentially in a single vessel, the process achieves remarkable efficiency. The reaction utilizes basic conditions to facilitate the displacement of the halogen atom by the secondary amine while simultaneously driving the condensation with the active methylene compound. This integrated approach has demonstrated yields as high as 85% in optimized examples, representing a substantial improvement over the 43-56% range of previous techniques. The elimination of the isolation of unstable intermediate aldehydes further enhances the overall process mass intensity, reducing solvent consumption and simplifying the downstream purification workflow, which is a critical factor for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Mediated Tandem Reaction
The core of this technological advancement lies in the synergistic interaction between the nucleophilic secondary amine and the electron-deficient aromatic ring activated by the nitro group. Under alkaline conditions, the secondary amine attacks the carbon bearing the halogen substituent in a nucleophilic aromatic substitution (SNAr) mechanism. Simultaneously, the base deprotonates the active methylene group of the cyanoacetate or cyanoacetamide, generating a carbanion that undergoes Knoevenagel condensation with the aldehyde moiety. This tandem sequence is highly sensitive to the electronic nature of the substituents, yet the patent data confirms its robustness across a wide range of substrates, including chloro and bromo benzaldehydes. The reaction proceeds efficiently at temperatures between 50°C and 100°C, avoiding the extreme thermal stress that often leads to decomposition in sensitive nitro-compounds. This mechanistic elegance ensures that the formation of the carbon-carbon double bond and the carbon-nitrogen bond are harmonized, minimizing the formation of side products such as hydrolysis byproducts or polymerization species.
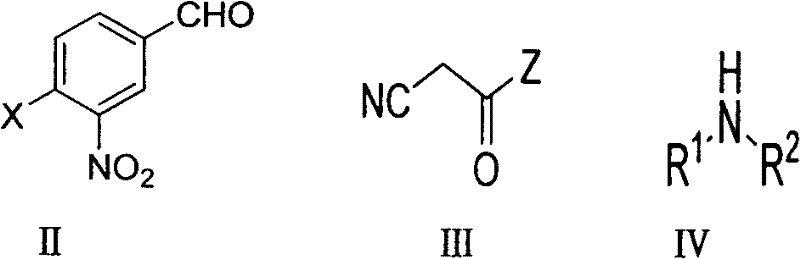
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity pharmaceutical intermediates. The absence of palladium residues removes a major class of genotoxic impurities that require costly scavenging resins or activated carbon treatments. Furthermore, the reaction byproducts are primarily inorganic salts and water, which can be easily removed during the aqueous workup or recrystallization steps described in the examples. The patent highlights that most crude products can be purified simply by ethanol recrystallization, bypassing the need for resource-intensive column chromatography which is often a bottleneck in scale-up. This simplicity in purification directly correlates to improved batch-to-batch consistency and reduced variability in the final impurity profile, a key metric for R&D directors evaluating process feasibility for regulatory filing.
How to Synthesize E-α-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives Efficiently
The operational simplicity of this synthesis route makes it highly attractive for both laboratory optimization and industrial implementation. The process begins by dissolving the 3-nitro-4-halobenzaldehyde in a selected solvent system, followed by the addition of the cyanoacetate derivative and the secondary amine in specific molar ratios ranging from 1:1 to 1:2.5 relative to the aldehyde. The mixture is then heated to reflux, with reaction progress monitored via thin-layer chromatography (TLC) using standard petroleum ether and ethyl acetate mobile phases. Upon completion, the solvent is removed, and the product is isolated through crystallization. For a comprehensive understanding of the specific parameters, reagents, and safety considerations required for execution, please refer to the detailed standardized synthesis guide provided below.
- Dissolve 3-nitro-4-halobenzaldehyde in a suitable organic solvent such as ethanol or DMF within a reaction vessel equipped with stirring.
- Add stoichiometric amounts of cyanoacetate (or cyanoacetamide) and the desired secondary amine (e.g., piperidine, morpholine) to the reaction mixture.
- Heat the mixture to reflux (50-100°C) until TLC indicates completion, then isolate the product via crystallization or standard purification methods.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical yield. The primary advantage is the drastic simplification of the raw material portfolio. By removing the requirement for palladium catalysts and specialized ligands, the supply chain becomes less susceptible to the volatility of precious metal markets and geopolitical constraints on rare earth elements. Additionally, the use of commodity chemicals like ethanol, piperidine, and morpholine ensures a stable and diversified supplier base, enhancing supply continuity. The ability to utilize ionic liquids as alternative solvents also opens avenues for green chemistry initiatives, potentially reducing waste disposal costs and aligning with corporate sustainability goals without compromising output quality.
- Cost Reduction in Manufacturing: The economic impact of eliminating transition metal catalysts cannot be overstated. Palladium and bipyridine represent significant cost centers in traditional synthesis, and their removal drastically lowers the Bill of Materials (BOM). Furthermore, the high yields achieved (up to 85%) mean less raw material is wasted per kilogram of finished product. The simplified purification process, which often relies on recrystallization rather than chromatography, reduces solvent usage and labor hours, leading to substantial cost savings in the overall manufacturing overhead. These efficiencies allow for more competitive pricing structures in the final API supply chain.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply reliability. Since the process does not require inert gas protection (such as nitrogen or argon blankets) or strictly anhydrous environments, the risk of batch failure due to atmospheric exposure is minimized. This tolerance to ambient conditions simplifies reactor requirements and allows for faster turnaround times between batches. The use of widely available solvents like ethanol and DMF ensures that production is not halted by shortages of specialized reagents, providing a resilient foundation for long-term supply agreements.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the exothermic nature of the condensation and the stability of the intermediates. The absence of heavy metals simplifies wastewater treatment and effluent management, reducing the environmental footprint of the manufacturing site. The ability to recycle solvents like ethanol further enhances the environmental profile. This alignment with environmental, social, and governance (ESG) criteria is increasingly important for multinational corporations seeking compliant and sustainable partners for their complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits and chemical scope for potential partners. Understanding these nuances is essential for integrating this method into existing production workflows and assessing its fit for specific project requirements.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, unlike conventional methods that rely on Palladium (Pd) and bipyridine ligands, this novel process operates under base-mediated conditions without transition metals, significantly reducing raw material costs and eliminating heavy metal residue concerns.
Q: What solvents are compatible with this one-pot reaction?
A: The process demonstrates high versatility, functioning effectively in common organic solvents like ethanol, acetonitrile, and DMF, as well as in ionic liquids such as [bmIm]OH, allowing for optimization based on environmental and cost requirements.
Q: Can this method be scaled for industrial production of EGFR inhibitor intermediates?
A: Yes, the reaction conditions are mild (50-100°C) and do not require inert gas protection or complex anhydrous environments, making it highly suitable for large-scale commercial manufacturing with robust safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of E-α-cyano-3-nitro-4-aminocinnamic acid derivatives meets the exacting standards required for GMP manufacturing. Our infrastructure is designed to handle the specific solvent systems and reaction conditions outlined in this advanced patent, delivering consistency and reliability.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive pharmaceutical landscape.
