Advanced One-Pot Synthesis of Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives for Commercial Scale-Up
Introduction to Patent CN102558098A
The pharmaceutical industry constantly seeks more efficient pathways to access complex heterocyclic scaffolds, particularly those serving as critical building blocks for kinase inhibitors. Patent CN102558098A introduces a robust and economically viable preparation method for α-cyano-3-nitro-4-substituted aminophenylacrylic acid derivatives. These compounds are not merely academic curiosities; they serve as indispensable intermediates in the total synthesis of potent anticancer agents targeting EGFR and HDAC enzymes. The disclosed technology represents a paradigm shift from multi-step, metal-dependent processes to a streamlined one-pot tandem reaction. By leveraging the intrinsic reactivity of 3-nitro-4-halobenzaldehydes, this method achieves high conversion rates under relatively mild thermal conditions, typically between 50°C and 100°C. For R&D directors and process chemists, this patent offers a blueprint for simplifying the supply chain of high-value oncology intermediates, ensuring that the transition from laboratory discovery to commercial manufacturing is both seamless and cost-effective.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for constructing this specific chemical architecture were fraught with inefficiencies and economic burdens that hindered large-scale adoption. Traditional routes often relied on a stepwise approach, necessitating the initial formation of the amine-substituted benzaldehyde before proceeding to the condensation step. A prominent example involved the use of palladium catalysts coupled with bipyridine ligands to facilitate the amination of the chloro-substituted precursor. 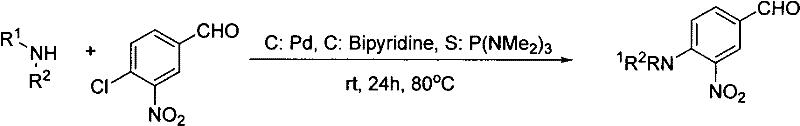 As illustrated in the reaction scheme above, this reliance on precious metals introduced significant cost volatility and created a major bottleneck in purification, as removing trace palladium to meet strict ICH Q3D guidelines for elemental impurities requires additional, costly scavenging steps. Furthermore, alternative methods utilizing cesium carbonate in DMSO suffered from moderate yields, often hovering around 43%, and required harsh conditions that could compromise the stability of sensitive functional groups. These legacy processes were not only environmentally taxing due to solvent waste but also operationally complex, demanding rigorous exclusion of moisture and oxygen to maintain catalyst activity.
As illustrated in the reaction scheme above, this reliance on precious metals introduced significant cost volatility and created a major bottleneck in purification, as removing trace palladium to meet strict ICH Q3D guidelines for elemental impurities requires additional, costly scavenging steps. Furthermore, alternative methods utilizing cesium carbonate in DMSO suffered from moderate yields, often hovering around 43%, and required harsh conditions that could compromise the stability of sensitive functional groups. These legacy processes were not only environmentally taxing due to solvent waste but also operationally complex, demanding rigorous exclusion of moisture and oxygen to maintain catalyst activity.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a convergent one-pot strategy that dramatically simplifies the synthetic topology. By combining 3-nitro-4-halobenzaldehyde, a secondary amine, and a cyanoacetate derivative simultaneously in a single reaction vessel, the process eliminates the need for isolating unstable intermediates.  The reaction proceeds through a cascade mechanism where the initial Knoevenagel condensation is followed rapidly by a nucleophilic aromatic substitution (SNAr), driven by the strong electron-withdrawing nature of the nitro group. This approach allows for the use of inexpensive, commodity-grade solvents such as ethanol, which are far easier to recover and recycle compared to high-boiling polar aprotic solvents like DMSO. The elimination of transition metal catalysts not only reduces the direct cost of goods sold (COGS) but also simplifies the regulatory filing process by removing the need for extensive heavy metal validation data. This streamlined workflow exemplifies how modern process chemistry can achieve superior outcomes through mechanistic understanding rather than brute-force reagent usage.
The reaction proceeds through a cascade mechanism where the initial Knoevenagel condensation is followed rapidly by a nucleophilic aromatic substitution (SNAr), driven by the strong electron-withdrawing nature of the nitro group. This approach allows for the use of inexpensive, commodity-grade solvents such as ethanol, which are far easier to recover and recycle compared to high-boiling polar aprotic solvents like DMSO. The elimination of transition metal catalysts not only reduces the direct cost of goods sold (COGS) but also simplifies the regulatory filing process by removing the need for extensive heavy metal validation data. This streamlined workflow exemplifies how modern process chemistry can achieve superior outcomes through mechanistic understanding rather than brute-force reagent usage.
Mechanistic Insights into Tandem Knoevenagel-SNAr Reaction
The success of this transformation hinges on the precise orchestration of two distinct chemical events within the same reaction medium. Initially, the secondary amine acts as a base to deprotonate the active methylene group of the cyanoacetate, generating a nucleophilic carbanion. This species attacks the aldehyde carbonyl carbon of the 3-nitro-4-halobenzaldehyde, initiating the Knoevenagel condensation to form an α-cyano-cinnamate intermediate with the elimination of water. Crucially, the presence of the ortho-nitro group on the aromatic ring significantly activates the para-position towards nucleophilic attack. This electronic activation lowers the energy barrier for the subsequent displacement of the halogen atom (chlorine or bromine) by the excess secondary amine present in the mixture. 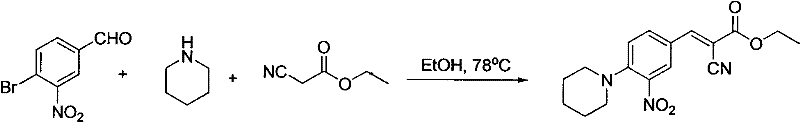 As demonstrated in the specific example above, when piperidine is employed, the reaction proceeds smoothly to yield the target E-isomer with high stereo-selectivity. The thermodynamic stability of the conjugated system drives the equilibrium towards the E-configuration, minimizing the formation of Z-isomer impurities which are often difficult to separate. This mechanistic pathway ensures that the final product possesses a clean impurity profile, a critical attribute for any reliable pharmaceutical intermediate supplier aiming to support downstream drug substance manufacturing.
As demonstrated in the specific example above, when piperidine is employed, the reaction proceeds smoothly to yield the target E-isomer with high stereo-selectivity. The thermodynamic stability of the conjugated system drives the equilibrium towards the E-configuration, minimizing the formation of Z-isomer impurities which are often difficult to separate. This mechanistic pathway ensures that the final product possesses a clean impurity profile, a critical attribute for any reliable pharmaceutical intermediate supplier aiming to support downstream drug substance manufacturing.
From an impurity control perspective, the choice of solvent and temperature plays a pivotal role in suppressing side reactions. In protic solvents like ethanol, the solvation of the ionic intermediates facilitates the SNAr step while minimizing hydrolysis of the ester or nitrile functionalities. The patent data indicates that maintaining the reaction temperature between 50°C and 100°C provides the optimal kinetic balance; temperatures below this range result in incomplete conversion, while excessive heat may promote polymerization or decomposition of the sensitive nitro-aromatic core. Furthermore, the use of a slight excess of the secondary amine serves a dual purpose: it acts as both the nucleophile for the substitution and the base for the condensation, thereby self-regulating the pH of the reaction medium. This self-buffering effect contributes to the reproducibility of the process, ensuring that batch-to-batch variability is kept to a minimum, which is essential for maintaining stringent purity specifications in a GMP environment.
How to Synthesize E-α-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives Efficiently
Implementing this synthesis in a pilot or production plant requires adherence to specific operational parameters to maximize yield and safety. The process begins by charging the reactor with the halogenated benzaldehyde substrate and the chosen solvent, typically ethanol or an ionic liquid depending on the specific solubility requirements of the amine component. Once the solution is homogenized, the cyanoacetate and the secondary amine are added sequentially or as a premixed solution. The detailed standardized synthesis steps, including precise molar ratios, addition rates, and workup procedures, are outlined below to ensure consistent quality.
- Dissolve 3-nitro-4-halobenzaldehyde in a suitable organic solvent such as ethanol or DMF within a reaction vessel equipped for heating.
- Add cyanoacetate or cyanoacetamide and the chosen secondary amine (e.g., piperidine, morpholine) to the reaction mixture in specific molar ratios.
- Heat the mixture to 50-100°C until reaction completion is confirmed by TLC, then isolate the product via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond simple unit price reductions. The most significant impact is observed in the simplification of the raw material portfolio. By eliminating the dependency on palladium catalysts and specialized ligands, the supply chain becomes more resilient to the geopolitical and market fluctuations that often plague the precious metals sector. This shift allows for a more predictable budgeting process and reduces the risk of production stoppages due to catalyst shortages. Moreover, the ability to use common solvents like ethanol facilitates easier integration into existing manufacturing infrastructure, as there is no need for specialized containment or recovery systems designed for exotic or highly toxic solvents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing train. In traditional methods, the removal of palladium residues often requires multiple filtration steps through specialized scavenger resins or treatment with sulfur-based precipitants, all of which add time, labor, and material costs. By completely removing the metal catalyst from the equation, this one-pot method eliminates these expensive purification stages entirely. Additionally, the higher isolated yields reported in the patent examples mean that less starting material is wasted, directly improving the mass intensity of the process. The reduction in solvent volume and the ability to use lower-cost alcohols instead of dipolar aprotic solvents further contribute to substantial cost savings in the overall cost of goods sold, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of this chemical route significantly enhances supply continuity. Because the reaction does not require inert gas protection, it can be performed in standard vessels without the need for complex nitrogen blanketing systems, reducing the technical barriers to entry for contract manufacturing organizations (CMOs). This operational simplicity means that the process can be easily transferred between different manufacturing sites without extensive re-validation of equipment capabilities. Furthermore, the starting materials, such as 3-nitro-4-chlorobenzaldehyde and common cyclic amines, are commodity chemicals available from multiple global vendors, mitigating the risk of single-source dependency. This diversification of the supply base ensures that production schedules remain uninterrupted even if one supplier faces logistical challenges.
- Scalability and Environmental Compliance: Scaling this reaction from the gram scale to multi-ton production is facilitated by its exothermic profile and lack of hazardous reagents. The absence of pyrophoric catalysts or moisture-sensitive organometallics simplifies the safety protocols required for large-scale operations, reducing the burden on EHS teams. From an environmental standpoint, the use of ethanol as a primary solvent aligns well with green chemistry principles, as it is biodegradable and has a lower environmental footprint compared to chlorinated or high-boiling amide solvents. The high atom economy of the tandem reaction minimizes waste generation, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising on output volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this one-pot method over traditional palladium-catalyzed routes?
A: The primary advantage is the elimination of expensive palladium catalysts and ligands, which significantly reduces raw material costs and removes the complex downstream processing required to scavenge heavy metal residues to meet pharmaceutical purity standards.
Q: Does this synthesis require inert atmosphere protection?
A: No, the patent explicitly states that the process does not require inert gas protection, which simplifies operational complexity and allows for easier implementation in standard glass-lined or stainless steel reactors without specialized nitrogen blanketing systems.
Q: What types of amines are compatible with this reaction scope?
A: The method demonstrates broad substrate tolerance, successfully accommodating both cyclic secondary amines like piperidine and morpholine, as well as acyclic amines, allowing for the rapid generation of diverse analog libraries for SAR studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your drug development pipeline depends heavily on the quality and reliability of your intermediate supply. We have thoroughly analyzed the technological potential of Patent CN102558098A and possess the technical expertise to execute this one-pot synthesis with precision. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or preparing for market launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Alpha-Cyano-3-Nitro-4-Substituted Aminophenylacrylic Acid Derivatives meets the exacting standards required for oncology drug synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this metal-free methodology. We encourage you to contact us today to obtain specific COA data for our reference batches and to receive comprehensive route feasibility assessments tailored to your timeline and budget constraints. Let us be your partner in delivering high-quality pharmaceutical intermediates that drive your innovation forward.
