Advanced Manufacturing of Alectinib Intermediate via Optimized Grignard Carboxylation
Advanced Manufacturing of Alectinib Intermediate via Optimized Grignard Carboxylation
The pharmaceutical industry's relentless pursuit of effective treatments for non-small cell lung cancer has placed Alectinib hydrochloride at the forefront of ALK inhibitor therapy. Central to the commercial viability of this life-saving drug is the efficient synthesis of its key precursor, 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid. Patent CN112047829B, published in May 2023, discloses a groundbreaking synthetic methodology that fundamentally reshapes the production landscape for this critical pharmaceutical intermediate. By leveraging a strategic combination of Grignard chemistry and electrophilic aromatic substitution, this novel approach circumvents the prohibitive costs and environmental hazards associated with legacy manufacturing protocols. For R&D directors and procurement specialists alike, understanding the mechanistic elegance and economic implications of this patent is essential for securing a robust supply chain. The following analysis dissects the technical superiority of this route, highlighting its potential to become the industry standard for high-purity API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
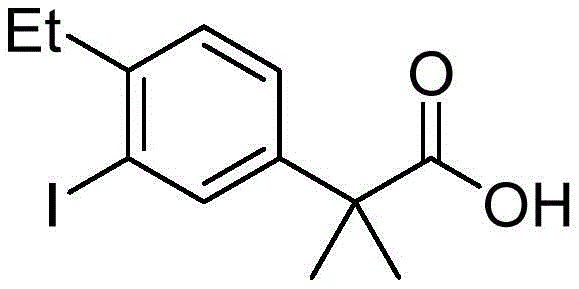
Historically, the synthesis of 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid has been plagued by significant technical and economic bottlenecks that hinder large-scale industrial adoption. One prominent prior art method, illustrated in US2012083488A1, relies heavily on palladium-catalyzed Suzuki coupling reactions. While chemically effective, this route necessitates the use of expensive organoboron reagents and precious metal catalysts, which not only inflate raw material costs but also introduce complex downstream purification challenges to remove trace heavy metals. Furthermore, alternative pathways often employ hazardous reagents such as thionyl chloride and anhydrous aluminum trichloride, generating corrosive acid gases and toxic wastewater that impose severe burdens on environmental compliance systems. Another common strategy involves the use of methyl iodide for methylation steps; however, the high volatility and confirmed mutagenicity of this reagent pose unacceptable health risks to operators in a commercial setting. These cumulative factors render many traditional synthesis routes economically unviable and operationally risky for modern pharmaceutical manufacturing.
The Novel Approach
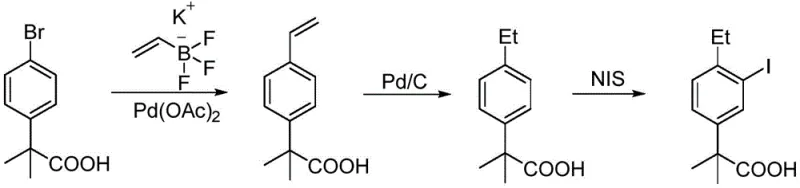
In stark contrast to these cumbersome legacy methods, the methodology disclosed in CN112047829B introduces a streamlined, three-step sequence that prioritizes both safety and cost-efficiency without compromising yield. The process initiates with the readily available and inexpensive starting material, 4-ethylacetophenone, thereby eliminating the dependency on complex, high-cost precursors. By utilizing a Grignard reaction followed by direct carboxylation with carbon dioxide, the synthesis effectively constructs the requisite quaternary carbon center while avoiding the use of toxic methylating agents entirely. The final iodination step is optimized to proceed under mild conditions using elemental iodine or N-iodosuccinimide, ensuring high regioselectivity for the desired 3-iodo position. This strategic redesign of the synthetic tree not only drastically reduces the consumption of hazardous chemicals but also simplifies the workup procedures, shifting the purification burden from energy-intensive distillations to more manageable recrystallizations. Consequently, this novel approach offers a compelling value proposition for manufacturers seeking to optimize their production economics and environmental footprint.
Mechanistic Insights into Grignard Carboxylation and Iodination
The core chemical innovation of this patent lies in the precise execution of the Grignard carboxylation sequence, which serves as the cornerstone for constructing the 2-methylpropanoic acid motif. The mechanism begins with the nucleophilic addition of a methyl Grignard reagent to the ketone carbonyl of 4-ethylacetophenone, forming a tertiary alcohol intermediate which is subsequently converted into a tertiary halide. This halide is then transformed into a sterically hindered Grignard reagent, a challenging transformation that requires careful control of initiation and temperature to prevent Wurtz-type coupling side reactions. Upon exposure to dry carbon dioxide, the organomagnesium species undergoes nucleophilic attack on the electrophilic carbon of CO2, followed by acidic workup to yield the carboxylic acid. This step is particularly advantageous as it utilizes a C1 building block that is both abundant and inexpensive, effectively replacing costly alkyl halides. The subsequent iodination relies on the activating effect of the ethyl group and the directing influence of the carboxyl-containing side chain to achieve selective substitution at the meta-position relative to the alkyl group, ensuring the correct substitution pattern required for downstream coupling in Alectinib synthesis.
Impurity control within this synthetic framework is achieved through the inherent selectivity of the reaction conditions and the physical properties of the intermediates. The use of specific solvents like tetrahydrofuran and diethyl ether facilitates the formation of stable Grignard complexes, minimizing the formation of homocoupling byproducts that often plague organometallic reactions. Furthermore, the patent specifies the use of oxidants such as periodic acid or potassium permanganate during the iodination step to regenerate active iodine species in situ, which helps maintain a consistent reaction rate and prevents the accumulation of reduced iodine byproducts. The final purification via recrystallization from solvent systems like ethanol and water exploits the solubility differences between the target acid and potential isomeric impurities, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. This robust control over the impurity profile is critical for R&D teams aiming to streamline regulatory filings and ensure batch-to-batch consistency.
How to Synthesize 2-(4-Ethyl-3-Iodophenyl)-2-Methylpropanoic Acid Efficiently
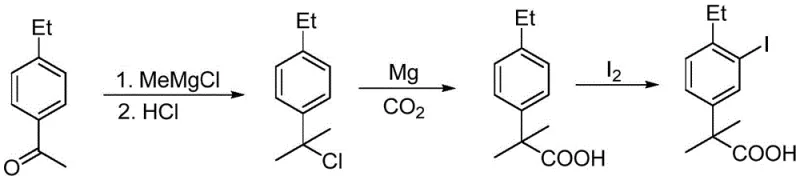
The practical implementation of this synthesis requires strict adherence to inert atmosphere techniques and precise temperature control to maximize yield and safety. The process is divided into three distinct operational stages: the formation of the halo-intermediate, the Grignard carboxylation, and the final electrophilic iodination. Each stage has been optimized in the patent examples to demonstrate scalability, with specific attention paid to molar ratios and reaction times that balance throughput with quality. Operators must ensure that all solvents are anhydrous prior to the Grignard step to prevent premature quenching of the organometallic reagent. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures derived directly from the patent examples, are outlined below for technical reference.
- React 4-ethylacetophenone with a methyl Grignard reagent followed by halogenation to form 2-halo-2-(4-ethylphenyl)propane.
- Convert the halo-intermediate into a Grignard reagent using magnesium metal and react with dry carbon dioxide to form the carboxylic acid.
- Perform electrophilic iodination on the acid using elemental iodine or NIS with an oxidant to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the radical simplification of the raw material portfolio; by relying on commodity chemicals like 4-ethylacetophenone, magnesium, and carbon dioxide, manufacturers can insulate themselves from the price volatility associated with specialized catalysts and complex boronate esters. This shift significantly reduces the cost of goods sold (COGS) and mitigates the risk of supply disruptions caused by the limited availability of niche reagents. Moreover, the elimination of palladium catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which further accelerates production cycles and reduces waste disposal costs. The overall process design favors continuous improvement and scale-up, making it an ideal candidate for long-term commercial partnerships.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-value inputs with low-cost alternatives. By removing the dependency on palladium catalysts and toxic methyl iodide, the direct material costs are substantially lowered. Additionally, the simplified purification workflow, which relies on recrystallization rather than multiple distillation steps, results in significant energy savings and reduced equipment wear. These factors combine to create a highly competitive cost structure that allows for better margin management in the final API production.
- Enhanced Supply Chain Reliability: Sourcing stability is markedly improved due to the use of widely available bulk chemicals. Unlike specialized organoboron compounds that may have long lead times and limited suppliers, the key starting materials for this route are produced by numerous global chemical manufacturers. This diversity in the supply base ensures continuity of supply even during market fluctuations. Furthermore, the reduced hazard profile of the reagents simplifies logistics and storage requirements, lowering the administrative burden associated with handling controlled or highly toxic substances.
- Scalability and Environmental Compliance: From an operational perspective, the process is designed for seamless scale-up from pilot plant to multi-ton commercial production. The absence of corrosive gases like HCl or SO2 in the main synthetic steps reduces the requirement for specialized corrosion-resistant reactors, allowing for the use of standard stainless steel equipment. Environmentally, the generation of less hazardous waste streams aligns with increasingly strict global regulations on pharmaceutical manufacturing, reducing the liability and cost associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent. These inquiries address common concerns regarding reaction safety, impurity profiles, and scalability. The answers provided are derived strictly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for your process development assessments.
Q: Why is the Grignard carboxylation route preferred over Suzuki coupling for this intermediate?
A: The Grignard route eliminates the need for expensive palladium catalysts and complex boronate esters, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: How does this method address the toxicity issues of traditional methylation?
A: By utilizing carbon dioxide gas for carboxylation instead of toxic methyl iodide, the process avoids carcinogenic reagents, enhancing operator safety and environmental compliance.
Q: What are the purification advantages of this synthetic pathway?
A: The final two steps primarily require recrystallization rather than energy-intensive distillation, lowering operational costs and facilitating easier scale-up for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Ethyl-3-Iodophenyl)-2-Methylpropanoic Acid Supplier
As the demand for next-generation ALK inhibitors continues to surge, securing a reliable source of high-quality intermediates is paramount for maintaining competitive advantage in the oncology market. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Grignard chemistry and air-sensitive reactions, ensuring that every batch of 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid meets stringent purity specifications. With rigorous QC labs and a commitment to process excellence, we deliver the consistency and reliability that global pharmaceutical partners demand.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical and commercial confidence.
