Advanced Synthesis of Erlotinib Intermediate for Commercial Scale-Up and Purity Control
Introduction to Next-Generation Kinase Inhibitor Synthesis
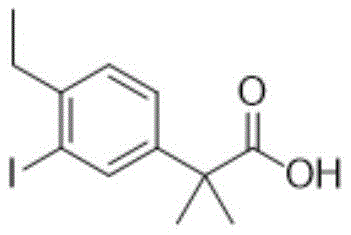
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of complex oncology drug intermediates. A pivotal development in this domain is detailed in patent CN109384664B, which discloses a highly optimized preparation method for 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid. This compound serves as a critical building block for potent kinase inhibitors used in the treatment of advanced non-small cell lung cancer. The significance of this patent lies not merely in the chemical transformation itself, but in its comprehensive approach to solving long-standing industrial pain points associated with heavy metal contamination, hazardous reagent usage, and process scalability. By shifting away from traditional palladium-catalyzed cross-couplings and corrosive Friedel-Crafts acylations, this technology offers a pathway that aligns perfectly with modern Green Chemistry principles while delivering superior product quality.
For R&D directors and process chemists, the implications of this methodology are profound. The ability to synthesize such a sterically hindered and functionally dense aromatic acid without relying on expensive transition metals or extreme reaction conditions represents a significant leap forward in process efficiency. The patent outlines a strategy that not only enhances the purity profile of the intermediate—crucial for downstream API synthesis—but also drastically simplifies the purification workflow. As we delve deeper into the technical specifics, it becomes evident that this approach provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates, ensuring supply chain continuity and cost-effectiveness for global manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
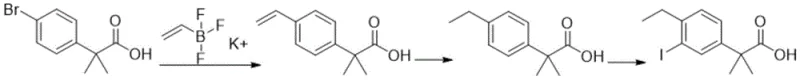
Historically, the synthesis of 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid has been plagued by significant technical and economic hurdles. One prominent prior art route, disclosed in WO2004/046080, relies on a Suzuki-Miyaura coupling catalyzed by palladium acetate to introduce the ethyl group, followed by hydrogenation and iodination. While chemically feasible, this method presents severe drawbacks for industrial application. The reliance on palladium catalysts introduces the persistent risk of heavy metal residue in the final product. Removing trace palladium to meet stringent pharmaceutical standards often requires additional, costly purification steps using specialized scavengers. Furthermore, palladium-catalyzed reactions typically demand strict anaerobic conditions to prevent catalyst deactivation, thereby increasing operational complexity and equipment costs.
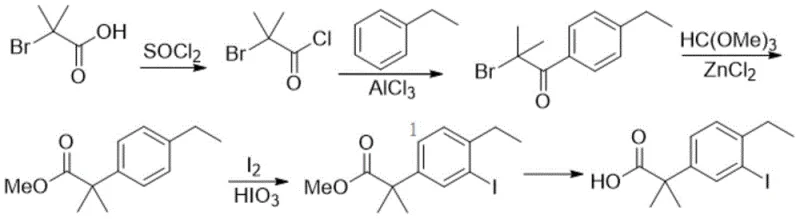
Another conventional approach, described in CN106946650A, utilizes a Friedel-Crafts acylation strategy involving thionyl chloride and aluminum trichloride. This route is particularly problematic from a safety and environmental perspective. The use of thionyl chloride generates hazardous acidic gases, while aluminum trichloride is highly corrosive and produces substantial amounts of acidic wastewater upon quenching. These factors necessitate the use of specialized corrosion-resistant reactors and complex waste treatment facilities, driving up capital expenditure and operational overhead. Moreover, the multi-step nature of these traditional routes often leads to cumulative yield losses and increased formation of difficult-to-remove impurities, compromising the overall quality of the intermediate.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN109384664B introduces a streamlined, copper-catalyzed coupling strategy that effectively bypasses the aforementioned limitations. The core innovation lies in the use of cuprous halides (such as cuprous iodide or bromide) to facilitate the coupling between bromoethylbenzene and a malonate derivative. This substitution of palladium with copper is transformative; copper is not only orders of magnitude cheaper but also far more tolerant of ambient conditions, eliminating the need for rigorous anaerobic setups. The subsequent steps—esterification, double methylation, and iodination—are optimized to proceed under mild conditions that preserve the integrity of the sensitive aromatic ring while ensuring high conversion rates.
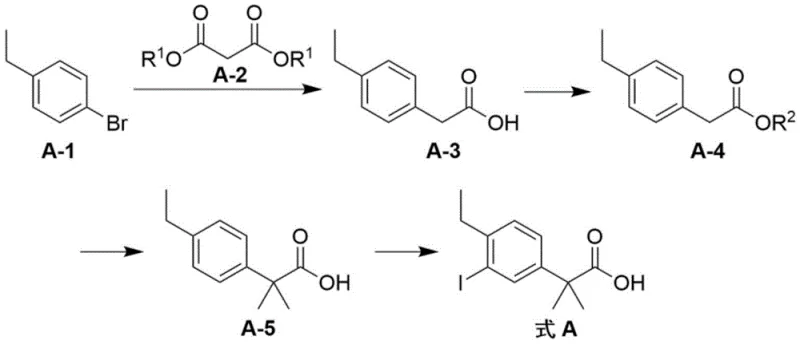
The strategic advantage of this new route is its modularity and robustness. By avoiding the use of corrosive Lewis acids like aluminum trichloride, the process significantly reduces the wear and tear on reaction vessels, extending equipment lifespan and lowering maintenance costs. Furthermore, the post-treatment procedures are simplified to standard aqueous workups and recrystallizations, removing the need for column chromatography which is impractical at the tonnage scale. This holistic optimization ensures that the final intermediate possesses a purity exceeding 99.5% with minimal single impurity content, providing a superior starting material for the synthesis of the final active pharmaceutical ingredient.
Mechanistic Insights into Copper-Catalyzed Coupling and Iodination
The heart of this synthetic innovation is the initial copper-catalyzed coupling reaction. Mechanistically, this step likely proceeds through a nucleophilic aromatic substitution or a copper-mediated radical pathway, where the cuprous species activates the aryl bromide bond. Unlike palladium cycles which involve oxidative addition and reductive elimination steps that are sensitive to oxygen and moisture, the copper system operates efficiently in ether solvents like 1,4-dioxane or tetrahydrofuran at moderate temperatures (60-80°C). The presence of a strong metal alkali base, such as sodium methoxide or potassium tert-butoxide, facilitates the deprotonation of the malonate nucleophile, driving the coupling forward with high regioselectivity. This mechanistic simplicity is key to the process's scalability, as it minimizes the formation of homocoupling byproducts often seen in palladium chemistry.
Following the coupling, the control of impurities is maintained through a carefully sequenced esterification and methylation protocol. The esterification step protects the carboxylic acid functionality, preventing unwanted side reactions during the subsequent alkylation. The double methylation is performed at low temperatures (-10 to 25°C) using methyl iodide and a metal base, ensuring precise installation of the gem-dimethyl group without over-alkylation or degradation of the ester. Finally, the iodination step utilizes N-iodosuccinimide (NIS) or elemental iodine in the presence of an acid catalyst. This electrophilic aromatic substitution is highly selective for the position ortho to the ethyl group, driven by the electronic activation of the ring. The mild acidic conditions prevent the hydrolysis of the ester or the decarboxylation of the acid, preserving the molecular scaffold's integrity throughout the synthesis.
How to Synthesize 2-(4-Ethyl-3-Iodophenyl)-2-Methylpropanoic Acid Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the coupling mixture, where the stoichiometry of the cuprous catalyst and base is critical for maximizing yield. Operators must monitor the reaction temperature closely during the exothermic addition of reagents to prevent thermal runaway. Following the coupling, the hydrolysis and acidification steps must be managed to ensure complete precipitation of the intermediate acid, which is then carried forward to esterification. The subsequent methylation requires careful handling of methyl iodide, a volatile and toxic reagent, necessitating closed-system operations. Finally, the iodination step demands precise control of the oxidant addition rate to manage heat evolution and ensure complete conversion without over-iodination.
- Perform a coupling reaction between bromoethylbenzene and diethyl malonate using a cuprous halide catalyst and metal alkali base in an ether solvent, followed by hydrolysis to obtain the phenylacetic acid derivative.
- Conduct an acid-catalyzed esterification of the resulting acid with an alcohol (such as methanol) to form the corresponding ester intermediate.
- Execute a double methylation reaction on the ester using methyl iodide and a strong metal base, followed by hydrolysis to generate the dimethylated acid precursor.
- Complete the synthesis via an electrophilic iodination reaction using N-iodosuccinimide or iodine in the presence of an acid catalyst to yield the final target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers compelling economic and logistical benefits. The primary driver for cost reduction is the elimination of palladium catalysts. Palladium is a precious metal with volatile pricing and significant supply chain risks; replacing it with abundant copper salts results in a drastic reduction in raw material costs. Furthermore, the removal of palladium scavenging agents and the associated filtration steps simplifies the manufacturing process, reducing labor hours and consumable usage. The avoidance of corrosive reagents like thionyl chloride also translates to lower equipment maintenance costs and reduced expenditure on hazardous waste disposal, contributing to a leaner and more sustainable cost structure for API manufacturing.
- Cost Reduction in Manufacturing: The shift from precious metal catalysis to base metal catalysis fundamentally alters the cost equation of intermediate production. By utilizing inexpensive cuprous halides instead of palladium acetate, manufacturers can achieve significant savings on bill of materials. Additionally, the simplified workup procedures eliminate the need for expensive chromatographic purification or specialized metal scavengers, further driving down the cost per kilogram. This economic efficiency allows for more competitive pricing of the final API, enhancing market positioning for generic and branded drug manufacturers alike.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are readily available from multiple global suppliers. Reagents such as bromoethylbenzene, diethyl malonate, and cuprous iodide are produced at massive scales for various industries, ensuring stable availability and mitigating the risk of shortages that often plague specialty catalysts. The robustness of the reaction conditions, which do not require inert atmosphere gloveboxes or specialized dry rooms, also means that production can be flexibly allocated across different manufacturing sites without extensive retrofitting, thereby securing supply continuity.
- Scalability and Environmental Compliance: The environmental profile of this process aligns with increasingly stringent global regulations regarding industrial emissions and waste. By generating less corrosive wastewater and avoiding the release of sulfur dioxide gas, the facility reduces its environmental footprint and regulatory burden. The high purity of the crude product (>99.5%) minimizes the need for energy-intensive recrystallization cycles, lowering the overall carbon footprint of the manufacturing process. This 'green' credential is increasingly valuable for pharmaceutical companies aiming to meet sustainability goals and secure approval in environmentally conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this method over legacy routes. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value proposition of this supply chain solution.
Q: Why is the copper-catalyzed route preferred over palladium-catalyzed Suzuki coupling for this intermediate?
A: The copper-catalyzed route eliminates the risk of heavy metal palladium residue, which is difficult to remove and can poison downstream catalysts or compromise API stability. Additionally, copper catalysts are significantly less expensive than palladium acetate, and the reaction does not require strict anaerobic conditions, simplifying operational complexity.
Q: How does this method improve environmental safety compared to traditional Friedel-Crafts acylation routes?
A: Traditional routes often utilize hazardous reagents like thionyl chloride and aluminum trichloride, which generate corrosive wastewater and require specialized corrosion-resistant equipment. The novel method avoids these dangerous reagents entirely, utilizing milder conditions that reduce equipment corrosion risks and simplify waste treatment protocols.
Q: What purity levels can be achieved with this optimized synthesis method?
A: The optimized process consistently yields the target intermediate with a purity exceeding 99.5%, with the maximum single impurity content controlled below 0.1%. This high level of purity is critical for ensuring the quality and safety of the final oncology drug product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Ethyl-3-Iodophenyl)-2-Methylpropanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology therapeutics depends on the reliability and quality of the supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid meets the highest international standards. Our infrastructure is designed to handle the specific safety and environmental requirements of this optimized copper-catalyzed process, providing a secure and compliant manufacturing environment.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to accelerate the delivery of life-saving medications to patients worldwide through superior chemical manufacturing excellence.
