Advanced Synthesis of Alectinib Intermediate for Commercial Scale-up and Cost Reduction
Advanced Synthesis of Alectinib Intermediate for Commercial Scale-up and Cost Reduction
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for the production of critical oncology intermediates. Patent CN112047829B, published in May 2023, discloses a novel synthetic methodology for 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid, a pivotal building block in the manufacture of Alectinib hydrochloride. This ALK inhibitor has demonstrated superior progression-free survival rates in non-small cell lung cancer patients compared to earlier generation treatments. The disclosed technology addresses long-standing challenges in prior art by utilizing a Grignard-based carboxylation strategy that bypasses the need for precious metal catalysts. By shifting from palladium-dependent cross-coupling to a more direct organometallic approach, this innovation offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to optimize their API manufacturing pipelines.
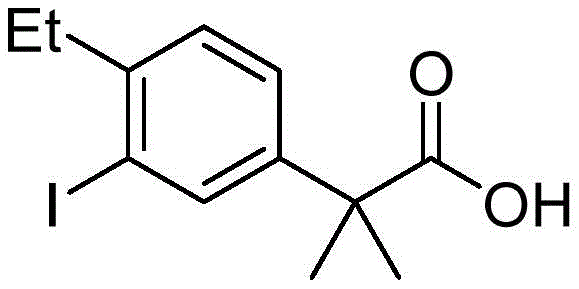
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate has relied on methodologies that impose significant economic and environmental burdens on the supply chain. One prominent prior art route, illustrated in US2012083488A1, employs a Suzuki-Miyaura coupling between a bromophenyl propanoic acid derivative and an ethyl boronate species. While chemically effective, this pathway necessitates the use of expensive palladium catalysts and specialized organoboron reagents. The residual palladium levels in the final product often require rigorous and costly purification steps to meet stringent regulatory limits for heavy metals in active pharmaceutical ingredients. Furthermore, alternative routes described in CN106946650A utilize Friedel-Crafts acylation with thionyl chloride and aluminum trichloride, generating substantial amounts of corrosive acidic waste and posing severe equipment corrosion risks. Another method involving methyl iodide for methylation introduces significant occupational health hazards due to the mutagenicity and volatility of the alkylating agent.
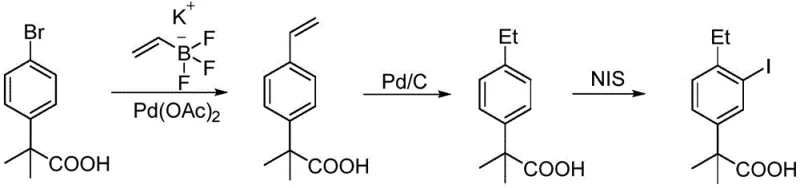
The Novel Approach
In stark contrast, the methodology outlined in CN112047829B presents a streamlined three-step sequence that fundamentally alters the cost structure of production. The process initiates with the readily available and inexpensive 4-ethylacetophenone, avoiding the need for pre-functionalized halogenated starting materials required in cross-coupling strategies. By leveraging a Grignard addition followed by a direct carboxylation with carbon dioxide, the synthesis constructs the quaternary carbon center and the carboxylic acid moiety in a highly atom-economical fashion. This approach not only eliminates the dependency on volatile and toxic methyl iodide but also removes the requirement for transition metal catalysts entirely. The final iodination step is achieved under controlled oxidative conditions, ensuring high regioselectivity without the need for complex protecting group strategies. This shift represents a paradigm change towards greener, safer, and more economically viable cost reduction in API manufacturing.
Mechanistic Insights into Grignard Carboxylation and Electrophilic Iodination
The core of this synthetic innovation lies in the precise manipulation of organomagnesium species to build molecular complexity. The first stage involves the nucleophilic attack of a methyl Grignard reagent on the carbonyl carbon of 4-ethylacetophenone. This addition forms a tertiary alcohol intermediate, which is subsequently converted into a tertiary halide using reagents such as trimethylchlorosilane or concentrated hydrochloric acid. This transformation is critical as it activates the benzylic position for the subsequent formation of the Grignard reagent. The conversion of the tertiary halide into the corresponding organomagnesium species must be carefully managed to prevent Wurtz-type coupling side reactions, typically achieved by controlling the addition rate and temperature in solvents like tetrahydrofuran or diethyl ether.
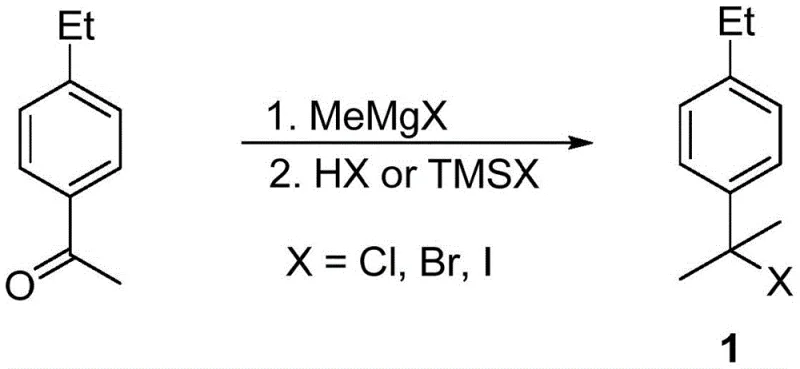
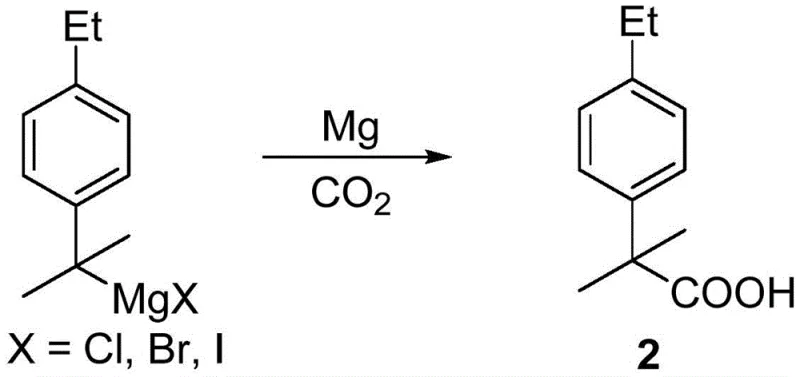
Following the formation of the Grignard reagent, the introduction of dry carbon dioxide gas serves as the electrophile to install the carboxylic acid group. This carboxylation step proceeds through a nucleophilic attack of the carbanion on the carbon of CO2, forming a magnesium carboxylate salt. Subsequent acidic workup liberates the free acid, yielding 2-(4-ethylphenyl)-2-methylpropanoic acid. The final transformation involves an electrophilic aromatic substitution where elemental iodine or N-iodosuccinimide acts as the iodinating agent. Crucially, the presence of an oxidant such as periodic acid or hydrogen peroxide regenerates the active iodinating species in situ, driving the reaction to completion and ensuring high conversion rates. This mechanistic pathway allows for excellent control over the impurity profile, as the final product can often be purified via simple recrystallization rather than energy-intensive distillation.
How to Synthesize 2-(4-Ethyl-3-iodophenyl)-2-methylpropanoic Acid Efficiently
Implementing this synthesis requires strict adherence to inert atmosphere conditions and temperature controls to maximize yield and safety. The process begins with the preparation of the tertiary halide precursor, followed by the generation of the Grignard reagent and its reaction with carbon dioxide. The final iodination step demands careful monitoring of oxidant stoichiometry to prevent over-oxidation or poly-iodination. Detailed operational parameters, including specific solvent choices, molar ratios, and workup procedures, are essential for reproducing the high purity levels reported in the patent examples. For a comprehensive guide on executing these steps in a GMP environment, please refer to the standardized protocol below.
- React 4-ethylacetophenone with methyl magnesium halide followed by halogenation to form 2-halo-2-(4-ethylphenyl)propane.
- Convert the halo-intermediate into a Grignard reagent and react with carbon dioxide to yield 2-(4-ethylphenyl)-2-methylpropanoic acid.
- Perform electrophilic iodination using elemental iodine or NIS with an oxidant to obtain the final 3-iodo substituted product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary driver for cost optimization is the substitution of high-value inputs with commodity chemicals. By replacing palladium catalysts and boronic acids with magnesium metal and carbon dioxide, the direct material cost is drastically simplified. Furthermore, the elimination of toxic methyl iodide reduces the regulatory burden associated with handling hazardous substances, thereby lowering compliance costs and insurance premiums. The reliance on recrystallization for purification in the final steps, as opposed to complex chromatographic separations or high-vacuum distillations, significantly reduces energy consumption and processing time. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for expensive scavenging resins and extensive heavy metal testing, directly improving the gross margin of the final intermediate. Additionally, the use of 4-ethylacetophenone as a starting material leverages existing bulk chemical infrastructure, ensuring stable pricing and availability compared to specialized cross-coupling partners.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as magnesium turnings, dry ice, and elemental iodine, are globally sourced commodities with robust supply networks. This diversification reduces the risk of single-source bottlenecks that often plague specialized reagent markets. The simplified workflow also shortens the overall production cycle time, allowing for faster response to fluctuating market demand for Alectinib APIs.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to Friedel-Crafts or methylation routes, aligning with modern green chemistry principles. The absence of corrosive acid gases and mutagenic alkylating agents simplifies waste treatment protocols and enhances worker safety. This environmental profile facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the key advantages of this synthesis method over Suzuki coupling?
A: This method eliminates the need for expensive palladium catalysts and complex boronate reagents, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: How is the regioselectivity of the iodination step controlled?
A: The process utilizes specific oxidants like periodic acid or potassium permanganate alongside elemental iodine or NIS to ensure selective substitution at the 3-position ortho to the ethyl group.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids highly toxic reagents like methyl iodide and corrosive thionyl chloride where possible, relying on recrystallization rather than complex distillation for purification, which enhances scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Ethyl-3-iodophenyl)-2-methylpropanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN112047829B can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-(4-ethyl-3-iodophenyl)-2-methylpropanoic acid meets the exacting standards required for oncology drug substance manufacturing.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us on optimizing this synthesis for your specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring a seamless integration of this advanced intermediate into your Alectinib production strategy.
