Revolutionizing Aryl Halide Production With Safe Organocatalytic Halogenation Technology
The chemical industry is constantly seeking more efficient and safer pathways to synthesize critical building blocks, and the recent disclosure in patent CN112573978A presents a transformative approach to aryl halide production. This technology addresses the longstanding challenges associated with aromatic halogenation by utilizing a novel organocatalytic system based on sulfoxides or nitrogen oxides. Unlike traditional methods that rely on hazardous elemental halogens or expensive transition metals, this invention leverages inexpensive and readily available catalysts to activate N-halosuccinimides, resulting in exceptional yields and selectivity. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline supply chains for high-purity pharmaceutical intermediates. The method operates under remarkably mild conditions, typically at 25°C, which not only reduces energy consumption but also minimizes the formation of unwanted by-products, ensuring a cleaner impurity profile for downstream processing.
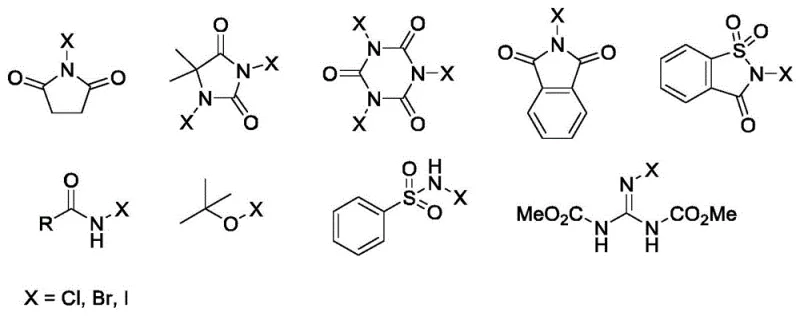
Furthermore, the versatility of this synthetic route allows for the functionalization of a broad spectrum of substrates, ranging from simple benzene derivatives to complex heterocyclic systems found in active pharmaceutical ingredients. The ability to introduce chlorine, bromine, or iodine atoms with such precision makes this technology a cornerstone for modern medicinal chemistry campaigns. By adopting this methodology, manufacturers can achieve reliable aryl halide supplier status, meeting the rigorous quality standards demanded by global regulatory bodies. The patent data explicitly highlights the superiority of this catalytic system over conventional Lewis acid catalysts, providing a compelling case for its integration into large-scale commercial processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl halides has been plagued by significant operational and safety hurdles that impact both cost and efficiency in pharmaceutical intermediate manufacturing. Traditional electrophilic aromatic substitution often requires the use of elemental halogens such as chlorine or bromine gas, which are highly toxic, corrosive, and difficult to handle on an industrial scale. Even when safer N-halo reagents are employed, the reaction frequently necessitates the addition of stoichiometric amounts of Lewis acids like iron(III) chloride or aluminum chloride to activate the halogen source. These conventional activators often suffer from poor atom economy, generate substantial amounts of acidic waste, and can lead to over-halogenation or regio-isomeric mixtures that are difficult to separate. Moreover, many standard protocols require harsh reaction conditions, including elevated temperatures or strongly acidic media, which can degrade sensitive functional groups present in complex drug molecules.
The Novel Approach
In stark contrast, the method described in CN112573978A utilizes a catalytic amount of dimethyl sulfoxide (DMSO) or specific nitrogen oxides to activate the halogenating agent, effectively bypassing the need for hazardous gases or heavy metal promoters. The experimental data provided in the patent illustrates a dramatic improvement in performance; for instance, in the chlorination of N-Bz-Tyr-OMe, the DMSO-catalyzed reaction achieved an impressive 88% yield, whereas traditional catalysts like FeCl3 resulted in a mere 3% yield under comparable conditions. 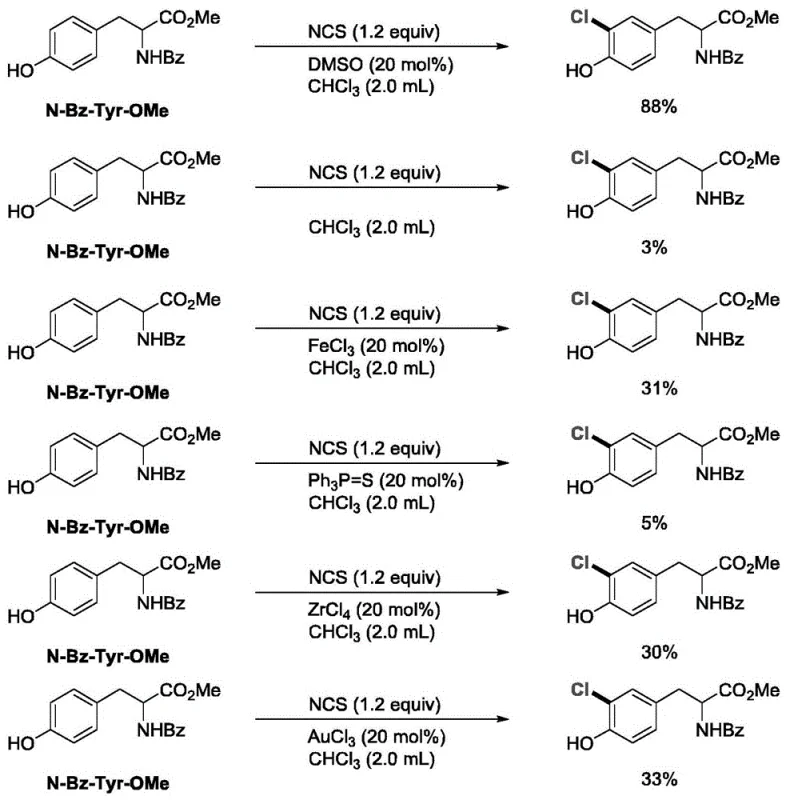
This substantial increase in efficiency translates directly to cost reduction in pharmaceutical intermediate manufacturing by maximizing raw material utilization and minimizing purification steps. Similarly, for the chlorination of Diclofenac, the novel method delivered an 87% yield compared to only 43% with gold chloride catalysts, demonstrating its robustness across diverse chemical scaffolds. The mild reaction environment preserves the integrity of the substrate, ensuring high selectivity and reducing the burden on quality control laboratories to remove trace metal contaminants.
Mechanistic Insights into Sulfoxide-Catalyzed Electrophilic Halogenation
The core innovation of this technology lies in the unique activation mechanism wherein the sulfoxide or nitrogen oxide catalyst interacts with the N-halosuccinimide to generate a highly reactive electrophilic halogen species in situ. 
Unlike Lewis acids which coordinate directly to the halogen atom to weaken the N-X bond, the oxygen atom of the sulfoxide catalyst likely forms a transient hypervalent intermediate that facilitates the transfer of the halogen cation to the electron-rich aromatic ring. This pathway lowers the activation energy of the rate-determining step, allowing the reaction to proceed rapidly even at room temperature. The catalytic cycle is regenerated efficiently, meaning that only 20 mol% of the catalyst is required to drive the transformation to completion, which is a significant advantage over stoichiometric activators. This mechanistic efficiency ensures that the reaction kinetics are favorable for scale-up, as heat generation can be managed more easily without the exothermic spikes often associated with elemental halogen additions.
From an impurity control perspective, this mechanism offers superior regioselectivity, predominantly targeting the most activated positions on the aromatic ring without causing poly-halogenation. The absence of strong Lewis acids prevents side reactions such as Friedel-Crafts alkylation or acylation that might occur if susceptible groups are present on the substrate. Furthermore, the by-product of the reaction is succinimide, a benign solid that is easily removed during aqueous workup or crystallization, unlike the metal salts generated by traditional methods which require complex chelation and filtration steps. This cleanliness of the reaction profile is critical for producing high-purity aryl halides that meet the stringent specifications required for clinical trial materials and commercial API production.
How to Synthesize 3-Chloro-indole-2-carboxylic Acid Ethyl Ester Efficiently
To implement this technology in a laboratory or pilot plant setting, the procedure is straightforward and relies on standard equipment familiar to organic chemists. The patent details a robust protocol where the aromatic substrate is dissolved in a common solvent such as chloroform, followed by the addition of the halogenating agent and the liquid catalyst. The mixture is then stirred at ambient temperature, eliminating the need for specialized heating or cooling infrastructure. Detailed standardized synthesis steps see the guide below for precise molar ratios and workup procedures tailored to specific substrates.
- Charge a reaction vessel with the aromatic substrate, N-halosuccinimide (1.2 equiv), and a catalytic amount of dimethyl sulfoxide (20 mol%) in chloroform.
- Stir the reaction mixture at 25°C for 2 to 12 hours, monitoring progress via TLC or HPLC to ensure complete conversion.
- Remove the solvent by rotary evaporation and purify the crude residue via column chromatography to isolate the high-purity aryl halide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfoxide-catalyzed halogenation method offers tangible benefits that extend beyond simple reaction yields. The shift away from elemental halogens and heavy metal catalysts fundamentally alters the risk profile of the manufacturing process, leading to enhanced supply chain reliability and reduced regulatory burden. By utilizing inexpensive organocatalysts like DMSO, companies can achieve substantial cost savings in raw material procurement while simultaneously simplifying waste management protocols. The high selectivity of the reaction reduces the volume of solvent and silica gel required for purification, further driving down the cost of goods sold. Additionally, the mild conditions allow for the use of a wider range of reactor materials, potentially extending the lifespan of existing manufacturing assets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as gold or iron salts removes a significant cost driver from the bill of materials, while the high yields ensure that valuable starting materials are not wasted on side products. The simplified workup procedure reduces labor hours and utility consumption associated with extensive purification, leading to a more economically viable process overall. Furthermore, the benign nature of the by-products lowers the cost of waste disposal and environmental compliance, contributing to a leaner operational budget.
- Enhanced Supply Chain Reliability: Sourcing hazardous gases like chlorine often involves complex logistics and strict safety regulations that can disrupt supply continuity; replacing these with stable solid N-halosuccinimides mitigates these risks significantly. The robustness of the catalytic system means that production batches are less likely to fail due to sensitivity to moisture or temperature fluctuations, ensuring consistent on-time delivery to customers. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding schedules of pharmaceutical clients.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with green chemistry principles, making it easier to obtain environmental permits for capacity expansion. The absence of heavy metal residues in the final product simplifies the validation process for regulatory filings, accelerating time-to-market for new drug candidates. Scalability is further supported by the exotherm control inherent in the mild reaction conditions, allowing for safe operation in large-scale reactors without the need for complex cooling systems.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this halogenation technology, based on the detailed experimental data provided in the patent documentation. Understanding these nuances is essential for process chemists evaluating the feasibility of this route for their specific projects. The answers reflect the proven capabilities of the method across a wide array of chemical structures.
Q: What are the primary advantages of using DMSO as a catalyst for halogenation?
A: Using DMSO as a catalyst significantly enhances reaction yields compared to traditional Lewis acids, often achieving over 85% yield where other methods fail below 35%, while operating under mild, metal-free conditions.
Q: Is this halogenation method suitable for complex API intermediates?
A: Yes, the method demonstrates excellent compatibility with complex molecules such as Diclofenac, Naproxen, and Clopidogrel, maintaining high selectivity without degrading sensitive functional groups.
Q: Does this process require hazardous elemental halogens?
A: No, the process utilizes solid N-halosuccinimides as safe halogen sources, eliminating the need for toxic and corrosive elemental chlorine or bromine gas, thereby improving workplace safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the sulfoxide-catalyzed halogenation method are translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aryl halide intermediate meets the highest industry standards. Our commitment to quality and safety makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to contact our technical procurement team to discuss how this advanced halogenation technology can be applied to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this metal-free protocol. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you achieve your production goals efficiently.
