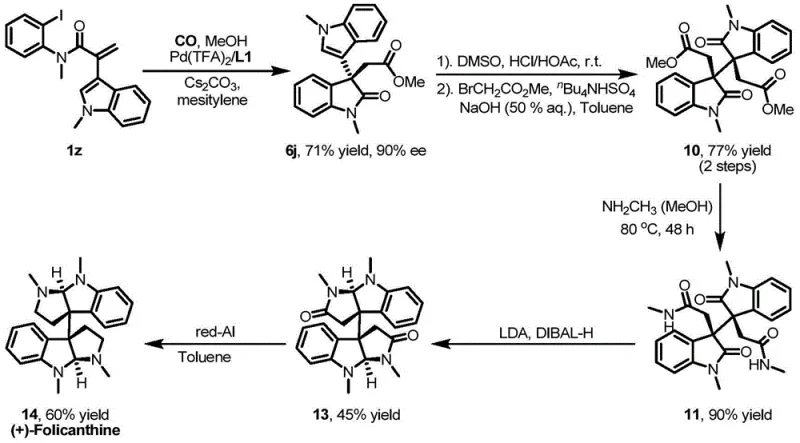
Advanced Phosphoramidite Ligands for High-Enantioselective Alkaloid Synthesis and Commercial Scale-Up
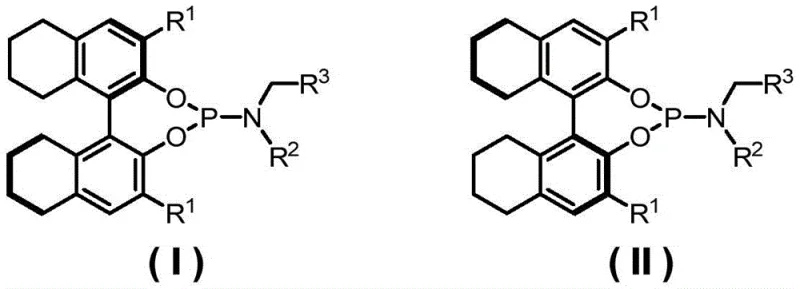
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel chiral monodentate phosphoramidite ligands, as detailed in patent CN113444125A. This groundbreaking technology addresses a long-standing challenge in the synthesis of complex natural products, specifically hexahydropyrrole indole alkaloids and dimeric cyclotryptamine alkaloids, by overcoming the limitations of traditional bidentate ligands. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal opportunity to enhance the enantioselectivity of domino Heck carbonylation reactions while streamlining the supply chain for high-purity pharmaceutical intermediates. The patent discloses a series of ligands represented by general structural formulas (I) and (II), which exhibit superior performance in palladium-catalyzed asymmetric transformations. By leveraging these advanced catalytic systems, manufacturers can achieve substantial cost reduction in pharmaceutical intermediates manufacturing through improved yields and simplified purification processes. The ability to utilize monodentate ligands effectively in carbonylation reactions, where carbon monoxide typically competes with chiral ligands for coordination sites, marks a distinct technological leap forward for the industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex alkaloid frameworks relied heavily on multidentate or bidentate chiral ligands, which often presented significant drawbacks in terms of reactivity and selectivity under carbonylation conditions. In asymmetric Heck carbonylation reactions, the strong competitive coordination between carbon monoxide and traditional chiral ligands with palladium catalysts frequently leads to the severe destruction of the chiral environment, resulting in disappointingly low enantiomeric excess values. For instance, early attempts by research groups to synthesize hexahydropyrrole indole alkaloids using bidentate Binap ligands yielded only 28% ee, which is commercially unacceptable for high-value active pharmaceutical ingredients. Furthermore, the rigid structure of bidentate ligands can limit the substrate scope, making it difficult to accommodate diverse nucleophilic reagents such as aryl amines or phenols without extensive optimization. These inefficiencies translate directly into higher production costs, increased waste generation, and prolonged lead times for high-purity pharmaceutical intermediates, creating bottlenecks for supply chain heads aiming to maintain continuous production schedules.
The Novel Approach
The novel approach introduced in this patent utilizes specially designed monodentate phosphoramidite ligands that maintain a robust chiral environment even in the presence of carbon monoxide, thereby enabling highly enantioselective domino Heck carbonylation reactions. This strategy not only resolves the coordination competition issue but also expands the versatility of the reaction system to include a wide array of nucleophilic reagents, including aryl boronic acids, aryl amines, and various alcohols. The structural flexibility of these monodentate ligands allows for fine-tuning of steric and electronic properties through the variation of R1, R2, and R3 substituents, ensuring optimal performance across different substrate classes. As demonstrated in the patent, this method efficiently solves the synthesis problem of hexahydropyrrole indole alkaloids and dimeric chromane alkaloids, which are critical scaffolds in drug discovery. For a reliable pharmaceutical intermediates supplier, adopting this methodology means offering clients a pathway to complex molecules with significantly reduced process steps and enhanced overall efficiency, positioning the supply chain for greater resilience and scalability in the face of market demands.
Mechanistic Insights into Pd-Catalyzed Asymmetric Domino Heck Carbonylation
The core of this technological advancement lies in the unique interaction between the palladium catalyst and the chiral phosphoramidite ligand during the catalytic cycle. Unlike bidentate ligands that occupy two coordination sites and may be displaced by carbon monoxide, the monodentate nature of the phosphoramidite ligand allows for a dynamic equilibrium that preserves the chiral information transfer to the substrate. The ligand structure, featuring a H8-BINOL backbone with bulky aryl substituents, creates a specific chiral pocket that guides the insertion of carbon monoxide and the subsequent nucleophilic attack with high stereocontrol. This mechanism ensures that the formation of the new carbon-carbon and carbon-heteroatom bonds occurs with exceptional enantioselectivity, often exceeding 90% ee across a broad range of substrates. The stability of the palladium-ligand complex under reaction conditions, typically ranging from 0 to 80 °C, further contributes to the robustness of the process, minimizing catalyst decomposition and metal leaching which are critical concerns for commercial scale-up of complex pharmaceutical intermediates.
Impurity control is another critical aspect where this mechanistic understanding provides a commercial advantage. The high chemoselectivity of the system minimizes the formation of side products such as homocoupling byproducts or non-carbonylated Heck products, which are common in less optimized systems. By precisely controlling the stoichiometry of the nucleophilic reagent and the pressure of carbon monoxide, typically between 0.1 to 5 atm, the reaction pathway is directed almost exclusively towards the desired chiral product. This inherent selectivity reduces the burden on downstream purification processes, such as chromatography or crystallization, leading to higher overall recovery rates and lower solvent consumption. For quality assurance teams, this translates to a cleaner crude product profile and more consistent batch-to-batch reproducibility, which are essential metrics for maintaining stringent purity specifications in the supply of high-purity OLED material or pharmaceutical precursors.

How to Synthesize Chiral Phosphoramidite Ligands Efficiently
The preparation of these high-performance ligands follows a logical and scalable three-step synthetic route that is well-suited for industrial adaptation. The process begins with the modification of the H8-BINOL backbone via Suzuki coupling, followed by the synthesis of the chiral amine component, and concludes with the phosphoramidite coupling step. Each stage has been optimized to maximize yield and minimize the use of hazardous reagents, aligning with modern green chemistry principles. Detailed standardized synthesis steps see the guide below, which outlines the specific conditions and reagents required to reproduce the ligands described in the patent examples. This transparency in process chemistry allows our technical team to rapidly assess feasibility and integrate the synthesis into our existing manufacturing lines, ensuring a reliable agrochemical intermediate supplier or pharma partner can meet volume requirements without compromising on quality.
- Perform Suzuki coupling on di-Br-H8-Binol with aryl boric acid using palladium catalysis to obtain the biaryl backbone intermediate.
- Synthesize the secondary amine component via condensation reduction of aniline and aldehyde or substitution of aniline with alkyl halides.
- React the secondary amine with phosphorus trichloride to form a chlorophosphine intermediate, then couple with the biaryl backbone under alkaline conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers profound benefits for procurement managers and supply chain heads focused on cost efficiency and reliability. The elimination of expensive and difficult-to-remove transition metal contaminants, often associated with less selective catalysts, significantly simplifies the downstream processing workflow. This reduction in purification complexity directly correlates to substantial cost savings in manufacturing, as it lowers solvent usage, reduces waste disposal costs, and shortens the overall production cycle time. Furthermore, the use of readily available starting materials for the ligand synthesis, such as substituted anilines and boronic acids, ensures a stable raw material supply base that is less susceptible to market volatility. These factors combined create a more resilient supply chain capable of sustaining long-term production contracts for critical intermediates.
- Cost Reduction in Manufacturing: The high enantioselectivity and yield of this process mean that less raw material is wasted on unwanted stereoisomers or side products, leading to a more atom-economical process. By avoiding the need for costly chiral resolution steps or extensive recrystallization to upgrade optical purity, manufacturers can achieve a drastically simplified production flow. The ability to operate at moderate temperatures and pressures also reduces energy consumption, contributing to a lower carbon footprint and reduced operational expenditures. These qualitative efficiencies compound over large production volumes, delivering significant financial value to the end client without the need for risky capital investment in new reactor hardware.
- Enhanced Supply Chain Reliability: The robustness of the palladium-phosphoramidite catalyst system ensures consistent performance across different batches, which is crucial for maintaining just-in-time delivery schedules. Since the ligand synthesis does not rely on exotic or single-source reagents, the risk of supply disruption is minimized, providing procurement teams with greater confidence in sourcing strategies. The scalability of the reaction from gram to kilogram scale has been demonstrated in the patent examples, indicating that the technology can be seamlessly transferred to pilot and commercial plants. This scalability ensures that as demand for the final alkaloid products grows, the supply of the key intermediate can be ramped up quickly to meet market needs without lengthy re-qualification periods.
- Scalability and Environmental Compliance: The process utilizes common organic solvents like toluene and mesitylene, which are well-understood in terms of handling and recycling within regulated chemical facilities. The high selectivity of the reaction reduces the generation of hazardous waste streams, making it easier to comply with increasingly strict environmental regulations regarding effluent discharge. Additionally, the potential for catalyst recycling or the use of lower catalyst loadings as the process is optimized further enhances the sustainability profile of the manufacturing route. This alignment with environmental, social, and governance (ESG) goals is becoming a key differentiator for suppliers seeking to partner with top-tier multinational corporations that prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric catalysis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the fit of this technology within their specific product portfolios and manufacturing capabilities. We encourage technical teams to review these points closely to identify opportunities for process optimization and cost avoidance in their current supply chains.
Q: What is the primary advantage of this monodentate ligand over bidentate ligands?
A: This monodentate phosphoramidite ligand solves the low enantioselectivity issue in asymmetric domino Heck carbonylation reactions, achieving significantly higher ee values compared to traditional bidentate ligands like Binap which often yield poor results in this specific transformation.
Q: Can this catalytic system accommodate diverse nucleophilic reagents?
A: Yes, the system demonstrates broad substrate scope, successfully accommodating aryl boronic acids, aryl amines, alcohols, and phenols as nucleophilic reagents, allowing for the synthesis of various functionalized hexahydropyrrole indole alkaloids.
Q: Is this process suitable for large-scale industrial production?
A: The reaction conditions utilize standard palladium catalysts and common solvents like toluene and mesitylene at moderate temperatures, indicating strong potential for commercial scale-up and integration into existing fine chemical manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoramidite Ligand Supplier
The technical potential of this novel phosphoramidite ligand system is immense, offering a clear pathway to high-value chiral intermediates that are currently difficult to source economically. NINGBO INNO PHARMCHEM stands ready as your CDMO expert to translate this patent innovation into commercial reality, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of ligand or intermediate meets the exacting standards required for pharmaceutical and fine chemical applications. We understand the critical nature of chiral purity and the impact it has on the efficacy and safety of the final drug product, which is why our quality management systems are designed to provide full traceability and consistency.
We invite you to initiate a dialogue with our technical procurement team to explore how this technology can optimize your specific synthesis routes. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic system for your projects. We are prepared to provide specific COA data and route feasibility assessments to support your internal validation processes. Let us collaborate to engineer a supply chain that is not only cost-effective but also technologically advanced and resilient against future market shifts.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
