Advanced Catalytic Hydrogenation Strategy for High-Purity Biapenem Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for the synthesis of carbapenem antibiotics, particularly biapenem, due to its broad-spectrum efficacy against gram-negative and gram-positive bacteria. Patent CN101768174B introduces a transformative approach to preparing biapenem that addresses critical bottlenecks in industrial scalability and purification efficiency. This technology leverages a specific catalytic hydrogenation protocol conducted in aprotic polar organic solvents, fundamentally altering the solubility dynamics of the reaction medium. By shifting away from traditional aqueous-buffer systems, the process enables the direct precipitation of high-purity biapenem without the need for complex ion-exchange resin chromatography or energy-intensive freeze-drying steps. The method demonstrates exceptional compatibility with various carboxy protective groups, ensuring versatility in upstream synthetic routes while maintaining a stable yield above 72% and purity exceeding 98.0%. For global supply chain stakeholders, this represents a pivotal advancement in reliable carbapenem antibiotic intermediate supplier capabilities, offering a pathway to significantly reduced manufacturing costs and enhanced production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deprotection of biapenem precursors has been plagued by inefficient solvent systems and cumbersome purification workflows that hinder commercial viability. Traditional methods often rely on zinc powder reduction or catalytic hydrogenation in mixed aqueous-organic solvents requiring phosphate buffers to maintain pH stability. These legacy processes suffer from severe solubility issues; the raw materials, possessing ionic structures and large hydrophobic groups, exhibit poor solubility in water and limited solubility in common organic solvents. Consequently, prior art methods necessitate massive solvent volumes, typically ranging from 30 to 100 times the weight of the raw material for organic solvents and 10 to 50 times for water. This excessive solvent usage drastically lowers the throughput of pressure reaction vessels, where producing merely 1 kilogram of product might require a 300-liter reactor. Furthermore, the downstream processing is equally problematic, often mandating macroporous adsorbent resin column chromatography, concentration of large aqueous volumes, and low-temperature freezing crystallization or lyophilization. These steps not only inflate operational expenditures but also expose the thermally sensitive biapenem to degradation risks, with literature indicating 10% to 20% degradation during concentration processes, ultimately compromising yield and purity.
The Novel Approach
In stark contrast, the methodology disclosed in CN101768174B utilizes aprotic polar organic solvents to create a highly efficient reaction environment that overcomes the solubility barriers of conventional techniques. By employing solvents such as N,N-dimethylformamide (DMF), N-methyl pyrrolidone (NMP), or dimethyl sulfoxide (DMSO), the process achieves complete dissolution of the ionic starting material with minimal solvent consumption, typically only 1 to 10 times the weight of the compound. This dramatic reduction in solvent volume allows for a massive increase in equipment utilization efficiency; a mere 10 to 20-liter pressure reactor can now produce 1 kilogram of biapenem, representing a fifteen-fold improvement in space-time yield compared to older methods. The reaction proceeds under mild catalytic hydrogenation conditions to remove the carboxy protective group, followed by a simplified workup where water is added to the mixture, the catalyst is filtered off, and a water-miscible organic solvent induces direct precipitation of the product. This eliminates the need for buffer reagents, ion-exchange resins, and freeze-drying, streamlining the entire operation into a few straightforward unit operations that are ideal for cost reduction in beta-lactam antibiotic manufacturing.
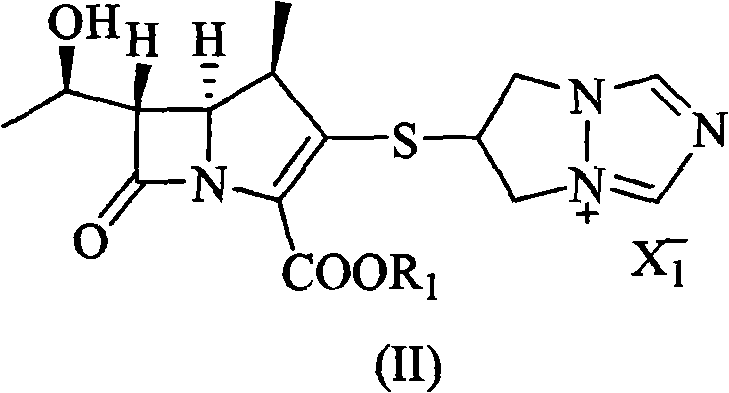
Mechanistic Insights into Aprotic Solvent-Mediated Catalytic Hydrogenation
The core innovation of this synthesis lies in the strategic selection of the reaction medium to facilitate the catalytic removal of the carboxy protecting group without compromising the stability of the sensitive beta-lactam ring. In this mechanism, the starting material, represented by Formula (II), contains a specific protecting group R1 that is susceptible to hydrogenolysis, such as p-nitrobenzyloxycarbonyl or benzyloxycarbonyl derivatives. When dissolved in aprotic polar solvents, the ionic nature of the quaternary ammonium salt in the triazole side chain is effectively stabilized, preventing aggregation or precipitation that would otherwise shield the reactive sites from the catalyst surface. The hydrogenation is typically catalyzed by palladium or platinum-based species, such as palladium on carbon (Pd/C), platinum on carbon (Pt/C), or platinum dioxide (PtO2), which activate molecular hydrogen to cleave the benzyl-oxygen or nitrobenzyl-oxygen bond. The reaction conditions are carefully controlled, with hydrogen pressures maintained between 1 and 10 Kg/cm² and temperatures ranging from 0°C to 80°C, preferably 20°C to 40°C, to ensure selective deprotection while minimizing hydrolysis of the beta-lactam moiety. This precise control over the reaction environment is critical for maintaining the stereochemical integrity of the chiral centers at positions 4, 5, and 6 of the carbapenem nucleus.
Furthermore, the purification mechanism relies on the differential solubility of the product versus impurities in the post-reaction solvent system. Upon completion of the hydrogenation, the addition of water serves a dual purpose: it quenches any residual reactive species and modifies the polarity of the medium to prepare for precipitation. Since the deprotected biapenem has distinct solubility characteristics compared to the organic byproducts and the catalyst support, simple filtration removes the solid catalyst effectively. Subsequent addition of a water-miscible organic solvent, such as ethanol, acetone, or isopropanol, to the aqueous filtrate reduces the solubility of the biapenem zwitterion, causing it to crystallize out of the solution directly. This "salting-out" or anti-solvent precipitation effect bypasses the need for thermal concentration, which is a major source of decomposition in carbapenem chemistry. The ability to isolate the product simply by filtration and drying ensures that the final material retains high purity levels above 98.0%, as the impurities remain dissolved in the mother liquor or are removed during the initial catalyst filtration step.
How to Synthesize Biapenem Efficiently
The synthesis of biapenem via this patented route offers a standardized protocol that balances reaction kinetics with operational simplicity, making it highly suitable for transfer to commercial manufacturing suites. The process begins with the careful selection of the aprotic solvent, ensuring it is anhydrous to prevent premature hydrolysis, followed by the dissolution of the Formula (II) intermediate at a concentration that maximizes reactor throughput. The detailed standardized synthesis steps见下方的指南 outline the precise stoichiometry of catalyst loading, hydrogen pressure ramping, and the critical anti-solvent addition rates required to control crystal morphology and particle size distribution. Adhering to these parameters ensures consistent batch-to-batch reproducibility and minimizes the formation of open-ring impurities that can occur under acidic or basic extremes.
- Dissolve the protected biapenem intermediate (Formula II) in an aprotic polar organic solvent such as DMF, NMP, or DMSO.
- Perform catalytic hydrogenation using Pd/C or Pt-based catalysts at mild pressures (1-10 Kg/cm²) and temperatures (20-40°C).
- Quench the reaction with water, filter off the catalyst, and precipitate the pure biapenem product by adding a water-miscible organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aprotic solvent hydrogenation technology translates into tangible improvements in cost structure and supply reliability. The most immediate impact is seen in capital expenditure optimization; because the process requires significantly less solvent volume per kilogram of product, existing pressure reactors can be utilized much more intensively, effectively multiplying production capacity without the need for new hardware investments. This efficiency gain is compounded by the elimination of expensive downstream processing units such as large-scale freeze-dryers or ion-exchange resin columns, which are both capital-intensive and maintenance-heavy. Additionally, the simplified workflow reduces the overall cycle time per batch, allowing for faster turnover and more responsive fulfillment of market demand for high-purity biapenem. The reduction in solvent usage also aligns with increasingly stringent environmental regulations, lowering the burden on waste treatment facilities and reducing the costs associated with solvent recovery and disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in solvent consumption and the removal of complex purification steps. By avoiding the use of macroporous adsorbent resins and eliminating the need for lyophilization, manufacturers can achieve substantial savings in both consumable materials and energy usage. The ability to recover and recycle the aprotic polar solvents further enhances the cost-efficiency profile, as these solvents, while initially more expensive than water, are used in such small quantities that their recovery is economically viable and environmentally sound. Moreover, the higher yield obtained through this method, consistently exceeding 72% compared to lower yields in degradation-prone conventional methods, directly improves the cost of goods sold by maximizing the output from expensive starting materials.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route mitigates risks associated with production bottlenecks and equipment downtime. The mild reaction conditions and the absence of corrosive buffer systems reduce wear and tear on reactor vessels and piping, leading to longer equipment lifecycles and fewer unplanned maintenance shutdowns. The simplified isolation procedure, which relies on standard filtration and drying rather than specialized membrane filtration or low-temperature crystallization, makes the process less susceptible to utility failures such as refrigeration loss. This operational stability ensures a continuous and predictable supply of biapenem intermediates, which is critical for maintaining the production schedules of downstream antibiotic formulation plants and preventing stockouts in the global pharmaceutical market.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more manageable due to the reduced inventory of hazardous solvents and the lower exothermic potential associated with smaller reaction volumes. The process generates significantly less wastewater compared to aqueous-buffer methods, as the bulk of the reaction mass consists of recoverable organic solvents rather than salt-laden aqueous streams. This reduction in effluent volume simplifies compliance with environmental discharge permits and lowers the operational costs of wastewater treatment plants. Furthermore, the high purity of the crude product obtained directly from precipitation reduces the need for reprocessing or re-crystallization, minimizing the generation of solid chemical waste and supporting the industry's move towards greener, more sustainable manufacturing practices for complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biapenem synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and quality outcomes expected when transitioning from traditional zinc powder or aqueous hydrogenation methods to this advanced aprotic solvent protocol. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What are the primary advantages of using aprotic polar solvents for biapenem deprotection?
A: Using aprotic polar solvents like DMF or NMP significantly increases the solubility of the ionic raw material, allowing for much higher reaction concentrations. This reduces the required reactor volume drastically compared to traditional aqueous-organic mixed solvent systems, thereby improving production throughput and reducing solvent recovery costs.
Q: Does this method require ion-exchange resin purification?
A: No, one of the key innovations of this process is the elimination of macroporous adsorbent resin column chromatography. The product can be directly precipitated from the reaction mixture by adjusting solvent polarity with water and alcohols, which simplifies the workflow and avoids product degradation associated with concentration steps.
Q: What is the expected yield and purity of biapenem using this catalytic method?
A: According to the patent data, this method consistently achieves yields exceeding 72%, with specific embodiments reaching up to 78.6%. The final product purity is reported to be greater than 98.0%, meeting stringent requirements for antibiotic intermediates without the need for freeze-drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biapenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for life-saving antibiotics like biapenem. Our technical team has extensively analyzed the advancements presented in CN101768174B and possesses the expertise to implement this aprotic solvent hydrogenation strategy at an industrial scale. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant with cGMP standards. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of biapenem intermediate meets the highest quality benchmarks required by global regulatory authorities.
We invite pharmaceutical partners to collaborate with us to leverage these technological improvements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and current manufacturing constraints. We encourage potential clients to contact us to obtain specific COA data from our pilot runs and to discuss route feasibility assessments that demonstrate how our optimized processes can enhance your overall production efficiency and profitability.
