Advanced Chromatography-Free Manufacturing Route for High-Purity Biapenem Intermediates
Advanced Chromatography-Free Manufacturing Route for High-Purity Biapenem Intermediates
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic viability, particularly for complex beta-lactam antibiotics like Biapenem. Patent CN101007816A introduces a transformative approach to Biapenem synthesis that addresses critical bottlenecks in traditional manufacturing. By strategically utilizing methanesulfonic acid salt formation, this method eliminates the need for column chromatography in key purification steps, a breakthrough that significantly enhances process throughput. This technical insight report analyzes the mechanistic advantages of this route, providing R&D directors and procurement leaders with a clear understanding of its potential for cost reduction in pharmaceutical intermediates manufacturing. The transition from chloride salts to mesylate salts fundamentally changes the physical properties of the intermediates, enabling crystallization-based purification which is far more amenable to industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Biapenem and related carbapenems has relied heavily on preparative column chromatography to isolate sensitive intermediates and the final active pharmaceutical ingredient. Conventional routes, such as those described in earlier literature by Kumagai et al., often involve the generation of side chains that are difficult to purify due to their polarity and instability. The reliance on silica gel chromatography introduces several severe limitations for commercial production. Firstly, it creates a significant bottleneck in throughput, as column loading capacities are inherently limited compared to crystallization vessels. Secondly, the extensive use of organic solvents required for elution generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs. Furthermore, the mechanical stress and exposure to silica can sometimes lead to product degradation or isomerization, negatively impacting the overall yield and purity profile of the high-purity API intermediate.
The Novel Approach
The methodology outlined in CN101007816A circumvents these challenges by introducing a specific salt exchange strategy early in the synthesis. By converting the bis(6,7-dihydro-5H-pyrazolo[1,2a][1,2,4]triazolium-6-yl)disulfide dichloride into its corresponding dimethanesulfonate salt, the inventors have created an intermediate that possesses superior crystallinity. This physical property allows the intermediate to be purified simply by precipitation and filtration, completely bypassing the need for chromatographic separation. 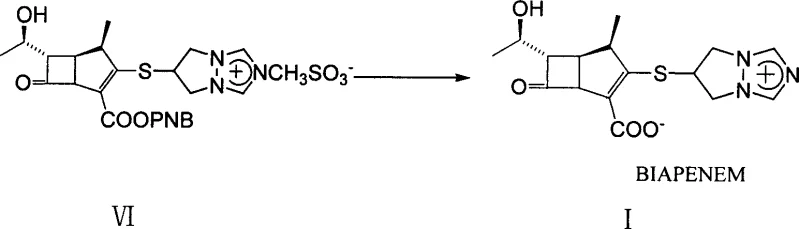 Moreover, this advantage extends to the final step of the synthesis. The hydrogenation of the protected condensate to yield the final Biapenem product is also designed to proceed without column chromatography. Instead, the process utilizes a combination of solvent evaporation, adsorption with agents like diatomaceous earth, and recrystallization. This holistic approach to purification ensures that the commercial scale-up of complex pharmaceutical intermediates is not hindered by purification bottlenecks, offering a reliable pathway for consistent supply.
Moreover, this advantage extends to the final step of the synthesis. The hydrogenation of the protected condensate to yield the final Biapenem product is also designed to proceed without column chromatography. Instead, the process utilizes a combination of solvent evaporation, adsorption with agents like diatomaceous earth, and recrystallization. This holistic approach to purification ensures that the commercial scale-up of complex pharmaceutical intermediates is not hindered by purification bottlenecks, offering a reliable pathway for consistent supply.
Mechanistic Insights into Salt-Mediated Purification and Hydrogenolysis
The core innovation of this process lies in the manipulation of ionic interactions to drive purification. In the initial step, the reaction of the disulfide dichloride with methanesulfonic acid in methanol facilitates an anion exchange where chloride ions are replaced by mesylate anions. The resulting dimethanesulfonate salt exhibits a distinct solubility curve in acetone compared to the starting dichloride and potential organic impurities. When acetone is added to the concentrated methanolic solution, the mesylate salt precipitates as an off-white solid, leaving impurities in the mother liquor. This crystallization mechanism is thermodynamically driven and highly selective, ensuring that the subsequent reduction step begins with a high-purity substrate. The reduction of the disulfide bond to the free thiol using tributylphosphine is then carried out in a biphasic system of tetrahydrofuran and water. The presence of the mesylate counter-ion stabilizes the resulting thiol species, preventing premature oxidation or decomposition that might occur with less stable salt forms.
Following the coupling of the thiol side chain with the beta-lactam core, the final deprotection step involves catalytic hydrogenolysis. This step is critical for removing the p-nitrobenzyl protecting group to reveal the active carboxylic acid. 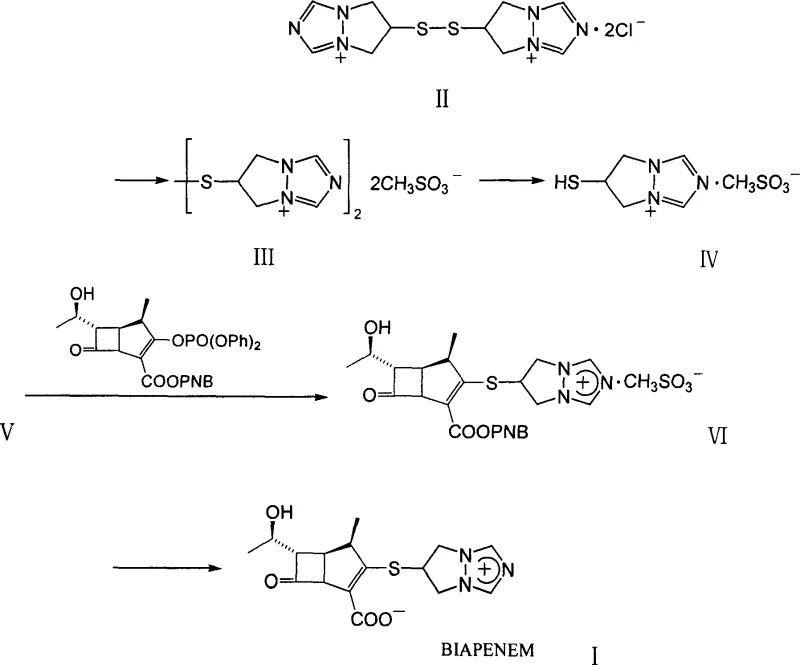 The patent specifies the use of palladium on carbon (Pd-C) in a mixture of n-butanol and water, with pH carefully controlled using phosphate buffers. Maintaining the pH between 4.5 and 5.5 is crucial to prevent the hydrolysis of the sensitive beta-lactam ring while ensuring efficient hydrogenolysis. Unlike traditional methods that might require chromatography to separate the product from the catalyst and deprotection byproducts, this method employs a sophisticated workup procedure. After filtration of the catalyst, the solution is concentrated, and specific adsorbents like anhydrous magnesium sulfate and diatomaceous earth are used to trap residual impurities and water. The final recrystallization from absolute ethanol yields Biapenem with the requisite pharmaceutical purity, demonstrating how careful control of reaction conditions and workup parameters can replace complex separation technologies.
The patent specifies the use of palladium on carbon (Pd-C) in a mixture of n-butanol and water, with pH carefully controlled using phosphate buffers. Maintaining the pH between 4.5 and 5.5 is crucial to prevent the hydrolysis of the sensitive beta-lactam ring while ensuring efficient hydrogenolysis. Unlike traditional methods that might require chromatography to separate the product from the catalyst and deprotection byproducts, this method employs a sophisticated workup procedure. After filtration of the catalyst, the solution is concentrated, and specific adsorbents like anhydrous magnesium sulfate and diatomaceous earth are used to trap residual impurities and water. The final recrystallization from absolute ethanol yields Biapenem with the requisite pharmaceutical purity, demonstrating how careful control of reaction conditions and workup parameters can replace complex separation technologies.
How to Synthesize Biapenem Efficiently
The synthesis of Biapenem via this improved route requires precise control over stoichiometry, temperature, and solvent composition to maximize the benefits of the chromatography-free design. The process begins with the strict preparation of the mesylate salt, where the ratio of methanesulfonic acid to the dichloride precursor must be optimized to ensure complete conversion without excess acid carryover. Subsequent reduction and coupling steps demand anhydrous conditions where appropriate, particularly during the activation of the beta-lactam core. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and temperature profiles for each stage from salt formation to final recrystallization, are provided in the guide below for technical reference.
- Convert bis(6,7-dihydro-5H-pyrazolo[1,2a][1,2,4]triazolium-6-yl)disulfide dichloride into its dimethanesulfonate salt (III) using methanesulfonic acid in methanol, followed by crystallization with acetone.
- Reduce the disulfide salt (III) to the corresponding thiol mesylate (IV) using tributylphosphine in a tetrahydrofuran and water mixture at low temperature.
- Couple the thiol mesylate (IV) with the bicyclic beta-lactam parent nucleus (V) using diisopropylethylamine to form the protected condensate (VI).
- Perform catalytic hydrogenation on the condensate (VI) using Pd-C in n-butanol and water, followed by filtration and recrystallization to obtain pure Biapenem without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift away from column chromatography represents a fundamental improvement in the economics of API production. Traditional chromatographic processes are capital intensive, requiring large columns, pumps, and significant volumes of high-grade solvents. By replacing these unit operations with crystallization and filtration, the novel process drastically simplifies the equipment requirements. This simplification translates directly into reduced capital expenditure for new production lines and lower maintenance costs for existing facilities. Furthermore, the reduction in solvent consumption aligns with green chemistry principles, potentially lowering the environmental compliance burden and waste disposal fees associated with manufacturing. The ability to produce high-purity intermediates without the variability inherent in manual or automated column packing enhances the reliability of the supply chain, ensuring consistent batch-to-batch quality.
- Cost Reduction in Manufacturing: The elimination of column chromatography removes one of the most expensive steps in fine chemical synthesis. Silica gel is a consumable cost that adds up quickly at scale, and the solvents required for elution often constitute a major portion of the variable manufacturing costs. By utilizing crystallization, the process leverages the intrinsic physical properties of the molecules for purification, which is inherently cheaper. Additionally, the recovery of solvents like acetone and ethanol is more straightforward in a crystallization workflow compared to the complex mixtures obtained from chromatographic fractions. This structural change in the process flow leads to substantial cost savings without compromising the quality of the final Biapenem product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the bottlenecks created by slow purification steps. Chromatography is time-consuming and limits the number of batches that can be processed in a given timeframe. The new method accelerates the production cycle time significantly, allowing for faster turnover of reactor vessels. This increased throughput capability means that suppliers can respond more agilely to market demand fluctuations. Moreover, the robustness of crystallization processes reduces the risk of batch failures due to column channeling or poor separation, thereby enhancing the overall reliability of the supply chain for this critical antibiotic intermediate.
- Scalability and Environmental Compliance: Scaling up a chromatographic process from the lab to the plant is notoriously difficult and risky. In contrast, crystallization scales linearly and predictably. The ability to simply increase vessel size without changing the fundamental physics of the separation makes this technology ideal for commercial scale-up. From an environmental perspective, the reduction in solvent usage and the elimination of spent silica gel waste contribute to a smaller environmental footprint. This aligns with the increasing regulatory pressure on pharmaceutical manufacturers to adopt sustainable practices, making this process not only economically attractive but also future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on how this method compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new manufacturing protocol.
Q: How does the mesylate salt formation improve the Biapenem synthesis yield?
A: The conversion of the dichloride salt to the dimethanesulfonate salt significantly alters the solubility profile of the intermediate. This allows for purification via simple crystallization rather than labor-intensive column chromatography, thereby reducing material loss and improving overall process efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the elimination of column chromatography steps makes this method highly scalable. Crystallization and filtration are standard unit operations in large-scale chemical manufacturing, offering better reproducibility and lower operational complexity compared to preparative chromatography.
Q: What are the key impurities controlled in this novel route?
A: The process controls impurities primarily through the high purity of the crystallized mesylate intermediate and the selective nature of the catalytic hydrogenation. The final recrystallization step further ensures the removal of residual catalysts and organic impurities, meeting stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biapenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antibiotics like Biapenem. Our team of expert chemists has extensively analyzed the technological advancements presented in CN101007816A and is well-positioned to implement this chromatography-free strategy. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing output. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Biapenem intermediate meets the highest global pharmaceutical standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized process can benefit your supply chain. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced Biapenem synthesis technology into your portfolio.
