Advanced DDQ-Mediated Synthesis of Meso-Substituted Porphyrin Derivatives for Commercial Scale
Advanced DDQ-Mediated Synthesis of Meso-Substituted Porphyrin Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for synthesizing complex aromatic systems, particularly porphyrin derivatives which serve as critical intermediates for next-generation photosensitizers. Patent CN110698487B introduces a groundbreaking preparation method for DDQ-mediated porphyrin meso-position carbon-oxygen and carbon-carbon coupled derivatives that fundamentally shifts the paradigm away from traditional transition metal catalysis. This innovation addresses the long-standing challenges of low yields and harsh reaction conditions associated with conventional porphyrin functionalization. By leveraging the oxidative power of 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ), this technology enables the direct modification of 5,15-diaryl substituted metalloporphyrins without the need for pre-functionalized groups. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates while drastically reducing the environmental footprint of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the derivatization of porphyrin compounds at the meso-position has been fraught with synthetic difficulties, primarily relying on transition metal-catalyzed cross-coupling reactions such as Suzuki-Miyaura or Kumada couplings. These traditional methods typically necessitate the use of expensive and toxic catalysts based on palladium, nickel, or iron, which not only inflate the raw material costs but also introduce severe downstream purification challenges. The removal of trace heavy metals to meet stringent pharmaceutical purity specifications often requires additional processing steps involving specialized scavengers or repeated chromatography, leading to substantial material loss and extended production timelines. Furthermore, many existing protocols require the porphyrin substrate to be pre-functionalized with halogen groups, adding extra synthetic steps that decrease the overall atom economy and generate unnecessary chemical waste. The reliance on sensitive organometallic reagents also imposes strict operational constraints, such as the need for anhydrous conditions and inert atmospheres, which complicates the scale-up process for commercial manufacturing.
The Novel Approach
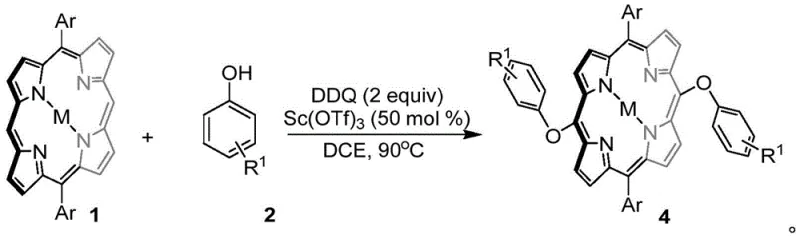
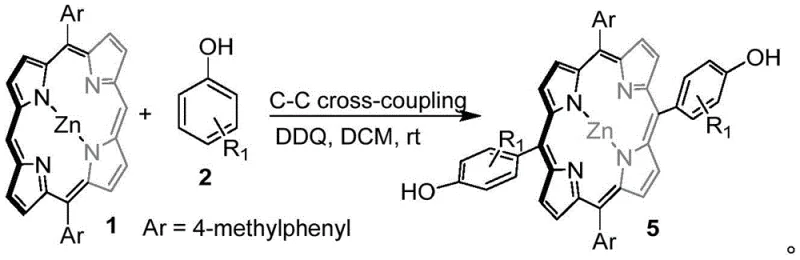
In stark contrast, the novel DDQ-mediated oxidative cross-coupling strategy described in the patent offers a streamlined, metal-free alternative that directly couples phenol derivatives with metalloporphyrins. This method successfully avoids self-coupling side reactions and the unwanted coupling between nucleophiles and oxidants, ensuring high selectivity for the desired meso-substituted products. The process is remarkably versatile, capable of forming either carbon-oxygen or carbon-carbon bonds simply by adjusting the reaction conditions and substrate sterics, thereby expanding the structural diversity accessible to medicinal chemists. By introducing polar oxygen atoms directly into the porphyrin conjugated system, this approach not only enhances the water solubility of the final derivatives but also lays a solid foundation for the development of new, more effective photosensitizers for photodynamic therapy. The elimination of transition metals simplifies the workflow significantly, making it an attractive option for reliable porphyrin derivative suppliers aiming to reduce lead time for high-purity intermediates.
Mechanistic Insights into DDQ-Mediated Oxidative Coupling
The core of this technological breakthrough lies in the unique ability of DDQ to act as a potent single-electron oxidant that facilitates the generation of radical species under mild conditions. In the presence of a Lewis acid catalyst such as scandium triflate (Sc(OTf)3), the reaction proceeds through a mechanism where the phenol substrate is activated to form a phenoxy radical or cationic intermediate, which then attacks the electron-rich meso-position of the metalloporphyrin ring. This oxidative cross-coupling pathway is highly efficient because it bypasses the need for transmetallation steps typical of palladium cycles, instead relying on the intrinsic nucleophilicity of the porphyrin macrocycle. The chemoselectivity of the reaction is finely tuned by the steric environment of the phenolic hydroxyl group; less hindered phenols tend to favor carbon-oxygen bond formation, while bulky substrates promote carbon-carbon coupling due to the difficulty of stabilizing the oxygen-centered radical. This mechanistic nuance allows for precise control over the product distribution, enabling the synthesis of distinct structural analogues from the same starting materials simply by varying the phenol substituent.
From an impurity control perspective, the absence of transition metals inherently reduces the risk of metal-contaminated byproducts, which is a critical quality attribute for API intermediates. The reaction conditions, typically involving dichloromethane or 1,2-dichloroethane as solvents at temperatures ranging from room temperature to 90°C, are温和 enough to preserve the integrity of the sensitive porphyrin macrocycle while providing sufficient energy to drive the oxidation. The use of Sc(OTf)3 as a co-catalyst further enhances the electrophilicity of the oxidant, ensuring complete conversion of the starting porphyrin within a short timeframe, usually around 3 hours for C-O coupling. This high efficiency minimizes the formation of oligomeric side products or decomposition materials, resulting in a cleaner crude profile that simplifies the subsequent purification via column chromatography. The robustness of this mechanism across various metal-coordinated porphyrins, including Zinc, Copper, and Nickel complexes, demonstrates its broad applicability for cost reduction in electronic chemical manufacturing and pharmaceutical synthesis.
How to Synthesize Meso-Substituted Porphyrin Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for producing these valuable compounds, emphasizing simplicity and scalability. The process begins with the dissolution of the metalloporphyrin substrate and the chosen phenolic coupling partner in a chlorinated solvent, followed by the sequential addition of the oxidant and Lewis acid catalyst. Reaction monitoring is straightforward, typically relying on TLC or HPLC to confirm the disappearance of the starting porphyrin material, after which the solvent is removed under reduced pressure. The resulting crude solid is then subjected to standard silica gel column chromatography to isolate the pure meso-substituted product, with yields often exceeding 60% and reaching as high as 93% for optimized substrates. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Dissolve 0.05mmol of porphyrin substrate and 0.25mmol of phenol in 50mL of 1,2-dichloroethane.
- Add 50 mol% Sc(OTf)3 and 0.1mmol of DDQ, then heat the mixture to 90°C and stir for 3 hours.
- Remove solvent via spin-drying and purify the crude product using column chromatography to isolate the target porphyrin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DDQ-mediated technology translates into tangible strategic benefits that go beyond mere technical feasibility. The most immediate impact is seen in the drastic simplification of the supply chain, as the process eliminates the dependency on volatile and expensive transition metal catalysts that are subject to geopolitical supply risks and price fluctuations. By removing the need for palladium or nickel, manufacturers can avoid the costly and time-consuming heavy metal clearance steps that often bottleneck production schedules, thereby enhancing supply chain reliability and ensuring consistent delivery timelines. Furthermore, the use of readily available phenol derivatives and common organic solvents means that raw material sourcing is stable and cost-effective, supporting the commercial scale-up of complex porphyrin derivatives without requiring specialized infrastructure.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a direct and significant reduction in raw material costs, as DDQ and scandium triflate are substantially cheaper and used in catalytic or stoichiometric amounts compared to the high loadings often required for palladium systems. Additionally, the simplified workup procedure reduces the consumption of purification media and solvents, leading to lower waste disposal costs and improved overall process economics. The high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, which further contributes to cost savings by maximizing the yield of the desired active pharmaceutical ingredient intermediate. This economic efficiency makes the method highly competitive for large-volume production where margin optimization is critical.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can often be a logistical challenge, but the reagents required for this DDQ-mediated process are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source dependencies, ensuring a continuous flow of materials for manufacturing operations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality or environmental factors, providing a more stable and predictable production output. Consequently, partners can rely on a more resilient supply chain capable of meeting fluctuating market demands for photosensitizers and related fine chemicals.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Scaling this process from laboratory to pilot plant and eventually to full commercial production is straightforward, as it does not require specialized high-pressure equipment or extreme temperatures. The reduced generation of hazardous metal-containing waste simplifies effluent treatment and lowers the environmental compliance burden for manufacturing facilities. This sustainability advantage not only reduces operational risks but also enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, based on the specific advantages and mechanisms detailed in the patent documentation. Understanding these aspects is crucial for evaluating the feasibility of integrating this technology into existing production workflows. The answers provided reflect the empirical data and theoretical framework established by the inventors, offering clarity on performance and applicability.
Q: What are the advantages of using DDQ over transition metal catalysts for porphyrin modification?
A: The DDQ-mediated method eliminates the need for expensive transition metals like palladium or nickel, thereby avoiding complex heavy metal removal steps and reducing overall production costs while maintaining high chemoselectivity.
Q: Can this method achieve both carbon-oxygen and carbon-carbon bonds?
A: Yes, the process is highly versatile; it forms carbon-oxygen bonds under Lewis acid catalysis at elevated temperatures and carbon-carbon bonds at room temperature depending on the steric hindrance of the phenolic substrate.
Q: Is this synthesis method suitable for large-scale manufacturing of photosensitizers?
A: Absolutely, the reaction utilizes commercially available reagents, operates under mild conditions without inert gas protection, and offers high yields, making it ideal for scaling from laboratory to industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this DDQ-mediated synthesis technology for the production of advanced photosensitizers and functional materials. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of porphyrin derivative delivered meets the highest standards required for pharmaceutical and electronic applications. We are committed to leveraging our technical expertise to optimize this metal-free route, delivering high-quality intermediates that empower our clients' innovation pipelines.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your product portfolio and reduce your manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and help you secure a competitive edge in the global market.
