Scalable Decitabine Production: Advanced Glycosylation and Isomer Separation Strategies
Introduction to Advanced Decitabine Manufacturing
The global demand for epigenetic therapies, particularly for the treatment of myelodysplastic syndromes (MDS), has placed Decitabine (5-aza-2'-deoxycytidine) at the forefront of oncology drug development. As a potent DNA methyltransferase inhibitor, its clinical efficacy is well-documented, yet its complex molecular architecture presents significant hurdles for large-scale production. Patent CN101497639B introduces a transformative approach to synthesizing this critical nucleoside analogue, addressing the longstanding bottlenecks of low yield and difficult purification that have plagued the industry. By shifting away from precious metal catalysts and cumbersome chromatographic separations, this technology offers a robust pathway for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolios. The core innovation lies in a strategic combination of p-toluoyl protection and a novel fractional crystallization technique that isolates the desired beta-anomer prior to deprotection.
This comprehensive report analyzes the technical merits of this patented process, providing deep insights for R&D directors evaluating process feasibility, procurement managers assessing cost structures, and supply chain heads planning for commercial scalability. The methodology described herein not only enhances the stereochemical control during the glycosylation step but also streamlines the downstream processing, thereby reducing the overall environmental footprint and operational complexity associated with producing high-purity Decitabine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Decitabine has been fraught with inefficiencies that render many routes unsuitable for industrial application. One prominent legacy method involves the isocyanate pathway, which relies heavily on silver isocyanate (AgNCO) to facilitate the formation of the nucleoside bond. As illustrated in the reaction scheme below, this approach is not only economically prohibitive due to the high cost of silver salts but also generates substantial heavy metal waste, complicating environmental compliance and waste disposal protocols.
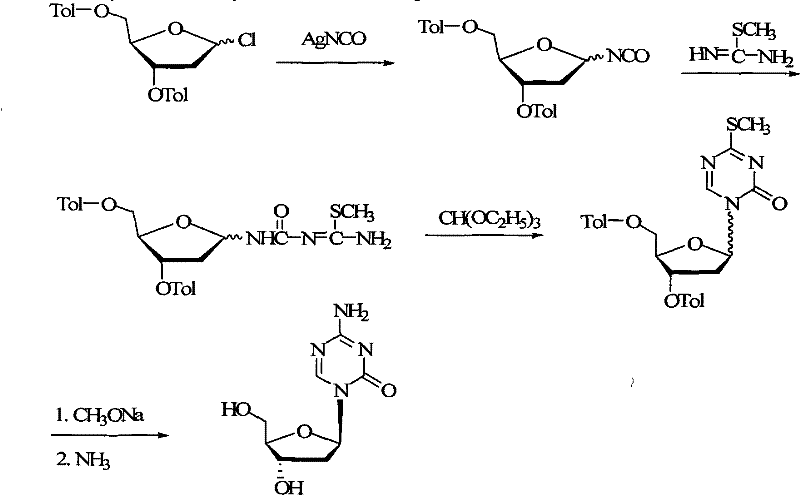
Furthermore, conventional routes often result in a mixture of alpha and beta anomers that are notoriously difficult to separate. Traditional strategies frequently employ column chromatography to isolate the active beta-isomer. While effective on a laboratory scale, chromatographic separation is a major bottleneck for commercial scale-up of complex pharmaceutical intermediates, leading to low throughput, high solvent consumption, and significant product loss. Other methods utilizing Fmoc protecting groups suffer from stability issues and high reagent costs, while acetyl-protected routes often yield poor stereocontrol, necessitating multiple recrystallization steps that erode overall yield.
The Novel Approach
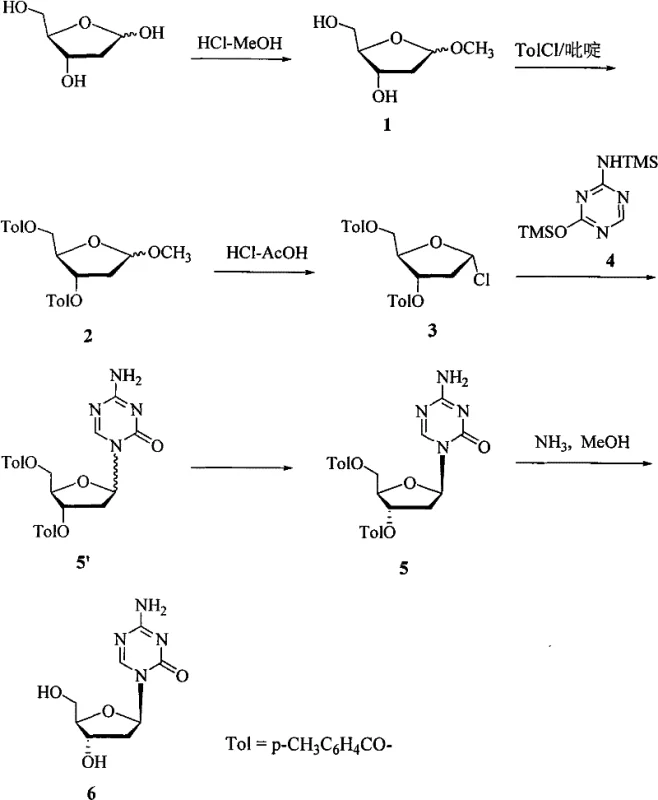
In stark contrast to these legacy techniques, the methodology outlined in CN101497639B leverages a p-toluoyl protection strategy that fundamentally alters the purification landscape. By employing p-toluoyl groups on the sugar moiety, the process introduces a chromophore that simplifies reaction monitoring via Thin Layer Chromatography (TLC), allowing for precise endpoint determination without sophisticated instrumentation. More critically, this protection scheme enables a unique purification strategy where the alpha/beta isomer mixture is subjected to fractional crystallization before the removal of protecting groups. This pre-deprotection separation is a game-changer, as it allows for the isolation of the pure beta-isomer (Compound 5) using common solvents like acetonitrile or ethyl acetate, completely bypassing the need for preparative HPLC or silica gel columns.
Mechanistic Insights into Vorbrüggen-style Glycosylation and Crystallization
The heart of this synthetic success lies in the meticulous control of the glycosylation reaction and the subsequent physical separation of isomers. The process begins with the silylation of 5-azacytosine using hexamethyldisilazane (HMDS) in the presence of ammonium sulfate, generating a highly reactive bis-trimethylsilyl derivative. This activated base then reacts with the glycosyl donor, 1-chloro-2-deoxy-3,5-di-O-p-toluoyl-alpha-D-ribofuranose (Compound 3). The choice of solvent during this coupling step is paramount; the patent data indicates that using acetonitrile favors the formation of the beta-isomer with an alpha:beta ratio of approximately 1:1.3, whereas chloroform yields a nearly 1:1 mixture. This solvent-dependent stereoselectivity suggests that polarity and solvation effects play a crucial role in stabilizing the transition state leading to the desired anomeric configuration.
Following the coupling reaction, the crude mixture contains both alpha and beta protected nucleosides. The innovative aspect of this technology is the exploitation of solubility differences between these diastereomers. By dissolving the mixture in acetonitrile or ethyl acetate and inducing crystallization, the less soluble beta-isomer precipitates out with high purity (>95%). This physical separation is far more efficient than chemical differentiation. Once the pure beta-protected intermediate is secured, the final deprotection is carried out using saturated ammonia in methanol at 0°C. This mild condition ensures the integrity of the sensitive triazine ring while efficiently cleaving the ester bonds, resulting in the final Decitabine product with a purity exceeding 98%.
How to Synthesize Decitabine Efficiently
The synthesis of Decitabine via this patented route involves a sequence of six distinct chemical transformations, starting from readily available 2-deoxy-D-ribose. The process is designed to maximize yield at each stage while minimizing the accumulation of impurities that are difficult to remove later. The initial steps focus on preparing the glycosyl donor with high stereochemical fidelity, followed by the coupling with the silylated base. The critical purification occurs immediately after coupling, ensuring that only the correct isomer proceeds to the final deprotection. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Methylation of 2-deoxy-D-ribose followed by acylation with p-toluoyl chloride to form the protected ribofuranose intermediate.
- Chlorination of the protected sugar to generate the glycosyl donor, followed by silylation of 5-azacytosine using HMDS.
- Glycosylation reaction to form the alpha/beta isomer mixture, followed by fractional crystallization to isolate the pure beta-isomer before final deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from chromatography-dependent methods to crystallization-based purification represents a substantial opportunity for cost reduction in pharmaceutical manufacturing. The elimination of silica gel columns and the associated large volumes of elution solvents drastically reduces the operational expenditure (OPEX) related to solvent purchase, recovery, and disposal. Furthermore, the removal of precious metal catalysts like silver from the process flow mitigates the risk of heavy metal contamination in the final API, thereby reducing the cost and complexity of downstream purification and quality control testing.
- Cost Reduction in Manufacturing: The replacement of expensive reagents such as silver isocyanate and Fmoc chlorides with commodity chemicals like p-toluoyl chloride and HMDS leads to significant raw material savings. Additionally, the ability to isolate the pure beta-isomer through simple recrystallization rather than preparative chromatography significantly lowers the cost per kilogram of the final product. This streamlined workflow reduces labor hours and equipment usage, contributing to a more lean and cost-effective production model that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 2-deoxy-D-ribose and standard organic solvents ensures a stable supply chain不受制于 specialty reagent shortages. By avoiding reagents that are difficult to source domestically or have volatile pricing, manufacturers can secure long-term supply contracts with greater confidence. The robustness of the crystallization step also means that the process is less sensitive to minor variations in reaction conditions, leading to more consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The transition from batch chromatography to crystallization is a critical enabler for commercial scale-up of complex nucleoside intermediates. Crystallization is inherently easier to scale from kilograms to tons compared to column chromatography, which faces physical limitations in bed height and flow rates. Moreover, the reduction in solvent waste and the absence of heavy metal residues align with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and reducing the environmental liability associated with large-scale chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decitabine synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and process robustness.
Q: How does this patent improve isomer separation compared to traditional methods?
A: Unlike prior art that relies on difficult column chromatography after deprotection, this method utilizes fractional crystallization of the protected intermediate (compound 5') using acetonitrile or ethyl acetate. This allows for the isolation of the pure beta-isomer before the final deprotection step, significantly simplifying purification.
Q: What are the key cost advantages of using p-toluoyl protecting groups?
A: The use of p-toluoyl groups eliminates the need for expensive reagents like silver isocyanate (AgNCO) or Fmoc chlorides found in older routes. Additionally, the aromatic nature of the toluoyl group acts as a chromophore, facilitating easy reaction monitoring via TLC, which reduces process development time and quality control costs.
Q: What purity levels can be achieved with this synthetic route?
A: The patent specifies that the final Decitabine product achieves a purity of greater than 98% (detected at UV 254nm). The intermediate beta-isomer obtained after crystallization also reaches a purity of over 95%, ensuring a high-quality final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Decitabine depends on a partner who can bridge the gap between innovative patent chemistry and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this crystallization-based route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the complex analytical requirements of nucleoside analogues, guaranteeing that every batch meets the highest international standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply of high-purity Decitabine is secure, cost-effective, and ready for clinical or commercial deployment.
