Scalable Decitabine Manufacturing: A Novel Pivaloyl Protection Strategy for High-Purity API Intermediates
Introduction to Advanced Decitabine Manufacturing
The pharmaceutical industry's demand for high-purity oncology intermediates continues to drive innovation in synthetic methodology, particularly for complex nucleoside analogs like Decitabine. Patent CN101712708A discloses a transformative approach to synthesizing Decitabine (5-aza-2'-deoxycytidine), a critical active pharmaceutical ingredient used in the treatment of myelodysplastic syndromes (MDS) and acute leukemia. This technology addresses long-standing bottlenecks in nucleoside chemistry by replacing traditional acetyl protection groups with robust pivaloyl moieties. The strategic implementation of this steric bulk not only enhances reaction selectivity but fundamentally alters the downstream processing requirements. By enabling the elimination of column chromatography in favor of direct recrystallization, this method offers a compelling value proposition for manufacturers seeking to optimize both yield and operational efficiency in the production of high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
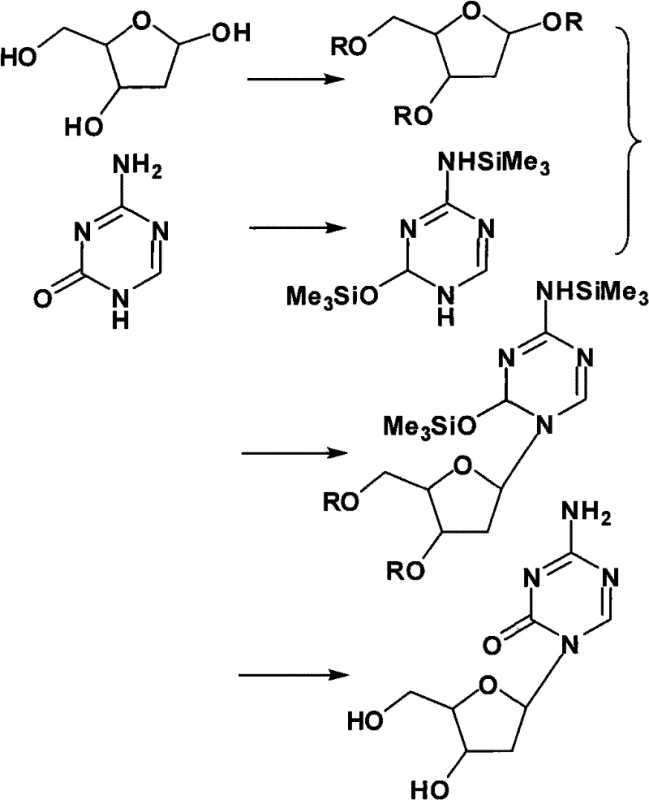
Historically, the industrial preparation of Decitabine has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional routes often rely on starting materials such as 2-halo-3-hydroxyl-4-hydroxymethylfurans, which are notoriously difficult to source commercially and exhibit poor availability in the global supply chain. Furthermore, alternative methods utilizing 2-deoxy-D-ribose protected with acetyl groups require the use of excessive amounts of trimethylsilyl trifluoromethanesulfonate (TMSOTf) to drive the glycosylation reaction. This reliance on stoichiometric or near-stoichiometric quantities of expensive Lewis acids drastically inflates raw material costs. Perhaps most critically, these conventional acetyl-protected routes typically generate complex mixtures of anomers and byproducts that necessitate purification via silica gel column chromatography. This purification step is a major bottleneck in process chemistry, consuming vast volumes of solvents, generating hazardous waste, and severely limiting the throughput capacity required for commercial manufacturing.
The Novel Approach
The methodology outlined in CN101712708A represents a paradigm shift by introducing pivaloyl (trimethylacetyl) groups as the protecting strategy for the ribose sugar. This seemingly simple substitution yields profound effects on the reaction kinetics and thermodynamics. The bulky nature of the pivaloyl group stabilizes the sugar intermediate and directs the subsequent coupling reaction with higher regioselectivity and stereoselectivity. Consequently, the process requires only a catalytic amount of TMSOTf (0.1 to 0.3 equivalents) rather than the large excesses needed in acetyl-based routes. This reduction in catalyst loading is not merely a cost-saving measure; it simplifies the workup procedure and reduces the formation of side products. The culmination of these improvements is a crude product of sufficient purity that it bypasses chromatographic purification entirely, relying instead on a scalable recrystallization process using dimethyl sulfoxide (DMSO) and anhydrous methanol.
Mechanistic Insights into Pivaloyl-Mediated Glycosylation
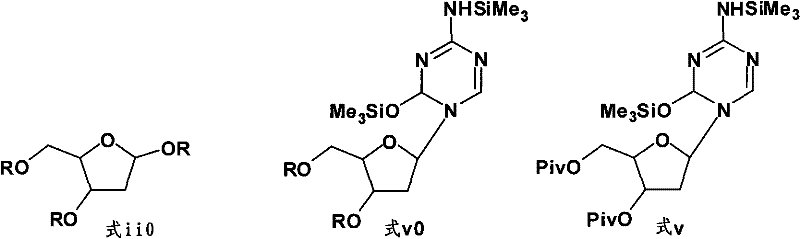
The core of this synthetic advancement lies in the electronic and steric influence of the pivaloyl group during the Vorbrüggen glycosylation step. In the initial phase, 2-deoxy-D-ribose is fully protected at the 1, 3, and 5 positions with pivaloyl groups using trimethylacetic anhydride. Unlike smaller acetyl groups, the tert-butyl moiety of the pivaloyl group exerts a significant steric shield around the sugar ring. When this protected sugar reacts with the bis-silylated 5-azacytosine derivative in the presence of TMSOTf, the activation of the anomeric center proceeds with greater control. The catalyst activates the anomeric pivaloyl group to form an oxocarbenium ion intermediate, which is then attacked by the silylated nitrogen base. The steric environment created by the adjacent pivaloyl groups favors the formation of the desired beta-anomer while suppressing the formation of alpha-anomers and other structural impurities. This inherent selectivity is the mechanistic foundation that allows the reaction to proceed cleanly without generating the complex impurity profiles typical of less hindered protecting groups.
Following the coupling reaction, the deprotection step utilizes sodium methoxide in methanol to cleave the pivaloyl esters. While pivaloyl esters are generally more resistant to hydrolysis than acetyl esters, the conditions employed (25% sodium methoxide solution at 20-40°C for 16-24 hours) are sufficient to liberate the free hydroxyl groups of the Decitabine molecule without degrading the sensitive triazine ring. The resulting crude solid contains the target API along with soluble pivalate salts and minor organic impurities. The final purification mechanism relies on the differential solubility of Decitabine in a DMSO/methanol binary solvent system. Heating the crude product in DMSO ensures complete dissolution, while the controlled addition of anhydrous methanol induces supersaturation. As the solution cools, pure Decitabine crystallizes out, leaving the remaining impurities in the mother liquor. This crystallization-driven purification is far more robust and scalable than adsorption-based chromatographic methods.
How to Synthesize Decitabine Efficiently
The synthesis of Decitabine via this pivaloyl-protected route involves a logical sequence of protection, activation, coupling, and deprotection steps that are highly amenable to standard reactor setups. The process begins with the per-acylation of 2-deoxy-D-ribose, followed by the silylation of 5-azacytosine to enhance its nucleophilicity. These two intermediates are then coupled under mild Lewis acid catalysis. The detailed operational parameters, including specific molar ratios, temperature gradients, and solvent volumes optimized for maximum yield and purity, are critical for successful replication. For process chemists and engineers looking to implement this technology, the following standardized guide outlines the essential workflow derived from the patent examples.
- Protect 2-deoxy-D-ribose with pivaloyl groups using trimethylacetic anhydride to form 1,3,5-tri-O-pivaloyl-2-deoxy-D-ribose.
- Silylate 5-azacytosine with hexamethyldisilazane and trimethylchlorosilane to generate the reactive bis-silylated derivative.
- Couple the protected sugar and silylated base using a catalytic amount of TMSOTf, followed by deprotection and recrystallization from DMSO/methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chromatography-dependent synthesis to crystallization-based purification represents a substantial opportunity for cost optimization and risk mitigation. The elimination of silica gel columns removes a significant variable cost associated with consumables and solvent recovery. Furthermore, the reduction in processing time associated with skipping the column loading, elution, and fraction collection steps directly translates to increased asset utilization and faster batch turnover. This efficiency gain is crucial for meeting the tight delivery schedules often required in the pharmaceutical supply chain. Additionally, the use of readily available starting materials like 2-deoxy-D-ribose and trimethylacetic anhydride ensures a stable supply base, reducing the risk of raw material shortages that can plague more exotic synthetic routes.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in catalyst consumption. By lowering the requirement for TMSOTf from large excesses to mere catalytic quantities (0.1-0.3 equivalents), the direct material cost of the glycosylation step is significantly lowered. Moreover, the avoidance of column chromatography eliminates the need for tons of silica gel and hundreds of liters of elution solvents per kilogram of product. This reduction in solvent usage also lowers the burden on waste treatment facilities and reduces the energy costs associated with solvent distillation and recovery, leading to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: The robustness of the pivaloyl protection strategy contributes to a more reliable supply chain by minimizing batch-to-batch variability. Chromatographic purifications are often sensitive to column packing quality and operator technique, which can lead to yield fluctuations. In contrast, recrystallization is a well-understood unit operation that scales predictably from the laboratory to the pilot plant and finally to commercial production. This predictability allows for more accurate production planning and inventory management. Furthermore, the high purity achieved (>99% by HPLC) reduces the need for re-processing or re-crystallization cycles, ensuring that finished goods are ready for shipment faster and with consistent quality specifications.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The reduction in solvent volume and the elimination of silica gel waste significantly decrease the environmental footprint of the manufacturing process. This is increasingly important as regulatory bodies impose stricter limits on pharmaceutical waste discharge. The process is inherently safer and easier to scale because it avoids the pressure drops and flow restrictions associated with large-scale chromatography columns. This facilitates the commercial scale-up of complex nucleoside analogs, allowing manufacturers to ramp up production capacity to meet market demand without requiring massive capital investment in specialized purification equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decitabine synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for evaluating the technology's feasibility for your specific production needs.
Q: Why is pivaloyl protection superior to acetyl protection in Decitabine synthesis?
A: Pivaloyl groups provide greater steric bulk and stability compared to acetyl groups. This enhanced stability allows for a significant reduction in the usage of the expensive TMSOTf catalyst during the glycosylation step and improves regioselectivity, ultimately eliminating the need for costly column chromatography purification.
Q: How does this method eliminate the need for column chromatography?
A: The use of pivaloyl protection results in a cleaner reaction profile with fewer byproducts. The patent demonstrates that the crude product obtained after hydrolysis can be directly purified via recrystallization using a DMSO/methanol solvent system, achieving high purity (>99%) without silica gel chromatography.
Q: What are the scalability advantages of this route?
A: By removing the chromatography step, the process becomes significantly more amenable to large-scale industrial production. The reliance on recrystallization rather than column separation reduces solvent consumption, processing time, and operational complexity, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value oncology intermediates like Decitabine. Our technical team has extensively analyzed the pivaloyl protection strategy disclosed in CN101712708A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications capable of handling the precise crystallization controls required to achieve the >99% purity levels demonstrated in this patent.
We invite pharmaceutical partners and contract manufacturers to collaborate with us on leveraging this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can deliver high-purity Decitabine with improved lead times and cost efficiency.
