Advanced Electrocatalytic Synthesis of Benzothiophene Derivatives for Commercial Scale-up
Advanced Electrocatalytic Synthesis of Benzothiophene Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, driven by the urgent need to reduce environmental impact and lower production costs. A groundbreaking development in this arena is detailed in Chinese Patent CN114540844A, which discloses a novel preparation method for benzothiophene derivatives utilizing an innovative electrocatalytic strategy. Benzothiophene scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for drugs targeting osteoporosis, cardiovascular diseases, and various cancers, yet their traditional synthesis often relies on harsh conditions and toxic reagents. This new technology leverages electricity as a traceless reagent to drive the cyclization of alkylphenylacetylenes and substituted benzenesulfonyl hydrazides, offering a metal-free alternative that operates under remarkably mild conditions. By replacing stoichiometric chemical oxidants with electrons, this process not only minimizes waste generation but also simplifies the downstream purification workflow, addressing key pain points for both research and manufacturing teams. The implications of this discovery extend far beyond the laboratory, presenting a viable pathway for the cost-effective and environmentally responsible manufacturing of high-value pharmaceutical intermediates on a global scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzothiophene core has heavily depended on transition metal catalysis, particularly palladium-mediated processes, which, while effective, introduce significant logistical and economic burdens to the supply chain. As illustrated in the prior art reaction scheme below, conventional methods typically involve the intramolecular cyclization of dibromo-substituted alkenyl ortho-thiophenols using palladium catalysts and additional phosphorus ligands.
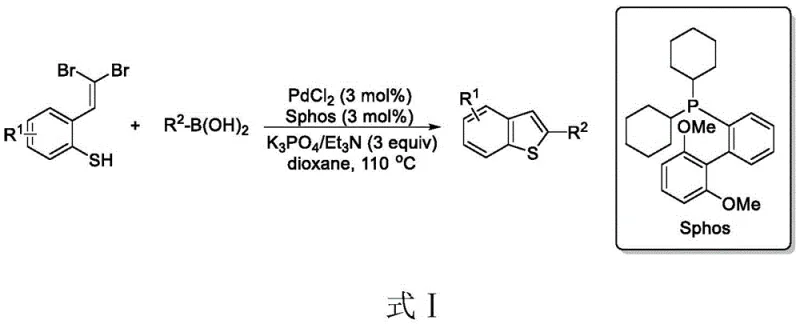
These traditional protocols suffer from several inherent drawbacks that hinder their efficiency in large-scale operations. Firstly, the requirement for precious metal catalysts like palladium drastically increases the raw material costs and necessitates rigorous post-reaction metal scavenging steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients (APIs). Secondly, these reactions often demand elevated temperatures and inert atmospheres, leading to higher energy consumption and increased operational complexity. Furthermore, the use of phosphorus ligands adds another layer of impurity profile complexity, requiring extensive chromatographic purification to remove phosphine oxides and other byproducts. The cumulative effect of these factors is a process with a high E-factor, significant safety risks associated with high-temperature operations, and a prolonged lead time for batch release due to complex quality control testing for heavy metals.
The Novel Approach
In stark contrast to the legacy methods, the electrocatalytic approach described in the patent offers a streamlined, green chemistry solution that fundamentally reimagines the bond-forming events required to access these valuable heterocycles. This novel strategy employs an undivided electrochemical cell where the oxidative cyclization is driven directly by an electric current, effectively replacing the need for external chemical oxidants or transition metal catalysts. The reaction proceeds at room temperature, eliminating the thermal energy input required by conventional thermal catalysis and significantly reducing the risk of thermal runaway or decomposition of sensitive functional groups. By utilizing graphite felt electrodes, the system ensures efficient electron transfer while maintaining chemical inertness, preventing the introduction of metal contaminants into the reaction mixture. This metal-free nature is a game-changer for procurement and quality assurance teams, as it removes the necessity for expensive metal scavengers and simplifies the analytical validation process. The result is a cleaner reaction profile with fewer side products, enabling a more direct path to high-purity benzothiophene derivatives suitable for immediate use in drug discovery and development pipelines.
Mechanistic Insights into Electrocatalytic Cyclization
The success of this electrocatalytic transformation lies in the precise interplay between the electrode potential, the solvent system, and the unique reactivity of the sulfonyl hydrazide substrate under anodic oxidation. The general reaction scheme, depicted below, outlines the conversion of alkylphenylacetylenes and substituted benzenesulfonyl hydrazides into the target benzothiophene dioxide derivatives through a radical-mediated pathway.
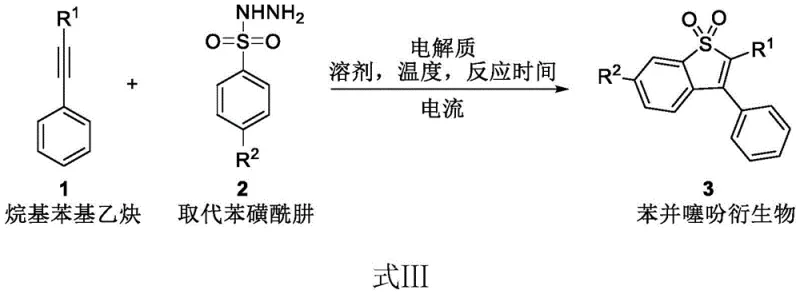
Mechanistically, the process initiates at the anode where the benzenesulfonyl hydrazide undergoes single-electron oxidation to generate a nitrogen-centered radical species. This highly reactive intermediate subsequently attacks the electron-rich triple bond of the alkylphenylacetylene, triggering a cascade of cyclization and elimination events that construct the fused thiophene ring system. The choice of solvent is critical to this mechanism; the patent specifies a binary mixture of hexafluoroisopropanol (HFIP) and nitromethane in a 47:3 volume ratio. HFIP is renowned in electro-organic synthesis for its ability to stabilize cationic and radical intermediates through strong hydrogen bonding, thereby lowering the oxidation potential of the substrate and enhancing the selectivity of the reaction. Meanwhile, nitromethane serves as a supporting co-solvent that improves the solubility of the organic substrates and the electrolyte, tetrabutylammonium hexafluorophosphate. The use of a constant current of 5 mA ensures a steady flux of electrons, preventing over-oxidation of the product which could lead to degradation. This controlled delivery of energy allows for the tolerance of diverse functional groups, including alkyl, cycloalkyl, and alkoxy substituents, as evidenced by the broad substrate scope explored in the patent examples. The mechanistic elegance of this approach ensures that the reaction stops precisely at the desired oxidation state, yielding the benzothiophene derivative with high fidelity and minimal formation of over-oxidized sulfone byproducts.
From an impurity control perspective, this electrocatalytic mechanism offers distinct advantages over thermal radical initiators or metal-catalyzed cross-couplings. Since the reaction does not involve metal salts, there is no risk of metal-ligand complexation or metal-induced side reactions that often plague traditional syntheses. The primary impurities are likely to be unreacted starting materials or simple dimerization products, which are generally easier to separate via standard silica gel chromatography than metal complexes. The mild room temperature conditions further suppress thermal degradation pathways, ensuring that the final product retains its structural integrity. For R&D directors, this means a more predictable impurity profile that is easier to characterize and control, facilitating faster regulatory filings. The ability to tune the reaction rate simply by adjusting the current provides an additional handle for process optimization, allowing chemists to balance reaction speed against selectivity in real-time. This level of control is rarely achievable in batch thermal processes, making the electrocatalytic method a superior choice for the synthesis of complex, sensitive pharmaceutical intermediates.
How to Synthesize Benzothiophene Derivatives Efficiently
Implementing this electrocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent to ensure reproducibility and optimal yield. The procedure involves setting up a simple undivided cell equipped with graphite felt electrodes, which provide a high surface area for efficient electrochemical turnover. The reactants, specifically the alkylphenylacetylene and the substituted benzenesulfonyl hydrazide, must be weighed out in a precise 1:3 molar ratio to drive the reaction to completion while minimizing the formation of side products. The electrolyte, tetrabutylammonium hexafluorophosphate, is added to ensure sufficient conductivity of the solution, and the specialized solvent system of HFIP and nitromethane is introduced to create the optimal medium for radical stabilization. Once the cell is assembled and purged with nitrogen to exclude oxygen, a constant current of 5 mA is applied, and the mixture is stirred at room temperature for approximately 8 hours. Following the electrolysis, the workup is straightforward, involving solvent removal via rotary evaporation and purification by flash column chromatography. For a detailed, step-by-step breakdown of the standardized operating procedure, please refer to the technical guide below.
- Prepare the electrolytic cell by adding alkylphenylacetylene and substituted benzenesulfonyl hydrazide in a 1: 3 molar ratio along with tetrabutylammonium hexafluorophosphate electrolyte.
- Add the solvent mixture consisting of hexafluoroisopropanol and nitromethane in a 47: 3 volume ratio to the reactor containing graphite felt electrodes.
- Apply a constant current of 5 mA at room temperature for 8 hours under nitrogen atmosphere, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrocatalytic technology translates into tangible strategic benefits that directly impact the bottom line and operational resilience. The most significant advantage is the complete elimination of precious metal catalysts, which removes a major source of cost volatility and supply chain risk associated with fluctuating palladium prices. Without the need for expensive metal scavengers or specialized filtration equipment to remove trace metals, the overall cost of goods sold (COGS) is significantly reduced. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during the workup phase, contributing to substantial cost savings in raw materials and waste disposal. The mild reaction conditions also imply lower energy costs, as there is no need for heating mantles or chillers to maintain extreme temperatures, allowing the process to run efficiently at ambient conditions. These factors combined create a leaner, more cost-effective manufacturing process that enhances the competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The removal of palladium catalysts and phosphorus ligands eliminates the need for costly metal scavenging resins and reduces the burden of heavy metal testing, leading to direct savings in both material and analytical costs. The simplified workup procedure requires fewer chromatography columns and less solvent, further driving down the variable costs per kilogram of product. Additionally, the use of electricity as a reagent is inherently cheaper and more sustainable than purchasing stoichiometric chemical oxidants, providing a long-term economic advantage. By streamlining the synthesis into a single pot with minimal additives, the process reduces labor hours and equipment occupancy time, maximizing facility throughput.
- Enhanced Supply Chain Reliability: Relying on electricity rather than scarce transition metals insulates the supply chain from geopolitical disruptions and price spikes in the precious metals market. The starting materials, alkylphenylacetylenes and benzenesulfonyl hydrazides, are commercially available and stable, ensuring a consistent feedstock supply for continuous production. The robustness of the reaction conditions means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or reagent quality. This reliability allows for more accurate demand forecasting and inventory planning, reducing the risk of stockouts or production delays. Moreover, the ease of scaling the electrochemical setup means that capacity can be increased rapidly by adding more cells in parallel, providing flexibility to meet surging market demand.
- Scalability and Environmental Compliance: The electrocatalytic method aligns perfectly with modern green chemistry principles, generating minimal hazardous waste and avoiding the use of toxic heavy metals that require special disposal protocols. The low E-factor of the process simplifies environmental compliance and reduces the regulatory burden associated with wastewater treatment. Scaling up electrochemical reactions is increasingly feasible with modern flow chemistry technologies, allowing for the safe transition from gram-scale laboratory synthesis to multi-kilogram commercial production. The absence of exothermic hazards associated with strong chemical oxidants enhances process safety, lowering insurance premiums and facilitating easier permitting for new manufacturing lines. This sustainable approach not only meets current environmental regulations but also future-proofs the supply chain against tightening global standards on chemical manufacturing.
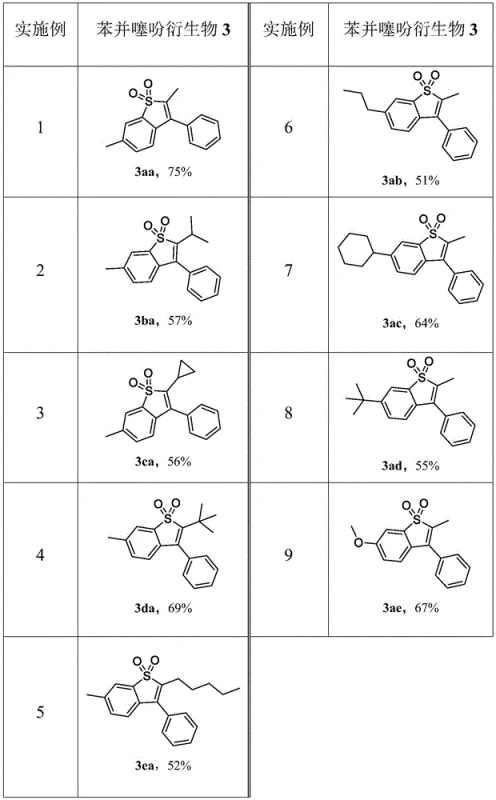
The versatility of this method is further demonstrated by the wide range of substrates successfully converted into benzothiophene derivatives, as summarized in the yield table below. The data shows consistent performance across various alkyl and aryl substitutions, confirming the robustness of the protocol for diverse chemical libraries.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled a list of frequently asked questions based on the patent data and practical implementation experience. These answers address common concerns regarding reaction scope, scalability, and product quality, providing a clear overview of what to expect when adopting this electrocatalytic route. Understanding these details is crucial for making informed decisions about process integration and resource allocation. We encourage stakeholders to review these insights closely to gauge the potential impact on their current R&D and manufacturing workflows.
Q: What are the primary advantages of this electrocatalytic method over traditional palladium-catalyzed routes?
A: The electrocatalytic method eliminates the need for expensive and toxic transition metal catalysts like palladium, operates at room temperature instead of high heat, and uses electrons as green reagents, resulting in higher purity products easier to separate.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the method utilizes simple undivided cells with graphite felt electrodes and mild conditions, making it highly suitable for commercial scale-up without the complex safety measures required for high-temperature or high-pressure reactions.
Q: What represents the typical yield range for these benzothiophene derivatives?
A: According to the patent data, the method consistently achieves high yields, with specific examples ranging from 51% to 75% depending on the substituents, demonstrating robust substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in the production of high-value pharmaceutical intermediates like benzothiophene derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We understand the critical importance of supply continuity and cost efficiency, and our team is dedicated to optimizing every step of the value chain to deliver superior results for our clients. By leveraging our expertise in green chemistry and process intensification, we can help you bring your benzothiophene-based drug candidates to market faster and more economically.
We invite you to collaborate with us to explore the full potential of this electrocatalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data, route feasibility assessments, and a comprehensive proposal that demonstrates how our capabilities can enhance your supply chain resilience. Let us be your trusted partner in navigating the complexities of modern chemical manufacturing and achieving your strategic business goals.
